Abstract
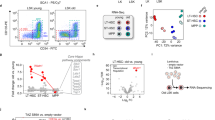
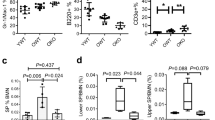
The differentiation of dendritic cells (DC) is affected by the aging process. However, the molecular mechanisms responsible for the alteration of DC development in aged mice have not been clarified. Recently, Wnt5a was reported to be an important aging-related molecule in hematopoietic systems. Here, we hypothesized that the increased expression of Wnt5a in aged hematopoietic precursors led to deficient DC differentiation in aged mice. The percentages and cell numbers of plasmacytoid DC (pDC) and CD172a−CD8α+conventional DC (cDC) were decreased in aged mice compared to young mice. Further analysis indicated that the hematopoietic precursors that gave rise to DC, including Flt3+ lymphoid-primed multipotent precursors (LMPP), common lymphoid progenitors (CLP) and common DC precursors (CDP), were all decreased in the bone marrow of aged mice. Overexpression of Wnt5a in hematopoietic precursors strongly affected the differentiation of cDC and pDC in vivo. Treatment of hematopoietic stem cells (HSC) with Wnt5a led to a significant decrease in the differentiation of the LMPP, CLP and CDP populations that was similar to the decrease observed in the bone marrow (BM) HSC of aged mice. Molecular studies demonstrated that Wnt5a negatively regulated the expression of an array of genes important for DC differentiation, including Flt3, Gfi-1, Ikaros, Bcl11a, and IL-7R, by activating the Wnt5a-Cdc42 pathway. Finally, we rejuvenated DC differentiation from aged precursors by blocking the non-canonical Wnt pathway. Our study identified the key roles of the non-canonical Wnt pathway in DC differentiation and DC aging.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Merad M, Sathe P, Helft J, Miller J, Mortha A . The dendritic cell lineage: ontogeny and function of dendritic cells and their subsets in the steady state and the inflamed setting . Annu Rev Immunol 2013 ; 31 : 563 – 604.
Mildner A, Jung S . Development and function of dendritic cell subsets . Immunity 2014 ; 40 : 642 – 656.
Wu L, Liu YJ . Development of dendritic-cell lineages . Immunity 2007 ; 26 : 741 – 750.
Shortman K, Naik SH . Steady-state and inflammatory dendritic-cell development . Nat Rev Immunol 2007 ; 7 : 19 – 30.
Adolfsson J, Månsson R, Buza-Vidas N, Hultquist A, Liuba K, Jensen CT et al . Identification of Flt3+ lympho-myeloid stem cells lacking erythro-megakaryocytic potential a revised road map for adult blood lineage commitment . Cell 2005 ; 121 : 295 – 306.
Liu K, Nussenzweig MC . Origin and development of dendritic cells . Immunol Rev 2010 ; 234 : 45 – 54.
Onai N, Obata-Onai A, Schmid MA, Ohteki T, Jarrossay D, Manz MG . Identification of clonogenic common Flt3+M-CSFR+ plasmacytoid and conventional dendritic cell progenitors in mouse bone marrow . Nat Immunol 2007 ; 8 : 1207 – 1216.
Sathe P, Vremec D, Wu L, Corcoran L, Shortman K . Convergent differentiation: myeloid and lymphoid pathways to murine plasmacytoid dendritic cells . Blood 2013 ; 121 : 11 – 19.
Chen YL, Chen TT, Pai LM, Wesoly J, Bluyssen HA, Lee CK . A type I IFN-Flt3 ligand axis augments plasmacytoid dendritic cell development from common lymphoid progenitors . J Exp Med 2013 ; 210 : 2515 – 2522.
Onai N, Kurabayashi K, Hosoi-Amaike M, Toyama-Sorimachi N, Matsushima K, Inaba K et al . A clonogenic progenitor with prominent plasmacytoid dendritic cell developmental potential . Immunity 2013 ; 38 : 943 – 957.
Shortman K, Sathe P . Another heritage for plasmacytoid dendritic cells . Immunity 2013 ; 38 : 845 – 846.
Bogunovic M, Ginhoux F, Helft J, Shang L, Hashimoto D, Greter M et al . Origin of the lamina propria dendritic cell network . Immunity 2009 ; 31 : 513 – 525.
