Abstract
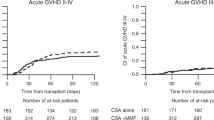
Previous studies have suggested that tacrolimus (TAC) is more potent than cyclosporine (CSA) for prophylaxis against acute GVHD after allogeneic hematopoietic stem cell transplantation (HSCT). However, the target blood concentrations of these drugs in these studies were not consistent with the current recommendations. Therefore, we performed a randomized controlled trial to compare CSA and TAC with target blood concentrations of 500 and 15 ng/ml, respectively, to prevent acute GVHD after unrelated HSCT. A total of 107 patients were randomized into a CSA group (n=53) or a TAC group (n=54). During the first 4 weeks after HSCT, more than 90% of the patients achieved a mean blood concentration of between 80 and 120% of the target concentration. The incidences of grade II–IV and grade III–IV acute GVHD were 39.6 and 7.5% for the CSA group and 33.3 and 9.4% for the TAC group, respectively (P=0.41 and P=0.76). Other clinical outcomes, including overall survival, disease-free survival and the incidences of relapse, non-relapse mortality, and organ toxicities, were also equivalent. We concluded that the combinations of CSA and TAC with strict dose adjustment showed similar efficacies and toxicities as prophylaxis against acute GVHD after unrelated HSCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ruutu T, Gratwohl A, de Witte T, Afanasyev B, Apperley J, Bacigalupo A et al. Prophylaxis and treatment of GVHD: EBMT-ELN working group recommendations for a standardized practice. Bone Marrow Transplant 2014; 49: 168–173.
Choi SW, Reddy P . Current and emerging strategies for the prevention of graft-versus-host disease. Nat Rev Clin Oncol 2014; 11: 536–547.
Hiraoka A, Ohashi Y, Okamoto S, Moriyama Y, Nagao T, Kodera Y et al. Phase III study comparing tacrolimus (FK506) with cyclosporine for graft-versus-host disease prophylaxis after allogeneic bone marrow transplantation. Bone Marrow Transplant 2001; 28: 181–185.
Nash RA, Antin JH, Karanes C, Fay JW, Avalos BR, Yeager AM et al. Phase 3 study comparing methotrexate and tacrolimus with methotrexate and cyclosporine for prophylaxis of acute graft-versus-host disease after marrow transplantation from unrelated donors. Blood 2000; 96: 2062–2068.
Ram R, Gafter-Gvili A, Yeshurun M, Paul M, Raanani P, Shpilberg O . Prophylaxis regimens for GVHD: systematic review and meta-analysis. Bone Marrow Transplant 2009; 43: 643–653.
Ratanatharathorn V, Nash RA, Przepiorka D, Devine SM, Klein JL, Weisdorf D et al. Phase III study comparing methotrexate and tacrolimus (prograf, FK506) with methotrexate and cyclosporine for graft-versus-host disease prophylaxis after HLA-identical sibling bone marrow transplantation. Blood 1998; 92: 2303–2314.
Yanada M, Emi N, Naoe T, Sakamaki H, Takahashi S, Hirabayashi N et al. Tacrolimus instead of cyclosporine used for prophylaxis against graft-versus-host disease improves outcome after hematopoietic stem cell transplantation from unrelated donors, but not from HLA-identical sibling donors: a nationwide survey conducted in Japan. Bone Marrow Transplant 2004; 34: 331–337.
Wingard JR, Nash RA, Przepiorka D, Klein JL, Weisdorf DJ, Fay JW et al. Relationship of tacrolimus (FK506) whole blood concentrations and efficacy and safety after HLA-identical sibling bone marrow transplantation. Biol Blood Marrow Transplant 1998; 4: 157–163.
Nakamura Y, Takeuchi H, Okuyama K, Akashi T, Jojima Y, Konno O et al. Evaluation of appropriate blood level in continuous intravenous infusion from trough concentrations after oral administration based on area under trough level in tacrolimus and cyclosporine therapy. Transplant Proc 2005; 37: 1725–1727.
Kimura S, Oshima K, Okuda S, Sato K, Sato M, Terasako K et al. Pharmacokinetics of CsA during the switch from continuous intravenous infusion to oral administration after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2010; 45: 1088–1094.
Ogawa N, Kanda Y, Matsubara M, Asano Y, Nakagawa M, Sakata-Yanagimoto M et al. Increased incidence of acute graft-versus-host disease with the continuous infusion of cyclosporine A compared to twice-daily infusion. Bone Marrow Transplant 2004; 33: 549–552.
Couriel DR, Thall P, Mickler K, De Lima M, Giralt S, Qazilbash MH et al. Phase II/III Randomized Study Comparing Two Different Tacrolimus Blood Levels for the Prevention of Graft-Versus-Host Disease (GVHD). Blood 2005; 106: 45a (abstract 142).
Mori T, Kato J, Shimizu T, Aisa Y, Nakazato T, Yamane A et al. Effect of early posttransplantation tacrolimus concentration on the development of acute graft-versus-host disease after allogeneic hematopoietic stem cell transplantation from unrelated donors. Biol Blood Marrow Transplant 2012; 18: 229–234.
Oshima K, Kanda Y, Nakasone H, Arai S, Nishimoto N, Sato H et al. Decreased incidence of acute graft-versus-host disease by continuous infusion of cyclosporine with a higher target blood level. Am J Hematol 2008; 83: 226–232.
