Abstract
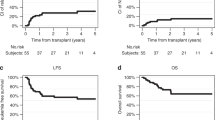
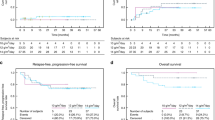
We report the toxicity of high-dose chemotherapy (HDC) based on etoposide, thiotepa and CY (ETC) in children with poor-prognosis Ewing's sarcoma family tumors (ESFTs). A total of 26 patients with high-risk ESFT (metastasis or axis localization or tumor volume >200 ml or necrosis <95%) were reviewed. The conditioning was based on etoposide (600 mg/m2), thiotepa (750 mg/m2) and CY (120 mg/kg) followed by autologous BM or PBSC rescue. The conditioning regimen was well tolerated, without any toxic deaths. The median time from transplant to a neutrophil count of >0.5 × 109/l was 10 days (range 6–27) and 22.5 days (range 9–114) for a plt count of >50 × 109/l. Oral mucositis was recorded in 20 patients, grade 1/2 in 19 and grade 3 in the last patient. Diarrhea grade 1/2 was recorded in four patients and grade 1/2 liver toxicity in four patients. Sepsis was documented in four cases and skin toxicity in three. Lung and tubular toxicity, respectively, were reported in one patient each. We conclude that the ETC regimen presented a limited and manageable toxicity. Further studies would confirm the role of ETC in high-risk ESFT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bernstein M, Heinrich K, Paulussen M, Randall RL, Schuck A, Teot LA et al. Ewing's sarcoma family of tumors: current management. Oncologist 2006; 11: 503–519.
Delattre O, Zucman J, Melot T, Garau XS, Zucker JM, Lenoir GM et al. The Ewing family of tumors: a subgroup of small-round-cell tumors defined by specific chimeric transcripts. N Engl J Med 1994; 331: 294–299.
Gurney JG, Swensen AR, Bulterys M . Malignant bone tumors. In: Ries LAG, Smith MA, Gumey JG, et al., (eds). Cancer Incidence and Survival Among Children and Adolescents: United States SEER Program 1975–1995. NIH Pub. No. 99–4649 National Cancer Institute, SEER Program: Bethesda, MD, 1999.
Paulussen M, Ahrens S, Burdach S, Craft A, Dockhorn-Dworniczak B, Dunst J et al. Primary metastatic (stage IV) Ewing tumor: survival analysis of 171 patients from the EICESS studies. European Intergroup Cooperative Ewing sarcoma studies. Ann Oncol 1998; 9: 275–281.
Craft AW, Cotterill SJ, Bullimore JA, Pearson D . Long-term results from the first UKCCSG Ewing's tumour study (ET-1). Eur J Cancer 1997; 33: 1061–1069.
Raney RB, Asmar L, Newton Jr WA, Bagwell C, Breneman JC, Crist W et al. Ewing's sarcoma of soft tissues in childhood: a report from the Intergroup Rhabdomyosarcoma Study, 1972–1991. J Clin Oncol 1997; 15: 574–582.
Göbel V, Jürgens H, Etspüler G, Kemperdick H, Jungblut RM, Stienen U et al. Prognostic significance of tumor volume in localized Ewing's sarcoma of bone in children and adolescents. J Cancer Res Clin Oncol 1987; 113: 187–191.
Picci P, Böhling T, Bacci G, Ferrari S, Sangiorgi L, Mercuri M et al. Chemotherapy-induced tumor necrosis as a prognostic factor in localized Ewing's sarcoma of the extremities. J Clin Oncol 1997; 15: 1553–1559.
Burdach S, Jürgens H, Peters C, Nürnberger W, Mauz-Körholz C, Körholz D et al. Myeloablative radiochemotherapy and hematopoietic stem-cell rescue in poor-prognosis Ewing's sarcoma. J Clin Oncol 1993; 11: 1482–1488.
