Abstract
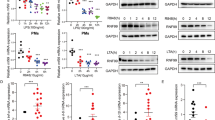
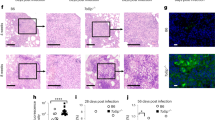
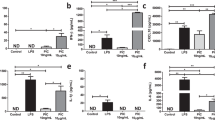
We demonstrate that Mycobacterium tuberculosis recombinant leucine-responsive regulatory protein (rLrp) inhibits lipopolysaccharide (LPS)-induced tumor necrosis factor alpha (TNF-α), interleukin-6, and interleukin-12 production and blocks the nuclear translocation of subunits of the nuclear-receptor transcription factor NF-κB (Nuclear factor-kappa B). Moreover, rLrp attenuated LPS-induced DNA binding and NF-κB transcriptional activity, which was accompanied by the degradation of inhibitory IκBα and a consequent decrease in the nuclear translocation of the NF-κB p65 subunit. RLrp interfered with the LPS-induced clustering of TNF receptor-associated factor 6 and with interleukin-1 receptor-associated kinase 1 binding to TAK1. Furthermore, rLrp did not attenuate proinflammatory cytokines or the expression of CD86 and major histocompatibility complex class-II induced by interferon-gamma in the macrophages of Toll-like receptor 2 deletion (TLR2−/−) mice and in protein kinase b (Akt)-depleted mouse cells, indicating that the inhibitory effects of rLrp were dependent on TLR2-mediated activation of the phosphatidylinositol 3-OH kinase (PI3K)/Akt pathway. RLrp could also activate the PI3K/Akt pathway by stimulating the rapid phosphorylation of PI3K, Akt, and glycogen synthase kinase 3 beta in macrophages. In addition, 19 amino acid residues in the N-terminus of rLrp were determined to be important and required for the inhibitory effects mediated by TLR2. The inhibitory function of these 19 amino acids of rLrp raises the possibility that mimetic inhibitory peptides could be used to restrict innate immune responses in situations in which prolonged TLR signaling has deleterious effects. Our study offers new insight into the inhibitory mechanisms by which the TLR2-mediated PI3K/Akt pathway ensures the transient expression of potent inflammatory mediators.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout











Similar content being viewed by others
References
Kaufmann SH, van Embden JD . Tuberculosis: a neglected disease strikes back. Trends Microbiol 1993; 1: 2–5.
Day M . Fighting talk: WHO gets tough on TB after criticism from scientists. New Sci 1998; 2127: 21 (Abstract).
Hingley-Wilson SM, Sly LM, Reiner NE, McMaster WR . The immunobiology of the mycobacterial infected macrophage. Mod Aspects Immunobiol 2000; 1: 96–101.
Fenton MJ, Vermeulen MW . Immunopathology of tuberculosis: roles of macrophages and monocytes. Infect Immun 1996; 64: 683–690.
Armstrong JA, Hart PD . Phagosome-lysosome interactions in cultured macrophages infected with virulent tubercle bacilli. Reversal of the usual nonfusion pattern and observations on bacterial survival. J Exp Med 1975; 142: 1–16.
Flynn JL, Chan J . Immunology of tuberculosis. Annu Rev Immunol 2001; 19: 93–129.
Orme IM, Cooper AM . Cytokine/chemokine cascades in immunity to tuberculosis. Immunol Today 1999; 20: 307–312.
van Crevel R, Ottenhoff TH, van der Meer JW . Innate immunity to Mycobacterium tuberculosis. Clin Microbiol Rev 2002; 15: 294–309.
Ottenhoff TH, Verreck FA, Lichtenauer-Kaligis EG, Hoeve MA, Sanal O, van Dissel JT . Genetics, cytokines and human infectious disease: lessons from weakly pathogenic mycobacteria and salmonellae. Nat Genet 2002; 32: 97–105.
Senaldi G, Yin S, Shaklee CL, Piguet PF, Mak TW, Ulich TR . Corynebacterium parvum- and Mycobacterium bovis Bacillus Calmette-Guerin-induced granuloma formation is inhibited in TNF receptor I (TNF-RI) knockout mice and by treatment with soluble TNF-RI. J Immunol 1996; 157: 5022–5026.
