Abstract
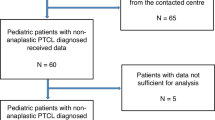
Allogeneic hematopoietic stem cell transplantation (allo-SCT) is a treatment option for relapsed anaplastic large cell lymphoma (ALCL) in children, but reports on its efficacy in this disease are still limited. We analyzed data concerning 34 patients under 18 years of age prospectively registered in the French SFGM-TC database, who had undergone an allo-SCT for the treatment of ALK+ ALCL between 1993 and 2011. At transplant, 28 patients (82.4%) were in CR, whereas 6 exhibited detectable disease. Conditioning regimens were mostly myelo-ablative (n=31). With a median follow-up of 6 years, 5-year overall and event-free survival rates were 70% (SE=8%) and 58% (SE=9%), respectively. The 5-year cumulative incidence of relapse and treatment-related mortality was 18% (SE=7%) and 24% (SE=8%), respectively. Six patients had relapsed (median time, 141 days (35–235)). A durable CR had been obtained in 4/6 patients after injection of donor lymphocytes (n=1) or vinblastine-corticosteroid therapy (n=3). Ten patients had died, eight due to transplant toxicity and two due to progressive disease. Allo-SCT is an efficient treatment for pediatric patients with high-risk relapsed ALK+ ALCL. However, the overall morbidity of allo-SCT raises questions about its place, given the efficacy of targeted agents currently under development in this disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Wright D, McKeever P, Carter R . Childhood non-Hodgkin lymphomas in the United Kingdom: findings from the UK Children’s Cancer Study Group. J Clin Pathol 1997; 50: 128–134.
Le Deley M-C, Rosolen A, Williams DM, Horibe K, Wrobel G, Attarbaschi A et al. Vinblastine in children and adolescents with high-risk anaplastic large-cell lymphoma: results of the randomized ALCL99-vinblastine trial. J Clin Oncol 2010; 28: 3987–3993.
Brugières L, Quartier P, Le Deley MC, Pacquement H, Perel Y, Bergeron C et al. Relapses of childhood anaplastic large-cell lymphoma: treatment results in a series of 41 children-a report from the French Society of Pediatric Oncology. Ann Oncol 2000; 11: 53–58.
Woessmann W, Peters C, Lenhard M, Burkhardt B, Sykora K-W, Dilloo D et al. Allogeneic haematopoietic stem cell transplantation in relapsed or refractory anaplastic large cell lymphoma of children and adolescents-a Berlin-Frankfurt-Münster group report. Br J Haematol. 2006; 133: 176–182.
Gross TG, Hale GA, He W, Camitta BM, Sanders JE, Cairo MS et al. Hematopoietic stem cell transplantation for refractory or recurrent non-Hodgkin lymphoma in children and adolescents. Biol Blood Marrow Transplant 2010; 16: 223–230.
Attarbaschi A, Dworzak M, Steiner M, Urban C, Fink F-M, Reiter A et al. Outcome of children with primary resistant or relapsed non-Hodgkin lymphoma and mature B-cell leukemia after intensive first-line treatment: a population-based analysis of the Austrian Cooperative Study Group. Pediatr Blood Cancer 2005; 44: 70–76.
Mori T, Kiyokawa N, Shimada H, Miyauchi J, Fujimoto J . Anaplastic large cell lymphoma in Japanese children: retrospective analysis of 34 patients diagnosed at the National Research Institute for Child Health and Development. Br J Haematol 2003; 121: 94–96.
Cesaro S, Pillon M, Visintin G, Putti MC, Gazzola MV, D’Amore E et al. Unrelated bone marrow transplantation for high-risk anaplastic large cell lymphoma in pediatric patients: a single center case series. Eur J Haematol 2005; 75: 22–26.
Fukano R, Mori T, Kobayashi R, Mitsui T, Fujita N, Iwasaki F et al. Haematopoietic stem cell transplantation for relapsed or refractory anaplastic large cell lymphoma: a study of children and adolescents in Japan. Br J Haematol 2014; 168: 557–563.
Woessmann W, Zimmermann M, Lenhard M, Burkhardt B, Rossig C, Kremens B et al. Relapsed or refractory anaplastic large-cell lymphoma in children and adolescents after Berlin-Frankfurt-Muenster (BFM)-type first-line therapy: a BFM-group study. J Clin Oncol 2011; 29: 3065–3071.
