Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
doi: https://doi.org/10.1038/d41586-021-01665-w
This article is part of Nature Outlook: The Spinoff Prize 2021, an editorially independent supplement produced with the financial support of third parties. About this content.
Updates & Corrections
-
Update 13 July 2021: This article has been updated to reflect the fact that HighT-Tech won The Spinoff Prize.
References
Yao, Y. et al. Science 359, 1489–1494 (2018).

 The Spinoff Prize 2021
The Spinoff Prize 2021
 Bilitech: Miniature organs to heal damaged livers
Bilitech: Miniature organs to heal damaged livers
 C2i Genomics: A sensitive strategy for tumour surveillance
C2i Genomics: A sensitive strategy for tumour surveillance
 Cambridge Photon Technology: Solar cells that make use of wasted light
Cambridge Photon Technology: Solar cells that make use of wasted light
 GrayMatters Health: Better brain training for treating psychological conditions
GrayMatters Health: Better brain training for treating psychological conditions
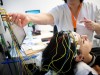 Neuronostics: Accelerating the diagnosis of epilepsy with computer modelling
Neuronostics: Accelerating the diagnosis of epilepsy with computer modelling
 ONA Therapeutics: A drug to block fat intake and combat cancer spread
ONA Therapeutics: A drug to block fat intake and combat cancer spread
 Sibylla Biotech: Turning transient structures into drug targets
Sibylla Biotech: Turning transient structures into drug targets
 The Spinoff Prize: Ones to watch
The Spinoff Prize: Ones to watch
 The Spinoff Prize 2020 finalists: where are they now?
The Spinoff Prize 2020 finalists: where are they now?