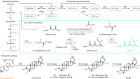
- NEWS
Biotech firm announces results from first US trial of genetically modified mosquitoes
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 604, 608-609 (2022)
doi: https://doi.org/10.1038/d41586-022-01070-x

 First genetically modified mosquitoes released in the United States
First genetically modified mosquitoes released in the United States
 US government approves 'killer' mosquitoes to fight disease
US government approves 'killer' mosquitoes to fight disease
 Florida abuzz over mosquito plan
Florida abuzz over mosquito plan