Fancke B, Suter M, Hochrein H, O’Keeffe M . M-CSF: a novel plasmacytoid and conventional dendritic cell poietin . Blood 2008 ; 111 : 150 – 159.
Karsunky H, Merad M, Cozzio A, Weissman IL, Manz MG . Flt3 ligand regulates dendritic cell development from Flt3+ lymphoid and myeloid-committed progenitors to Flt3+ dendritic cells in vivo . J Exp Med 2003 ; 198 : 305 – 313.
Waskow C, Liu K, Darrasse-Jèze G, Guermonprez P, Ginhoux F, Merad M et al . The receptor tyrosine kinase Flt3 is required for dendritic cell development in peripheral lymphoid tissues . Nat Immunol 2008 ; 9 : 676 – 683.
Brasel K, De Smedt T, Smith JL, Maliszewski CR . Generation of murine dendritic cells from flt3-ligand-supplemented bone marrow cultures . Blood 2000 ; 96 : 3029 – 3039.
McKenna HJ1, Stocking KL, Miller RE, Brasel K, De Smedt T, Maraskovsky E et al . Mice lacking flt3 ligand have deficient hematopoiesis affecting hematopoietic progenitor cells, dendritic cells, and natural killer cells . Blood 2000 ; 95 : 3489 – 3497.
Vogt TK, Link A, Perrin J, Finke D, Luther SA . Novel function for interleukin-7 in dendritic cell development . Blood 2009 ; 113 : 3961 – 3968.
Staal FJ, Luis TC, Tiemessen MM . WNT signalling in the immune system: WNT is spreading its wings . Nat Rev Immunol 2008 ; 8 : 581 – 593.
Logan CY, Nusse R . The Wnt signaling pathway in development and disease . Nat Rev Immunol 2008 ; 8 : 581 – 593.
Luis TC, Naber BA, Roozen PP, Brugman MH, de Haas EF, Ghazvini M et al . Canonical wnt signaling regulates hematopoiesis in a dosage-dependent fashion . Cell Stem Cell 2011 ; 9 : 345 – 356.
Sugimura R, He XC, Venkatraman A, Arai F, Box A, Semerad C et al . Noncanonical Wnt signaling maintains hematopoietic stem cells in the niche . Cell 2012 ; 150 : 351 – 365.
Semerad CL, Mercer EM, Inlay MA, Weissman IL, Murre C . E2A proteins maintain the hematopoietic stem cell pool and promote the maturation of myelolymphoid and myeloerythroid progenitors . Proc Natl Acad Sci U S A 2009 ; 106 : 1930 – 1935.
Nemeth MJ, Topol L, Anderson SM, Yang Y, Bodine DM . Wnt5a inhibits canonical Wnt signaling in hematopoietic stem cells and enhances repopulation . Proc Natl Acad Sci U S A 2007 ; 104 : 15436 – 15441.
Florian MC, Nattamai KJ, Dörr K, Marka G, Uberle B, Vas V et al . A canonical to non-canonical Wnt signalling switch in haematopoietic stem-cell ageing . Nature 2013 ; 503 : 392 – 396.
Oderup C, LaJevic M, Butcher EC . Canonical and noncanonical Wnt proteins program dendritic cell responses for tolerance . J Immunol 2013 ; 190 : 6126 – 6134.
Zhou J, Cheng P, Youn JI, Cotter MJ, Gabrilovich DI . Notch and wingless signaling cooperate in regulation of dendritic cell differentiation . Immunity 2009 ; 30 : 845 – 859.
Wong CP, Magnusson KR, Ho E . Aging is associated with altered dendritic cells subset distribution and impaired proinflammatory cytokine production . Exp Gerontol 2010 ; 45 : 163 – 169.
Jing Y, Shaheen E, Drake RR, Chen N, Gravenstein S, Deng Y . Aging is associated with a numerical and functional decline in plasmacytoid dendritic cells, whereas myeloid dendritic cells are relatively unaltered in human peripheral blood . Hum Immunol 2009 ; 70 : 777 – 784.
Agrawal A, Gupta S . Impact of aging on dendritic cell functions in humans . Ageing Res Rev 2011 ; 10 : 336 – 345.
