- TECHNOLOGY FEATURE
The overlooked variable in animal studies: why diet makes a difference

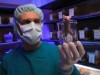
The behaviour of cuttlefish and other cephalopods can be influenced by their diets. Credit: Adam Glanzman/The Washington Post/Getty
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 605, 778-779 (2022)
doi: https://doi.org/10.1038/d41586-022-01393-9
References
Colman, R. J. et al. Science 325, 201–204 (2009).
Colman, R. J. et al. Nature Commun. 5, 3557 (2014).
Mattison, J. A. et al. Nature 489, 318–321 (2012).
Mattison, J. A. et al. Nature Commun. 8, 14063 (2017).
American Institute of Nutrition Ad Hoc Committee on Standards for Nutritional Studies. J. Nutr. 107, 1340–1348 (1977).
Tuck, C. J. et al. Sci. Rep. 10, 17784 (2020).
Stuhr, N. L. & Curran, S. P. Commun. Biol. 3, 653 (2020).

 Squeaky clean mice could be ruining research
Squeaky clean mice could be ruining research
 A mouse’s house may ruin experiments
A mouse’s house may ruin experiments
 NatureTech hub
NatureTech hub