Roach DR, Bean AG, Demangel C, France MP, Briscoe H, Britton WJ . TNF regulates chemokine induction essential for cell recruitment, granuloma formation, and clearance of mycobacterial infection. J Immunol 2002; 168: 4620–4627.
Giacomini E, Iona E, Ferroni L, Miettinen M, Fattorini L, Orefici G et al. Infection of human macrophages and dendritic cells with Mycobacterium tuberculosis induces a differential cytokine gene expression that modulates T cell response. J Immunol 2001; 166: 7033–7041.
Nau GJ, Richmond JF, Schlesinger A, Jennings EG, Lander ES, Young RA . Human macrophage activation programs induced by bacterial pathogens. Proc Natl Acad Sci U S A 2002; 99: 1503–1508.
Koul A, Herget T, Klebl B, Ullrich A . Interplay between mycobacteria and host signalling pathways. Nat Rev Microbiol 2004; 2: 189–202.
Beltan E, Horgen L, Rastogi N . Secretion of cytokines by human macrophages upon infection by pathogenic and non-pathogenic mycobacteria. Microb Pathog 2000; 28: 313–318.
Falcone V, Bassey EB, Toniolo A, Conaldi PG, Collins FM . Differential release of tumor necrosis factor-alpha from murine peritoneal macrophages stimulated with virulent and avirulent species of mycobacteria. FEMS Immunol Med Microbiol 1994; 8: 225–232.
Brightbill HD, Libraty DH, Krutzik SR, Yang RB, Belisle JT, Bleharski JR et al. Host defense mechanisms triggered by microbial lipoproteins through toll-like receptors. Science 1999; 285: 732–736.
Imler JL, Hoffmann JA . Toll receptors in innate immunity. Trends Cell Biol 2001; 11: 304–311.
Lien E, Sellati TJ, Yoshimura A, Flo TH, Rawadi G, Finberg RW et al. Toll-like receptor 2 functions as a pattern recognition receptor for diverse bacterial products. J Biol Chem 1999; 274: 33419–33425.
Underhill DM, Ozinsky A, Smith KD, Aderem A . Toll-like receptor-2 mediates mycobacteria-induced proinflammatory signaling in macrophages. Proc Natl Acad Sci U S A 1999; 96: 14459–14463.
Bauer S, Kirschning CJ, Hacker H, Redecke V, Hausmann S, Akira S et al. Human TLR9 confers responsiveness to bacterial DNA via species-specific CpG motif recognition. Proc Natl Acad Sci U S A 2001; 98: 9237–9242.
Means TK, Lien E, Yoshimura A, Wang S, Golenbock DT, Fenton MJ . The CD14 ligands lipoarabinomannan and lipopolysaccharide differ in their requirement for Toll-like receptors. J Immunol 1999; 163: 6748–6755.
Muzio M, Ni J, Feng P, Dixit VM . IRAK (Pelle) family member IRAK-2 and MyD88 as proximal mediators of IL-1 signaling. Science 1997; 278: 1612–1615.
Suzuki N, Suzuki S, Millar DG, Unno M, Hara H, Calzascia T et al. A critical role for the innate immune signaling molecule IRAK-4 in T cell activation. Science 2006; 311: 1927–1932.
Yamamoto M, Takeda K, Akira S . TIR domain-containing adaptors define the specificity of TLR signaling. Mol Immunol 2004; 40: 861–868.
Fukao T, Tanabe M, Terauchi Y, Ota T, Matsuda S, Asano T et al. PI3K-mediated negative feedback regulation of IL-12 production in DCs. Nat Immunol 2002; 3: 875–881.
Guha M, Mackman N . The phosphatidylinositol 3-kinase-Akt pathway limits lipopolysaccharide activation of signaling pathways and expression of inflammatory mediators in human monocytic cells. J Biol Chem 2002; 277: 32124–32132.
Cooper AM, Dalton DK, Stewart TA, Griffin JP, Russell DG, Orme IM . Disseminated tuberculosis in interferon gamma gene-disrupted mice. J Exp Med 1993; 178: 2243–2247.
