Abstract
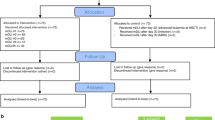
Peripheral T-cell lymphoma carries a poor prognosis. To document a possible graft-versus-lymphoma effect in this setting, we evaluated the impact of immunomodulation in 63 patients with peripheral T-cell lymphoma who relapsed after allogeneic transplant in 27 SFGM-TC centers. Relapse occurred after a median of 2.8 months. Patients were then treated with non-immunologic strategies (chemotherapy, radiotherapy) and/or immune modulation (donor lymphocyte infusions (DLI) and/or discontinuation of immunosuppressive therapy). Median overall survival (OS) after relapse was 6.1 months (DLI group: 23.6 months, non-DLI group: 3.6 months). Among the 14 patients who received DLI, 9 responded and 2 had stable disease. Among the remaining 49 patients, a complete response accompanied by extensive chronic GvHD was achieved in two patients after tapering of immunosuppressive drugs. Thirty patients received radio-chemotherapy, with an overall response rate of 50%. In multivariate analysis, chronic GvHD (odds ratio: 11.25 (2.68–48.21), P=0.0009) and skin relapse (odds ratio: 4.15 (1.04–16.50), P=0.043) were associated with a better response to treatment at relapse. In a time-dependent analysis, the only factor predictive of OS was the time from transplantation to relapse (hazards ratio: 0.33 (0.17–0.640), P=0.0009). This large series provides encouraging evidence of a true GvL effect in this disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Abouyabis AN, Shenoy PJ, Sinha R, Flowers CR, Lechowicz MJ . A systematic review and meta-analysis of front-line anthracycline-based chemotherapy regimens for peripheral T-cell lymphoma. ISRN Hematol 2011; 2011: 623924.
Reimer P, Rüdiger T, Geissinger E, Weissinger F, Nerl C, Schmitz N et al. Autologous stem-cell transplantation as first-line therapy in peripheral T-cell lymphomas: results of a prospective multicenter study. J Clin Oncol 2009; 27: 106–113.
Kyriakou C, Canals C, Finke J, Kobbe G, Harousseau J-L, Kolb H-J et al. Allogeneic stem cell transplantation is able to induce long-term remissions in angioimmunoblastic T-cell lymphoma: a retrospective study from the lymphoma working party of the European group for blood and marrow transplantation. J Clin Oncol 2009; 27: 3951–3958.
Gouill SL, Milpied N, Buzyn A, Latour RPD, Vernant J-P, Mohty M et al. Graft-versus-lymphoma effect for aggressive t-cell lymphomas in adults: a study by the Société Française de Greffe de Moëlle et de Thérapie Cellulaire. J Clin Oncol 2008; 26: 2264–2271.
Dodero A, Spina F, Narni F, Patriarca F, Cavattoni I, Benedetti F et al. Allogeneic transplantation following a reduced-intensity conditioning regimen in relapsed/refractory peripheral T-cell lymphomas: long-term remissions and response to donor lymphocyte infusions support the role of a graft-versus-lymphoma effect. Leukemia 2012; 26: 520–526.
Goldberg JD, Chou JF, Horwitz S, Teruya-Feldstein J, Barker JN, Boulad F et al. Long-term survival in patients with peripheral T-cell non-Hodgkin lymphomas after allogeneic hematopoietic stem cell transplant. Leuk Lymphoma 2012; 53: 1124–1129.
Duarte RF, Canals C, Onida F, Gabriel IH, Arranz R, Arcese W et al. Allogeneic hematopoietic cell transplantation for patients with mycosis fungoides and Sézary syndrome: a retrospective analysis of the Lymphoma Working Party of the European Group for Blood and Marrow Transplantation. J Clin Oncol 2010; 28: 4492–4499.
Jacobsen ED, Kim HT, Ho VT, Cutler CS, Koreth J, Fisher DC et al. A large single-center experience with allogeneic stem-cell transplantation for peripheral T-cell non-Hodgkin lymphoma and advanced mycosis fungoides/Sezary syndrome. Ann Oncol 2011; 22: 1608–1613.
Zain J, Palmer JM, Delioukina M, Thomas S, Tsai N-C, Nademanee A et al. Allogeneic hematopoietic cell transplant for peripheral T-cell non-Hodgkin lymphoma results in long-term disease control. Leuk Lymphoma 2011; 52: 1463–1473.
Loirat M, Chevallier P, Leux C, Moreau A, Bossard C, Guillaume T et al. Upfront allogeneic-stem cell transplantation for patients with non-localized untreated peripheral T-cell lymphoma: an intention-to-treat analysis from a single center. Ann Oncol 2014; 26: 386–392.