Jaffe ES . Anaplastic large cell lymphoma: the shifting sands of diagnostic hematopathology. Mod Pathol 2001; 14: 219–228.
Brugières L, Deley MC, Pacquement H, Meguerian-Bedoyan Z, Terrier-Lacombe MJ, Robert A et al. CD30(+) anaplastic large-cell lymphoma in children: analysis of 82 patients enrolled in two consecutive studies of the French Society of Pediatric Oncology. Blood 1998; 92: 3591–3598.
Kim HT . Cumulative incidence in competing risks data and competing risks regression analysis. Clin Cancer Res. 2007; 13 (2 Pt 1): 559–565.
Fine JP, Gray RJ . A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc 1999; 94: 496–509.
Giulino-Roth L, Ricafort R, Kernan NA, Small TN, Trippett TM, Steinherz PG et al. Ten-year follow-up of pediatric patients with non-Hodgkin lymphoma treated with allogeneic or autologous stem cell transplantation. Pediatr Blood Cancer 2013; 60: 2018–2024.
Pulford K, Falini B, Banham AH, Codrington D, Roberton H, Hatton C et al. Immune response to the ALK oncogenic tyrosine kinase in patients with anaplastic large-cell lymphoma. Blood 2000; 96: 1605–1607.
Ait-Tahar K, Cerundolo V, Banham AH, Hatton C, Blanchard T, Kusec R et al. B and CTL responses to the ALK protein in patients with ALK-positive ALCL. Int J Cancer 2006; 118: 688–695.
Ait-Tahar K, Damm-Welk C, Burkhardt B, Zimmermann M, Klapper W, Reiter A et al. Correlation of the autoantibody response to the ALK oncoantigen in pediatric anaplastic lymphoma kinase-positive anaplastic large cell lymphoma with tumor dissemination and relapse risk. Blood 2010; 115: 3314–3319.
Passoni L, Gallo B, Biganzoli E, Stefanoni R, Massimino M, Di Nicola M et al. In vivo T-cell immune response against anaplastic lymphoma kinase in patients with anaplastic large cell lymphomas. Haematologica 2006; 91: 48–55.
Chiarle R, Martinengo C, Mastini C, Ambrogio C, D’Escamard V, Forni G et al. The anaplastic lymphoma kinase is an effective oncoantigen for lymphoma vaccination. Nat Med 2008; 14: 676–680.
Ballen KK, Gluckman E, Broxmeyer HE . Umbilical cord blood transplantation: the first 25 years and beyond. Blood 2013; 122: 491–498.
Mateos MK, O’Brien TA, Oswald C, Gabriel M, Ziegler DS, Cohn RJ et al. Transplant-related mortality following allogeneic hematopoeitic stem cell transplantation for pediatric acute lymphoblastic leukemia: 25-year retrospective review. Pediatr Blood Cancer 2013; 60: 1520–1527.
Satwani P, Cooper N, Rao K, Veys P, Amrolia P . Reduced intensity conditioning and allogeneic stem cell transplantation in childhood malignant and nonmalignant diseases. Bone Marrow Transplant 2008; 41: 173–182.
Pulsipher MA, Boucher KM, Wall D, Frangoul H, Duval M, Goyal RK et al. Reduced-intensity allogeneic transplantation in pediatric patients ineligible for myeloablative therapy: results of the Pediatric Blood and Marrow Transplant Consortium Study ONC0313. Blood 2009; 114: 1429–1436.
Ohta H, Kusuki S, Yoshida H, Sato E, Hashii Y, Ozono K . Allogeneic hematopoietic stem cell transplantation with reduced intensity conditioning for a child with recurrent anaplastic large cell lymphoma. Int J Hematol 2010; 92: 190–193.
Brugières L, Pacquement H, Le Deley M-C, Leverger G, Lutz P, Paillard C et al. Single-drug vinblastine as salvage treatment for refractory or relapsed anaplastic large-cell lymphoma: a report from the French Society of Pediatric Oncology. J Clin Oncol 2009; 27: 5056–5061.