Rathinam C, Geffers R, Yücel R, Buer J, Welte K, Möröy T et al . The transcriptional repressor Gfi1 controls STAT3-dependent dendritic cell development and function . Immunity 2005 ; 22 : 717 – 728.
Wu L, Nichogiannopoulou A, Shortman K, Georgopoulos K . Cell-autonomous defects in dendritic cell populations of Ikaros mutant mice point to a developmental relationship with the lymphoid lineage . Immunity 1997 ; 7 : 483 – 492.
Wu X, Satpathy AT, Kc W, Liu P, Murphy TL, Murphy KM . Bcl11a controls Flt3 expression in early hematopoietic progenitors and is required for pDC development in vivo . PLoS One 201 ; 8 : e64800.
Carotta S, Dakic A, D’Amico A, Pang SH, Greig KT, Nutt SL et al . The transcription factor PU.1 controls dendritic cell development and Flt3 cytokine receptor expression in a dose-dependent manner . Immunity 2010 ; 32 : 628 – 641.
Sathe P, Metcalf D, Vremec D, Naik SH, Langdon WY, Huntington ND et al . Lymphoid tissue and plasmacytoid dendritic cells and macrophages do not share a common macrophage-dendritic cell-restricted progenitor . Immunity 2014 ; 41 : 104 – 115.
Rossi DJ, Bryder D, Zahn JM, Ahlenius H, Sonu R, Wagers AJ et al . Cell intrinsic alterations underlie hematopoietic stem cell aging . Proc Natl Acad Sci U S A 2005 ; 102 : 9194 – 9199.
Peterson JR, Lebensohn AM, Pelish HE, Kirschner MW . Biochemical suppression of small-molecule inhibitors: a strategy to identify inhibitor targets and signaling pathway components . Chem Biol 2006 ; 13 : 443 – 452.
Florian MC, Dörr K, Niebel A, Daria D, Schrezenmeier H, Rojewski M et al . Cdc42 activity regulates hematopoietic stem cell aging and rejuvenation . Cell Stem Cell 2012 ; 10 : 520 – 530.
Shurin MR, Shurin GV, Chatta GS . Aging and the dendritic cell system: implications for cancer . Crit Rev Oncol Hematol 2007 ; 64 : 90 – 105.
Agrawal A, Sridharan A, Prakash S, Agrawal H . Dendritic cells and aging: consequences for autoimmunity . Expert Rev Clin Immunol 2012 ; 8 : 73 – 80.
Grolleau-Julius A, Harning EK, Abernathy LM, Yung RL . Impaired dendritic cell function in aging leads to defective antitumor immunity . Cancer Res 2008 ; 68 : 6341 – 6349.
Montecino-Rodriguez E, Berent-Maoz B, Dorshkind K . Causes, consequences, and reversal of immune system aging . J Clin Invest 2013 ; 123 : 958 – 965.
Acknowledgements
This study was supported by a Key Project Grant from the National Natural Science Foundation of China (No. 31330027), a Tsinghua University Initiative Scientific Research Program Research Fund (No. 20111080963) and the Tsinghua-Peking Center for Life Sciences.
Author information
Authors and Affiliations
Additional information
Supplementary Information accompanies the paper on Cellular & Molecular Immunology’s website (http://www.nature.com/cmi).
Supplementary information
Rights and permissions
About this article
Cite this article
Xiao, J., Zhou, H., Wu, N. et al. The non-canonical Wnt pathway negatively regulates dendritic cell differentiation by inhibiting the expansion of Flt3+ lymphocyte-primed multipotent precursors. Cell Mol Immunol 13, 593–604 (2016). https://doi.org/10.1038/cmi.2015.39
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2015.39
Keywords
This article is cited by
-
Trait − driven analysis of the 2p15p16.1 microdeletion syndrome suggests a complex pattern of interactions between candidate genes
Genes & Genomics (2023)
-
Abnormal level of CUL4B-mediated histone H2A ubiquitination causes disruptive HOX gene expression
Epigenetics & Chromatin (2019)
-
The non-canonical Wnt pathway leads to aged dendritic cell differentiation
Cellular & Molecular Immunology (2018)
-
p38α has an important role in antigen cross-presentation by dendritic cells
Cellular & Molecular Immunology (2018)