Robles M, Vigouroux S, Tabrizi R, Bouabdallah K, Dilhuydy M-S, Parrens M et al. Allogeneic SCT for patients with high-risk peripheral T-cell lymphoma in first response. Bone Marrow Transplant 2013; 48: 1484–1485.
Corradini P, Dodero A, Zallio F, Caracciolo D, Casini M, Bregni M et al. Graft-versus-lymphoma effect in relapsed peripheral T-cell non-Hodgkin’s lymphomas after reduced-intensity conditioning followed by allogeneic transplantation of hematopoietic cells. J Clin Oncol 2004; 22: 2172–2176.
Itonaga H, Tsushima H, Taguchi J, Fukushima T, Taniguchi H, Sato S et al. Treatment of relapsed adult T-cell leukemia/lymphoma after allogeneic hematopoietic stem cell transplantation: the Nagasaki Transplant Group experience. Blood 2013; 121: 219–225.
Kanakry JA, Kasamon YL, Gocke CD, Tsai H-L, Davis-Sproul J, Ghosh N et al. Outcomes of related donor HLA-identical or HLA-haploidentical allogeneic blood or marrow transplantation for peripheral T cell lymphoma. Biol Blood Marrow Transplant 2013; 19: 602–606.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. 1994 Consensus conference on acute GVHD grading. Bone Marrow Transplant 1995; 15: 825–828.
Mamez AC, Souchet L, Roos-Weil D, Uzunov M, Brun AL, Algrin C et al. Graft-versus-T-cell lymphoma effect: a sustained CR after tapering immunosuppressive drugs in a patient with angioimmunoblastic T-cell lymphoma in relapse after allogeneic transplantation. Bone Marrow Transplant 2014; 50: 304–306.
Molina A, Zain J, Arber DA, Angelopolou M, O’Donnell M, Murata-Collins J et al. Durable clinical, cytogenetic, and molecular remissions after allogeneic hematopoietic cell transplantation for refractory Sezary syndrome and mycosis fungoides. J Clin Oncol 2005; 23: 6163–6171.
Yuan L, Sun L, Bo J, Zhou Y, Li H, Yu L et al. Durable remission in a patient with refractory subcutaneous panniculitis-like T-cell lymphoma relapse after allogeneic hematopoietic stem cell transplantation through withdrawal of cyclosporine. Ann Transplant 2011; 16: 135–138.
Kako S, Izutsu K, Oshima K, Sato H, Kanda Y, Motokura T et al. Regression of the tumor after withdrawal of cyclosporine in relapsed extranodal natural killer/T cell lymphoma following allogeneic hematopoietic stem cell transplantation. Am J Hematol 2007; 82: 937–939.
Chakraverty R, Mackinnon S . Allogeneic transplantation for lymphoma. J Clin Oncol 2011; 29: 1855–1863.
Schetelig J, Thiede C, Bornhauser M, Schwerdtfeger R, Kiehl M, Beyer J et al. Evidence of a graft-versus-leukemia effect in chronic lymphocytic leukemia after reduced-intensity conditioning and allogeneic stem-cell transplantation: the Cooperative German Transplant Study Group. J Clin Oncol 2003; 21: 2747–2753.
Horwitz SM, Advani RH, Bartlett NL, Jacobsen ED, Sharman JP, O’Connor OA et al. Objective responses in relapsed T-cell lymphomas with single-agent brentuximab vedotin. Blood 2014; 123: 3095–3100.
Acknowledgements
ACM1 and SN20 designed the study and wrote the paper; VL2 and MB2 performed the statistical analysis, VL2 and MB2 contributed to the analysis of the results, PC, DB, SV, AX, NF, NC, YB, NI, CEB, FS, IYA, PT, ED, TL, JYC, AH, SM, KB, MO, JOB, GG, NM, MM, TD, JHB, FR, RO, CJ contributed to the data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Mamez, AC., Lévy, V., Chevallier, P. et al. Effect of immune modulation in relapsed peripheral T-cell lymphomas after post-allogeneic stem cell transplantation: a study by the Société Française de Greffe de Moelle et de Thérapie Cellulaire (SFGM-TC). Bone Marrow Transplant 51, 358–364 (2016). https://doi.org/10.1038/bmt.2015.280
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2015.280
This article is cited by
-
Romidepsin-induced durable remission for relapsed nodal peripheral T-cell lymphoma with T follicular helper phenotype after allogeneic hematopoietic cell transplantation
International Journal of Hematology (2023)
-
Allogeneic stem cell transplantation for peripheral T cell lymphomas: a retrospective study in 285 patients from the Société Francophone de Greffe de Moelle et de Thérapie Cellulaire (SFGM-TC)
Journal of Hematology & Oncology (2020)