Flynn JL, Chan J, Triebold KJ, Dalton DK, Stewart TA, Bloom BR . An essential role for interferon gamma in resistance to Mycobacterium tuberculosis infection. J Exp Med 1993; 178: 2249–2254.
Schneeberger EE, DeFerrari M, Skoskiewicz MJ, Russell PS, Colvin RB . Induction of MHC-determined antigens in the lung by interferon-gamma. Lab Invest 1986; 55: 138–144.
Gorvel JP, Moreno E . Brucella intracellular life: from invasion to intracellular replication. Vet Microbiol 2002; 90: 281–297.
Kohler S, Michaux-Charachon S, Porte F, Ramuz M, Liautard JP . What is the nature of the replicative niche of a stealthy bug named Brucella? Trends Microbiol 2003; 11: 215–219.
Fruth U, Solioz N, Louis JA . Leishmania major interferes with antigen presentation by infected macrophages. J Immunol 1993; 150: 1857–1864.
Noss EH, Harding CV, Boom WH . Mycobacterium tuberculosis inhibits MHC class II antigen processing in murine bone marrow macrophages. Cell Immunol 2000; 201: 63–74.
Prina E, Jouanne C, de Souza Lao S, Szabo A, Guillet JG, Antoine JC . Antigen presentation capacity of murine macrophages infected with Leishmania amazonensis amastigotes. J Immunol 1993; 151: 2050–2061.
Noss EH, Pai RK, Sellati TJ, Radolf JD, Belisle J, Golenbock DT et al. Toll-like receptor 2-dependent inhibition of macrophage class II MHC expression and antigen processing by 19-kDa lipoprotein of Mycobacterium tuberculosis. J Immunol 2001; 167: 910–918.
Pai RK, Convery M, Hamilton TA, Boom WH, Harding CV . Inhibition of IFN-gamma-induced class II transactivator expression by a 19-kDa lipoprotein from Mycobacterium tuberculosis: a potential mechanism for immune evasion. J Immunol 2003; 171: 175–184.
Gehring AJ, Dobos KM, Belisle JT, Harding CV, Boom WH . Mycobacterium tuberculosis LprG (Rv1411c): a novel TLR-2 ligand that inhibits human macrophage class II MHC antigen processing. J Immunol 2004; 173: 2660–2668.
Pecora ND, Gehring AJ, Canaday DH, Boom WH, Harding CV . Mycobacterium tuberculosis LprA is a lipoprotein agonist of TLR2 that regulates innate immunity and APC function. J Immunol 2006; 177: 422–429.
Chu RS, Askew D, Noss EH, Tobian A, Krieg AM, Harding CV . CpG oligodeoxynucleotides down-regulate macrophage class II MHC antigen processing. J Immunol 1999; 163: 1188–1194.
Tobian AA, Potter NS, Ramachandra L, Pai RK, Convery M, Boom WH et al. Alternate class I MHC antigen processing is inhibited by Toll-like receptor signaling pathogen-associated molecular patterns: Mycobacterium tuberculosis 19-kDa lipoprotein, CpG DNA, and lipopolysaccharide. J Immunol 2003; 171: 1413–1422.
Pathak SK, Basu S, Basu KK, Banerjee A, Pathak S, Bhattacharyya A et al. Direct extracellular interaction between the early secreted antigen ESAT-6 of Mycobacterium tuberculosis and TLR2 inhibits TLR signaling in macrophages. Nat Immunol 2007; 8: 610–618.
Deng W, Wang H, Xie J . Regulatory and pathogenesis roles of Mycobacterium Lrp/AsnC family transcriptional factors. J Cell Biochem 2011; 112: 2655–2662.
Dey A, Ramachandran R . Cloning, overexpression, purification and preliminary X-ray analysis of a feast/famine regulatory protein (Rv2779c) from Mycobacterium tuberculosis H37Rv. Acta Crystallogr F Struct Biol Commun 2014; 70: 97–100.
Yu DH, Hu XD, Cai H . A combined DNA vaccine encoding BCSP31, SOD, and L7/L12 confers high protection against Brucella abortus 2308 by inducing specific CTL responses. DNA Cell Biol 2007; 26: 435–443.
