- NEWS AND VIEWS
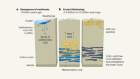
Martian water stored underground
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 552, 339-340 (2017)
doi: https://doi.org/10.1038/d41586-017-08670-y
References
Lammer, H. et al. Space Sci. Rev. 174, 113–154 (2013).
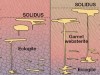
Wade, J., Dyck, B., Palin, R. M., Moore, J. D. P. & Smye, A. J. Nature 552, 391–394 (2017).
Herd, C. D. K. et al. Geochim. Cosmochim. Acta 66, 2025–2036 (2002).
Usui, T., Alexander, C. M. O’D., Wang, J., Simon, J. I. & Jones, J. H. Earth Planet. Sci. Lett. 357–358, 119–129 (2012).
Saal, A. E., Hauri, E. H., Langmuir, C. H. & Perfit, M. R. Nature 419, 451–455 (2002).
Filiberto, J. & Dasgupta, R. J. Geophys. Res. Planets 120, 109–122 (2015).
Kurokawa, H. et al. Earth Planet. Sci. Lett. 394, 179–185 (2014).
Mouginot, J., Pommerol, A., Beck, P., Kofman, W. & Clifford, S. M. Geophys. Res. Lett. 39, L02202 (2012).
Usui, T., Alexander, C. M. O’D., Wang, J., Simon, J. I. & Jones, J. H. Earth Planet. Sci. Lett. 410, 140–151 (2015).
Weiss, D. K. & Head, J. W. Icarus 288, 120–147 (2017).

 Read the paper: The divergent fates of primitive hydrospheric water on Earth and Mars
Read the paper: The divergent fates of primitive hydrospheric water on Earth and Mars
 Megafloods downsized
Megafloods downsized
 Evaporating asteroid
Evaporating asteroid