Storb R, Deeg HJ, Whitehead J, Appelbaum F, Beatty P, Bensinger W et al. Methotrexate and cyclosporine compared with cyclosporine alone for prophylaxis of acute graft versus host disease after marrow transplantation for leukemia. N Engl J Med 1986; 314: 729–735.
Morishima Y, Morishita Y, Tanimoto M, Ohno R, Saito H, Horibe K et al. Low incidence of acute graft-versus-host disease by the administration of methotrexate and cyclosporine in Japanese leukemia patients after bone marrow transplantation from human leukocyte antigen compatible siblings; possible role of genetic homogeneity. The Nagoya Bone Marrow Transplantation Group. Blood 1989; 74: 2252–2256.
Kawamura K, Wada H, Yamasaki R, Ishihara Y, Sakamoto K, Ashizawa M et al. Prophylactic role of long-term ultra-low-dose acyclovir for varicella zoster virus disease after allogeneic hematopoietic stem cell transplantation. Int J Infect Dis 2014; 19: 26–32.
Kawamura K, Wada H, Yamasaki R, Ishihara Y, Sakamoto K, Ashizawa M et al. Low-dose acyclovir prophylaxis for the prevention of herpes simplex virus disease after allogeneic hematopoietic stem cell transplantation. Transpl Infect Dis 2013; 15: 457–465.
Kanda Y, Yamashita T, Mori T, Ito T, Tajika K, Mori S et al. A randomized controlled trial of plasma real-time PCR and antigenemia assay for monitoring CMV infection after unrelated BMT. Bone Marrow Transplant 2009; 45: 1325–1332.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. 1994 Consensus conference on acute GVHD grading. Bone Marrow Transplant 1995 15: 825–828.
Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol Blood Marrow Transplant 2005; 11: 945–956.
Nishida T, Murayama T, Hirai H, Okamoto S, Sao H, Hara M et al. Phase II study of tacrolimus and methotrexate for prophylaxis of acute graft-versus-host disease after HLA-A, B, and DRB1 genotypically mismatched unrelated bone marrow transplantation among Japanese patients. Int J Hematol 2009; 89: 98–105.
Gray RJ . A class of k-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat 1988; 16: 1141–1154.
Kanda Y . Investigation of the freely available easy-to-use software 'EZR' for medical statistics. Bone Marrow Transplant 2013; 48: 452–458.
A systematic collaborative overview of randomized trials comparing idarubicin with daunorubicin (or other anthracyclines) as induction therapy for acute myeloid leukaemia. AML Collaborative Group. Br J Haematol 1998; 103: 100–109.
Ohtake S, Miyawaki S, Fujita H, Kiyoi H, Shinagawa K, Usui N et al. Randomized study of induction therapy comparing standard-dose idarubicin with high-dose daunorubicin in adult patients with previously untreated acute myeloid leukemia: the JALSG AML201 Study. Blood 2011; 117: 2358–2365.
Ram R, Storer B, Mielcarek M, Sandmaier BM, Maloney DG, Martin PJ et al. Association between calcineurin inhibitor blood concentrations and outcomes after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 2012; 18: 414–422.
Kanda Y, Hyo R, Yamashita T, Fujimaki K, Oshima K, Onoda M et al. Effect of blood cyclosporine concentration on the outcome of hematopoietic stem cell transplantation from an HLA-matched sibling donor. Am J Hematol 2006; 81: 838–844.
Acknowledgements
We thank the staffs at the participating centers, including Tokyo Medical University Hospital, Yokohama City University Medical Center, Jikei University Hospital and Yokohama City University Hospital. We also thank the staffs at the Japan Clinical Research Support Unit and the data center of the Kanto Study Group for Cell Therapy.
Author contributions
YK designed the study and analyzed the data. TK, TM, MT, CN, AY, RW, SK, KK, JK, ND, MA, S-IK, MK, HK and SO participated in the study and gathered the data. AT contributed as a data manager. YK wrote the first draft of the paper and all other authors contributed to the final version.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
YK and CN received an honorarium and a research grant from Astellas Pharma Inc. and Novartis Pharma KK. SO received research a grant from Astellas Pharma Inc. Division of Hematology, Department of Medicine, Keio University School of Medicine had courses endowed by Novartis Pharmaceuticals.
Rights and permissions
About this article
Cite this article
Kanda, Y., Kobayashi, T., Mori, T. et al. A randomized controlled trial of cyclosporine and tacrolimus with strict control of blood concentrations after unrelated bone marrow transplantation. Bone Marrow Transplant 51, 103–109 (2016). https://doi.org/10.1038/bmt.2015.222
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2015.222
This article is cited by
-
Substitution of calcineurin inhibitors with corticosteroids after allogeneic hematopoietic cell transplantation
International Journal of Hematology (2023)
-
Clinical trials: design, endpoints and interpretation of outcomes
Bone Marrow Transplantation (2022)
-
Risk factors for adverse outcomes following haploidentical hematopoietic cell transplantation with posttransplant cyclophosphamide: a two-center analysis
Annals of Hematology (2022)
-
Negative impact of chronic graft-versus-host disease and glucocorticoid on the recovery of physical function after allogeneic hematopoietic stem cell transplantation
Bone Marrow Transplantation (2019)
-
Pharmacokinetic comparison of cyclosporin A and tacrolimus in graft-versus-host disease prophylaxis
Annals of Hematology (2017)