Chen ST, Li JY, Zhang Y, Gao X, Cai H . Recombinant MPT83 derived from Mycobacterium tuberculosis induces cytokine production and upregulates the function of mouse macrophages through TLR2. J Immunol 2012; 188: 668–677.
Khan N, Ghousunnissa S, Jegadeeswaran SM, Thiagarajan D, Hasnain SE, Mukhopadhyay S . Anti-B7-1/B7-2 antibody elicits innate-effector responses in macrophages through NF-kappaB-dependent pathway. Int Immunol 2007; 19: 477–486.
Ashall L, Horton CA, Nelson DE, Paszek P, Harper CV, Sillitoe K et al. Pulsatile stimulation determines timing and specificity of NF-kappaB-dependent transcription. Science 2009; 324: 242–246.
Lawrence T, Gilroy DW, Colville-Nash PR, Willoughby DA . Possible new role for NF-kappaB in the resolution of inflammation. Nat Med 2001; 7: 1291–1297.
Murphy TL, Cleveland MG, Kulesza P, Magram J, Murphy KM . Regulation of interleukin 12 p40 expression through an NF-kappa B half-site. Mol Cell Biol 1995; 15: 5258–5267.
Joyce DA, Gimblett G, Steer JH . Targets of glucocorticoid action on TNF-alpha release by macrophages. Inflamm Res 2001; 50: 337–340.
Xiong Y, Qiu F, Piao W, Song C, Wahl LM, Medvedev AE . Endotoxin tolerance impairs IL-1 receptor-associated kinase (IRAK) 4 and TGF-beta-activated kinase 1 activation, K63-linked polyubiquitination and assembly of IRAK1, TNF receptor-associated factor 6, and IkappaB kinase gamma and increases A20 expression. J Biol Chem 2011; 286: 7905–7916.
Qian Y, Commane M, Ninomiya-Tsuji J, Matsumoto K, Li X . IRAK-mediated translocation of TRAF6 and TAB2 in the interleukin-1-induced activation of NFkappa B. J Biol Chem 2001; 276: 41661–41667.
Takaesu G, Ninomiya-Tsuji J, Kishida S, Li X, Stark GR, Matsumoto K . Interleukin-1 (IL-1) receptor-associated kinase leads to activation of TAK1 by inducing TAB2 translocation in the IL-1 signaling pathway. Mol Cell Biol 2001; 21: 2475–2484.
Cantley LC . The phosphoinositide 3-kinase pathway. Science 2002; 296: 1655–1657.
Gutierrez MG, Master SS, Singh SB, Taylor GA, Colombo MI, Deretic V . Autophagy is a defense mechanism inhibiting BCG and Mycobacterium tuberculosis survival in infected macrophages. Cell 2004; 119: 753–766.
Singh CR, Moulton RA, Armitige LY, Bidani A, Snuggs M, Dhandayuthapani S et al. Processing and presentation of a mycobacterial antigen 85B epitope by murine macrophages is dependent on the phagosomal acquisition of vacuolar proton ATPase and in situ activation of cathepsin D. J Immunol 2006; 177: 3250–3259.
Holscher C, Atkinson RA, Arendse B, Brown N, Myburgh E, Alber G et al. A protective and agonistic function of IL-12p40 in mycobacterial infection. J Immunol 2001; 167: 6957–6966.
Takeda K, Kaisho T, Akira S . Toll-like receptors. Annu Rev Immunol 2003; 21: 335–376.
Stauffer F, Petrow EP, Burgmann H, Graninger W, Georgopoulos A . Release of TNF alpha and IL6 from human monocytes infected with Mycobacterium kansasii: a comparison to Mycobacterium avium. Infection 1994; 22: 326–329.
Fattorini L, Xiao Y, Li B, Santoro C, Ippoliti F, Orefici G . Induction of IL-1 beta, IL-6, TNF-alpha, GM-CSF and G-CSF in human macrophages by smooth transparent and smooth opaque colonial variants of Mycobacterium avium. J Med Microbiol 1994; 40: 129–133.
