Abstract
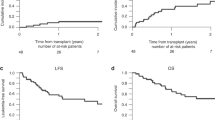
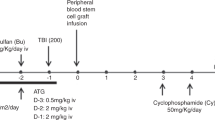
We report the results of a pilot study of a BU–fludarabine–alemtuzumab (BFA)-reduced toxicity conditioning (RTC) followed by allogeneic hematopoietic SCT (AlloHSCT) in 12 children and adolescents (<21 years) with malignant and non-malignant diseases. Stem cell sources were: two unrelated cord blood, one unrelated BM, two related and seven unrelated PBSC. Positive CD34 selection was performed in five unrelated PBSC grafts. RCT was carried out with BFA, and GVHD prophylaxis was FK506 and mycophenolate mofetil. The median time for neutrophil and platelet engraftment was 16 and 31 days, respectively. The P of developing ⩾grade II, ⩾grade III aGVHD and cGVHD was 41.6, 25 and 9%, respectively. Only 1 out of 12 developed ⩾grade III toxicity. There was one primary and no secondary graft failure. Mixed donor chimerism on day 100 and 1 year was median 99 and 96%, respectively; ⩾90% of recipients achieved ⩾80% donor chimerism. The 3-year overall survival (OS) in all patients was 91.7±8% (100% for malignant vs 80% for non-malignant diseases, ns). In all, 11 (91%) patients remain alive at median 2.8 (0.3–6.8) years. RTC followed by AlloHSCT, based on BFA conditioning, is feasible and tolerable in children and adolescents, and results in prompt achievement of durable mixed donor chimerism and excellent OS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ciurea SO, Andersson BS . Busulfan in hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2009; 15: 523–536.
Satwani P, Harrison L, Morris E, Del Toro G, Cairo MS . Reduced-intensity allogeneic stem cell transplantation in adults and children with malignant and nonmalignant diseases: end of the beginning and future challenges. Biol Blood Marrow Transplant 2005; 11: 403–422.
Slavin S, Nagler A, Naparstek E, Kapelushnik Y, Aker M, Cividalli G et al. Nonmyeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and nonmalignant hematologic diseases. Blood 1998; 91: 756–763.
Del Toro G, Satwani P, Harrison L, Cheung YK, Brigid Bradley M, George D et al. A pilot study of reduced intensity conditioning and allogeneic stem cell transplantation from unrelated cord blood and matched family donors in children and adolescent recipients. Bone Marrow Transplant 2004; 33: 613–622.
Jacobsohn DA, Duerst R, Tse W, Kletzel M . Reduced intensity haemopoietic stem-cell transplantation for treatment of non-malignant diseases in children. Lancet 2004; 364: 156–162.
Sandmaier BM, Mackinnon S, Childs RW . Reduced intensity conditioning for allogeneic hematopoeitic cell transplantation: current perspectives. Biol Blood Marrow Transplant 2009; 13 (suppl 1): 87–97.
Satwani P, Morris E, Bradley MB, Bhatia M, van de Ven C, Cairo MS . Reduced intensity and non-myeloablative allogeneic stem cell transplantation in children and adolescents with malignant and non-malignant diseases. Pediatr Blood Cancer 2008; 50: 1–8.
Lim ZY, Ingram W, Brand R, Akthari M, Milojkovic D, Ho AY et al. Clonal gammopathies following alemtuzumab-based reduced intensity conditioning haematopoietic stem cell transplantation: association with chronic graft-versus-host disease and improved overall survival. Bone Marrow Transplant 2007; 40: 747–752.
Horn B, Baxter-Lowe LA, Englert L, McMillan A, Quinn M, Desantes K et al. Reduced intensity conditioning using intravenous busulfan, fludarabine and rabbit ATG for children with nonmalignant disorders and CML. Bone Marrow Transplant 2006; 37: 263–269.
Ho AY, Pagliuca A, Kenyon M, Parker JE, Mijovic A, Devereux S et al. Reduced-intensity allogeneic hematopoietic stem cell transplantation for myelodysplastic syndrome and acute myeloid leukemia with multilineage dysplasia using fludarabine, busulphan, and alemtuzumab (FBC) conditioning. Blood 2004; 104: 1616–1623.