Williams DM, Hobson R, Imeson J, Gerrard M, McCarthy K, Pinkerton CR et al. Anaplastic large cell lymphoma in childhood: analysis of 72 patients treated on The United Kingdom Children’s Cancer Study Group chemotherapy regimens. Br J Haematol 2002; 117: 812–820.
Fanin R, Ruiz de Elvira MC, Sperotto A, Baccarani M, Goldstone A . Autologous stem cell transplantation for T and null cell CD30-positive anaplastic large cell lymphoma: analysis of 64 adult and paediatric cases reported to the European Group for Blood and Marrow Transplantation (EBMT). Bone Marrow Transplant 1999; 23: 437–442.
Pro B, Advani R, Brice P, Bartlett NL, Rosenblatt JD, Illidge T et al. Brentuximab vedotin (SGN-35) in patients with relapsed or refractory systemic anaplastic large-cell lymphoma: results of a phase II study. J Clin Oncol 2012; 30: 2190–2196.
Mossé YP, Lim MS, Voss SD, Wilner K, Ruffner K, Laliberte J et al. Safety and activity of crizotinib for paediatric patients with refractory solid tumours or anaplastic large-cell lymphoma: a Children’s Oncology Group phase 1 consortium study. Lancet Oncol 2013; 14: 472–480.
Lakshmaiah KC, Guruprasad B, Shah A, Kavitha S, Abraham LJ, Govindbabu K et al. Anaplastic large cell lymphoma: a single institution experience from India. J Cancer Res Ther 2013; 9: 649–652.
Stockklausner C, Behnisch W, Mechtersheimer G, Möller P, Kulozik AE . Long-term remission of children with relapsed and secondary anaplastic large cell non-Hodgkin lymphoma (ALCL) following treatment with pulsed dexamethasone and low dose etoposide. Pediatr Blood Cancer 2008; 50: 126–129.
Mikles B, Levine J, Gindin T, Bhagat G, Satwani P . Brentuximab vedotin (SGN-35) in a 3-year-old child with relapsed systemic anaplastic large cell lymphoma. J Pediatr Hematol Oncol 2014; 36: e85–e87.
Deng C, Pan B, O’Connor OA . Brentuximab vedotin. Clin Cancer Res 2013; 19: 22–27.
Foyil KV, Bartlett NL . Brentuximab vedotin and crizotinib in anaplastic large-cell lymphoma. Cancer J. 2012; 18: 450–456.
Gandhi L, Jänne PA . Crizotinib for ALK-rearranged non-small cell lung cancer: a new targeted therapy for a new target. Clin Cancer Res 2012; 18: 3737–3742.
Mussolin L, Damm-Welk C, Pillon M, Zimmermann M, Franceschetto G, Pulford K et al. Use of minimal disseminated disease and immunity to NPM-ALK antigen to stratify ALK-positive ALCL patients with different prognosis. Leukemia 2013; 27: 416–422.
Damm-Welk C, Mussolin L, Zimmermann M, Pillon M, Klapper W, Oschlies I et al. Early assessment of minimal residual disease identifies patients at very high relapse risk in NPM-ALK-positive anaplastic large-cell lymphoma. Blood 2014; 123: 334–337.
Acknowledgements
We thank the children and parents who participated in the study and Lorna Saint Ange for editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Bone Marrow Transplantation website
Supplementary information
Rights and permissions
About this article
Cite this article
Strullu, M., Thomas, C., Le Deley, MC. et al. Hematopoietic stem cell transplantation in relapsed ALK+ anaplastic large cell lymphoma in children and adolescents: a study on behalf of the SFCE and SFGM-TC. Bone Marrow Transplant 50, 795–801 (2015). https://doi.org/10.1038/bmt.2015.57
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2015.57
This article is cited by
-
Refractory Anaplastic Large Cell Lymphoma Rescued by the Combination of the Second-Generation ALK Inhibitor Brigatinib, High-dose Chemotherapy and Allogeneic Stem Cell Transplantation: A Case Report and Review of the Literature
Clinical Hematology International (2023)
-
Successful outcome with reduced-intensity condition regimen followed by allogeneic hematopoietic stem cell transplantation for relapsed or refractory anaplastic large-cell lymphoma
International Journal of Hematology (2019)