Egan LJ, Mays DC, Huntoon CJ, Bell MP, Pike MG, Sandborn WJ et al. Inhibition of interleukin-1-stimulated NF-kappaB RelA/p65 phosphorylation by mesalamine is accompanied by decreased transcriptional activity. J Biol Chem 1999; 274: 26448–26453.
Sakurai H, Chiba H, Miyoshi H, Sugita T, Toriumi W . IkappaB kinases phosphorylate NF-kappaB p65 subunit on serine 536 in the transactivation domain. J Biol Chem 1999; 274: 30353–30356.
Akira S, Takeda K . Toll-like receptor signalling. Nat Rev Immunol 2004; 4: 499–511.
Yoshimura S, Bondeson J, Brennan FM, Foxwell BM, Feldmann M . Role of NFkappaB in antigen presentation and development of regulatory T cells elucidated by treatment of dendritic cells with the proteasome inhibitor PSI. Eur J Immunol 2001; 31: 1883–1893.
Brown AM, Linhoff MW, Stein B, Wright KL, Baldwin AS Jr, Basta PV et al. Function of NF-kappa B/Rel binding sites in the major histocompatibility complex class II invariant chain promoter is dependent on cell-specific binding of different NF-kappa B/Rel subunits. Mol Cell Biol 1994; 14: 2926–2935.
Aliprantis AO, Yang RB, Mark MR, Suggett S, Devaux B, Radolf JD et al. Cell activation and apoptosis by bacterial lipoproteins through toll-like receptor-2. Science 1999; 285: 736–739.
Hou L, Sasaki H, Stashenko P . Toll-like receptor 4-deficient mice have reduced bone destruction following mixed anaerobic infection. Infect Immun 2000; 68: 4681–4687.
Cook DN, Pisetsky DS, Schwartz DA . Toll-like receptors in the pathogenesis of human disease. Nat Immunol 2004; 5: 975–979.
Kobayashi K, Hernandez LD, Galan JE, Janeway CA Jr, Medzhitov R, Flavell RA . IRAK-M is a negative regulator of Toll-like receptor signaling. Cell 2002; 110: 191–202.
Han J, Ulevitch RJ . Limiting inflammatory responses during activation of innate immunity. Nat Immunol 2005; 6: 1198–1205.
Choi KC, Lee YS, Lim S, Choi HK, Lee CH, Lee EK et al. Smad6 negatively regulates interleukin 1-receptor-Toll-like receptor signaling through direct interaction with the adaptor Pellino-1. Nat Immunol 2006; 7: 1057–1065.
Harte MT, Haga IR, Maloney G, Gray P, Reading PC, Bartlett NW et al. The poxvirus protein A52R targets Toll-like receptor signaling complexes to suppress host defense. J Exp Med 2003; 197: 343–351.
Pai RK, Convery M, Hamilton TA, Boom WH, Harding CV . Inhibition of IFN-gamma-induced class II transactivator expression by a 19-kDa lipoprotein from Mycobacterium tuberculosis: a potential mechanism for immune evasion. J Immunol 2003; 171: 175–184.
Neyrolles O, Gould K, Gares MP, Brett S, Janssen R, O’Gaora P et al. Lipoprotein access to MHC class I presentation during infection of murine macrophages with live mycobacteria. J Immunol 2001; 166: 447–457.
Ozinsky A, Underhill DM, Fontenot JD, Hajjar AM, Smith KD, Wilson CB et al. The repertoire for pattern recognition of pathogens by the innate immune system is defined by cooperation between toll-like receptors. Proc Natl Acad Sci U S A 2000; 97: 13766–13771.
Underhill DM, Ozinsky A, Hajjar AM, Stevens A, Wilson CB, Bassetti M et al. The Toll-like receptor 2 is recruited to macrophage phagosomes and discriminates between pathogens. Nature 1999; 401: 811–815.
Sing A, Rost D, Tvardovskaia N, Roggenkamp A, Wiedemann A, Kirschning CJ et al. Yersinia V-antigen exploits toll-like receptor 2 and CD14 for interleukin 10-mediated immunosuppression. J Exp Med 2002; 196: 1017–1024.
Fukao T, Koyasu S . PI3K and negative regulation of TLR signaling. Trends Immunol 2003; 24: 358–363.