Bradley MB, Satwani P, Baldinger L, Morris E, van de Ven C, Del Toro G et al. Reduced intensity allogeneic umbilical cord blood transplantation in children and adolescent recipients with malignant and non-malignant diseases. Bone Marrow Transplant 2007; 40: 621–631.
Bullock JM, Smith PF, Booker BM, Loughner J, Capozzi D, McCarthy Jr PL et al. Development of a pharmacokinetic and Bayesian optimal sampling model for individualization of oral busulfan in hematopoietic stem cell transplantation. Ther Drug Monit 2006; 28: 62–66.
Fraser JK, Cairo MS, Wagner EL, McCurdy PR, Baxter-Lowe LA, Carter SL et al. Cord Blood Transplantation Study (COBLT): cord blood bank standard operating procedures. J Hematother 1998; 7: 521–561.
Kurtzberg J, Cairo MS, Fraser JK, Baxter-Lowe L, Cohen G, Carter SL et al. Results of the cord blood transplantation (COBLT) study unrelated donor banking program. Transfusion 2005; 45: 842–855.
Rubinstein P, Dobrila L, Rosenfield RE, Adamson JW, Migliaccio G, Migliaccio AR et al. Processing and cryopreservation of placental/umbilical cord blood for unrelated bone marrow reconstitution. Proc Natl Acad Sci USA 1995; 92: 10119–10122.
Kasow KA, Sims-Poston L, Eldridge P, Hale GA . CD34(+) hematopoietic progenitor cell selection of bone marrow grafts for autologous transplantation in pediatric patients. Biol Blood Marrow Transplant 2007; 13: 608–614.
Osunkwo I, Bessmertny O, Harrison L, Cheung YK, Van de Ven C, del Toro G et al. A pilot study of tacrolimus and mycophenolate mofetil graft-versus-host disease prophylaxis in childhood and adolescent allogeneic stem cell transplant recipients. Biol Blood Marrow Transplant 2004; 10: 246–258.
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA et al. Clinical manifestations of graft-versus-host disease in human recipients of marrow from HL-A-matched sibling donors. Transplantation 1974; 18: 295–304.
Waxman IM, Militano O, Baldinger L, Roman E, Qualter E, Morris E et al. Sequential administration of sargramostim and filgrastim in pediatric allogeneic stem cell transplantation recipients undergoing myeloablative conditioning. Pediatr Transplant 2009; 13: 464–474.
Roman E, Osunkwo I, Militano O, Cooney E, van de Ven C, Cairo MS . Liposomal amphotericin B prophylaxis of invasive mold infections in children post allogeneic stem cell transplantation. Pediatr Blood Cancer 2008; 50: 325–330.
Shereck EB, Cooney E, van de Ven C, Della-Lotta P, Cairo MS . A pilot phase II study of alternate day ganciclovir and foscarnet in preventing cytomegalovirus (CMV) infections in at-risk pediatric and adolescent allogeneic stem cell transplant recipients. Pediatr Blood Cancer 2007; 49: 306–312.
Horn B, Soni S, Khan S, Petrovic A, Breslin N, Cowan M et al. Feasibility study of preemptive withdrawal of immunosuppression based on chimerism testing in children undergoing myeloablative allogeneic transplantation for hematologic malignancies. Bone Marrow Transplant 2009; 43: 469–476.
Hardy NM, Hakim F, Steinberg SM, Krumlauf M, Cvitkovic R, Babb R et al. Host T cells affect donor T cell engraftment and graft-versus-host disease after reduced-intensity hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2007; 13: 1022–1030.
Kaplan EL, Meier P . Non parametric estimation for incomplete observations. J Am Stat Assoc 1958; 53: 457–481.
Bhatia M, Militano O, Jin Z, Figurski M, Shaw L, Moore V et al. An age-dependent pharmacokinetic study of intravenous and oral mycophenolate mofetil in combination with tacrolimus for GVHD prophylaxis in pediatric allogeneic stem cell transplantation recipients. Biol Blood Marrow Transplant; 2010; 16: 333–343.
