- NEWS AND VIEWS
A 4D road map for the formation of hair follicles
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 594, 500-501 (2021)
doi: https://doi.org/10.1038/d41586-021-01482-1
References
Hadjantonakis, K. & Solnica-Krezel, L. Dev. Biol. 341, 2–4 (2010).
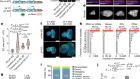
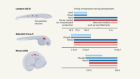
Morita, R. et al. Nature 594, 547–552 (2021).
Saxena, N., Mok, K.-W. & Rendl, M. Exp. Dermatol. 28, 332–344 (2019).
Kollar, E. J. J. Invest. Dermatol. 55, 374–378 (1970).
Millar, S. E. J. Invest. Dermatol. 118, 216–225 (2002).
Ji, S., Zhu, Z., Sun, X. & Fu, X. Signal Transduct. Target. Ther. 6, 66 (2021).
Lee, J. et al. Nature 582, 399–404 (2020).
Heitman, N., Saxena, N. & Rendl, M. Curr. Opin. Cell Biol. 55, 87–95 (2018).
Ruiz-Losada, M., Blom-Dahl, D., Córdoba, S. & Estella, C. J. Dev. Biol. 6, 17 (2018).
Tumbar, T. et al. Science 303, 359–363 (2004).
Morris, R. J. et al. Nature Biotechnol. 22, 411–417 (2004).
Fuchs, E. & Blau, H. M. Cell Stem Cell 27, 532–556 (2020).
Ouspenskaia, T., Matos, I., Mertz, A. F., Fiore, V. F. & Fuchs, E. Cell 164, 156–169 (2016).
Mok, K.-W. et al. Dev. Cell 48, 32–48 (2019).
Competing Interests
The authors declare no competing interests.

 Read the paper: Tracing the origin of hair follicle stem cells
Read the paper: Tracing the origin of hair follicle stem cells
 Relax to grow more hair
Relax to grow more hair
 Regenerative medicine could pave the way to treating baldness
Regenerative medicine could pave the way to treating baldness