Arbibe L, Mira JP, Teusch N, Kline L, Guha M, Mackman N et al. Toll-like receptor 2-mediated NF-kappa B activation requires a Rac1-dependent pathway. Nat Immunol 2000; 1: 533–540.
Martin M, Rehani K, Jope RS, Michalek SM . Toll-like receptor-mediated cytokine production is differentially regulated by glycogen synthase kinase 3. Nat Immunol 2005; 6: 777–784.
Hazeki K, Kinoshita S, Matsumura T, Nigorikawa K, Kubo H, Hazeki O . Opposite effects of wortmannin and 2-(4-morpholinyl)-8-phenyl-1(4H)-benzopyran-4-one hydrochloride on toll-like receptor-mediated nitric oxide production: negative regulation of nuclear factor-{kappa}B by phosphoinositide 3-kinase. Mol Pharmacol 2006; 69: 1717–1724.
Fukao T, Yamada T, Tanabe M, Terauchi Y, Ota T, Takayama T et al. Selective loss of gastrointestinal mast cells and impaired immunity in PI3K-deficient mice. Nat Immunol 2002; 3: 295–304.
Martin M, Schifferle RE, Cuesta N, Vogel SN, Katz J, Michalek SM . Role of the phosphatidylinositol 3 kinase-Akt pathway in the regulation of IL-10 and IL-12 by Porphyromonas gingivalis lipopolysaccharide. J Immunol 2003; 171: 717–725.
Wang H, Brown J, Martin M . Glycogen synthase kinase 3: a point of convergence for the host inflammatory response. Cytokine 2011; 53: 130–140.
Imai K, Kurita-Ochiai T, Ochiai K . Mycobacterium bovis bacillus Calmette-Guerin infection promotes SOCS induction and inhibits IFN-gamma-stimulated JAK/STAT signaling in J774 macrophages. FEMS Immunol Med Microbiol 2003; 39: 173–180.
Qasimi P, Ming-Lum A, Ghanipour A, Ong CJ, Cox ME, Ihle J et al. Divergent mechanisms utilized by SOCS3 to mediate interleukin-10 inhibition of tumor necrosis factor alpha and nitric oxide production by macrophages. J Biol Chem 2006; 281: 6316–6324.
Qin H, Wilson CA, Roberts KL, Baker BJ, Zhao X, Benveniste EN . IL-10 inhibits lipopolysaccharide-induced CD40 gene expression through induction of suppressor of cytokine signaling-3. J Immunol 2006; 177: 7761–7771.
Chowdhury RP, Saraswathi R, Chatterji D . Mycobacterial stress regulation: the Dps “twin sister” relationship. IUBMB Life 2010; 62: 67–77.
Schorey JS, Cooper AM . Macrophage signalling upon mycobacterial infection: the MAP kinases lead the way. Cell Microbiol 2003; 5: 133–142.
Smeulders MJ, Keer J, Speight RA, Williams HD . Adaptation of Mycobacterium smegmatis to stationary phase. J Bacteriol 1999; 181: 270–283.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Cellular & Molecular Immunology’s website (http://www.nature.com/cmi).
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, Y., Li, JY., Chen, ST. et al. The rLrp of Mycobacterium tuberculosis inhibits proinflammatory cytokine production and downregulates APC function in mouse macrophages via a TLR2-mediated PI3K/Akt pathway activation-dependent mechanism. Cell Mol Immunol 13, 729–745 (2016). https://doi.org/10.1038/cmi.2015.58
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2015.58
Keywords
This article is cited by
-
Mycobacterium tuberculosis Rv1043c regulates the inflammatory response by inhibiting the phosphorylation of TAK1
International Microbiology (2023)
-
Bacterial inhibition of Fas-mediated killing promotes neuroinvasion and persistence
Nature (2022)
-
Comprehensive lipid and lipid-related gene investigations of host immune responses to characterize metabolism-centric biomarkers for pulmonary tuberculosis
Scientific Reports (2022)
-
Integrative genomics of the mammalian alveolar macrophage response to intracellular mycobacteria
BMC Genomics (2021)
-
The Lrp of Mycobacterium tuberculosis regulates the innate immune response of macrophages
Cellular & Molecular Immunology (2018)