Landgren O, Gilbert ES, Rizzo JD, Socie G, Banks PM, Sobocinski KA et al. Risk factors for lymphoproliferative disorders after allogeneic hematopoietic cell transplantation. Blood 2009; 113: 4992–5001.
Styczynski J, Einsele H, Gil L, Ljungman P . Outcome of treatment of Epstein-Barr virus-related post-transplant lymphoproliferative disorder in hematopoietic stem cell recipients: a comprehensive review of reported cases. Transpl Infect Dis 2009; 11: 383–392.
Styczynski J, Reusser P, Einsele H, de la Camara R, Cordonnier C, Ward KN et al. Management of HSV, VZV and EBV infections in patients with hematological malignancies and after SCT: guidelines from the Second European Conference on Infections in Leukemia. Bone Marrow Transplant 2009; 43: 757–770.
Kalwak K, Gorczynska E, Toporski J, Turkiewicz D, Slociak M, Ussowicz M et al. Immune reconstitution after haematopoietic cell transplantation in children: immunophenotype analysis with regard to factors affecting the speed of recovery. Br J Haematol 2002; 118: 74–89.
Small TN . Immunologic reconstitution following stem cell transplantation. Curr Opin Hematol 1996; 3: 461–465.
Shah AJ, Kapoor N, Crooks GM, Weinberg KI, Azim HA, Killen R et al. The effects of Campath 1H upon graft-versus-host disease, infection, relapse, and immune reconstitution in recipients of pediatric unrelated transplants. Biol Blood Marrow Transplant 2007; 13: 584–593.
Koehl U, Bochennek K, Zimmermann SY, Lehrnbecher T, Sorensen J, Esser R et al. Immune recovery in children undergoing allogeneic stem cell transplantation: absolute CD8+ CD3+ count reconstitution is associated with survival. Bone Marrow Transplant 2007; 39: 269–278.
Schwinger W, Weber-Mzell D, Zois B, Rojacher T, Benesch M, Lackner H et al. Immune reconstitution after purified autologous and allogeneic blood stem cell transplantation compared with unmanipulated bone marrow transplantation in children. Br J Haematol 2006; 135: 76–84.
Acknowledgements
This work was supported in part by the Pediatric Cancer Research Foundation, Marisa Fund, Sonia Scaramella Fund, Paul Luisi Foundation, Dream for Discovery and Cure Fund, Brittany Barron Fund (MSC), and by a grant from Children's Medical Care Foundation (JSt).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
LMS is a member of the scientific advisory board of Saladax Biomedical Inc. MBB is Associate Director of Global Clinical Research in Oncology at Bristol-Myers Squibb. Rest of the authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Styczynski, J., Tallamy, B., Waxman, I. et al. A pilot study of reduced toxicity conditioning with BU, fludarabine and alemtuzumab before the allogeneic hematopoietic SCT in children and adolescents. Bone Marrow Transplant 46, 790–799 (2011). https://doi.org/10.1038/bmt.2010.209
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2010.209
Keywords
This article is cited by
-
Worked to the bone: antibody-based conditioning as the future of transplant biology
Journal of Hematology & Oncology (2022)
-
Is biological therapy in systemic sclerosis the answer?
Rheumatology International (2020)
-
Prophylaxis and treatment with mycophenolate mofetil in children with graft-versus-host disease undergoing allogeneic hematopoietic stem cell transplantation: a nationwide survey in Japan
International Journal of Hematology (2019)
-
Single centre results of targeted busulphan, fludarabine and serotherapy conditioning in haematopoietic stem cell transplantation for haemophagocytic lymphohistiocytosis
Bone Marrow Transplantation (2018)
-
Safety of liposomal cytarabine CNS prophylaxis in children, adolescent and young adult hematopoietic stem cell transplant recipients with acute leukemia and non-Hodgkin lymphoma
Bone Marrow Transplantation (2016)