Atra A, Whelan JS, Calvagna V, Shankar AG, Ashley S, Shepherd V et al. High-dose busulphan/melphalan with autologous stem cell rescue in Ewing's sarcoma. Bone Marrow Transplant 1997; 20: 843–846.
Ladenstein R, Lasset C, Pinkerton R, Zucker JM, Peters C, Burdach S et al. Impact of megatherapy in children with high-risk Ewing's tumours in complete remission: a report from the EBMT solid tumour registry. Bone Marrow Transplant 1995; 15: 697–705.
Rosen G, Caparros B, Huvos AG, Kosloff C, Nirenberg A, Cacavio A et al. Preoperative chemotherapy for osteogenic sarcoma: selection of postoperative adjuvant chemotherapy based on the response of the primary tumor to preoperative chemotherapy. Cancer 1982; 49: 1221–1230.
Milano GM, Cozza R, Ilari I, De Sio L, Boldrini R, Jenkner A et al. High istologic, overall response to dose intensification of ifosfamide, carboplatin, etoposide with cyclophophamide, doxorubicin, vincristine in patients with high-risk Ewing sarcoma family tumors. Cancer 2006; 106: 1838–1845.
International Common Toxicity Criteria Version 3 (CTC.3). Available from URL http://www.fda.gov/cder/cancer/toxicityframe.htmCTC.
Bearman SI, Appelbaum FR, Back A, Petersen FB, Buckner CD, Sullivan KM et al. Regimen related toxicity in patients undergoing bone marrow transplantation. J Clin Oncol 1988; 6: 1562–1568.
Kaplan EL, Meier P . Nonparametric estimation from incomplete observation. J Am Stat Assoc 1959; 53: 457–481.
Rodriguez-Galindo C, Spunt SL, Pappo AS . Treatment of Ewing sarcoma family of tumors: current status and outlook for the future. Med Pediatr Oncol 2003; 40: 276–287.
Ahrens S, Hoffman C, Jabar S, Braun-Munzinger G, Paulussen M, Dunst J et al. Evaluation of prognostic factors in a tumor volume-adapted treatment strategy for localized Ewing sarcoma of the bone: the CESS 86 experience. Med Ped Oncol 1999; 32: 186–195.
Rosito P, Mancini AF, Rondelli R, Abate ME, Pession A, Bedei L et al. Italian Cooperative Study for the treatment of children and young adults with localized Ewing sarcoma of bone: a preliminary report of 6 years of experience. Cancer 1999; 86: 421–428.
Cotterill SJ, Ahrens S, Paulussen M, Jürgens HF, Voûte PA, Gadner H et al. Prognostic factors in Ewing's tumor of bone: analysis of 975 patients from the European Intergroup Cooperative Ewing's Sarcoma Study Group. J Clin Oncol 2000; 18: 3108–3114.
Paulussen M, Ahrens S, Dunst J, Winkelmann W, Exner GU, Kotz R et al. Localized Ewing tumor of bone: final results of the Cooperative Ewing's Sarcoma Study CESS 86. J Clin Oncol 2001; 19: 1818–1829.
Paulussen M, Craft AW, Lewis I, Hackshaw A, Douglas C, Dunst J et al. Results of the EICESS-92 study: two randomized trials of Ewing's sarcoma treatment—cyclophosphamide compared with ifosfamide in standard-risk patients and assessment of benefit of etoposide added to standard treatment in high-risk patients. J Clin Oncol 2008; 26: 4385–4393.
Rodriguez-Galindo C, Liu T, Krasin MJ, Wu J, Billups CA, Daw NC et al. Analysis of prognostic factors in Ewing sarcoma family tumors: review of St. Jude Children's Research Hospital studies. Cancer 2007; 110: 375–384.
Burdach S, Jurgens H, Peters C, Nürnberger W, Mauz-Körholz C, Körholz D et al. Myeloablative radiochemotherapy and hematopoietic stem-cell rescue in poor prognosis Ewing's sarcoma. J Clin Oncol 1993; 11: 1482–1488.
Ladenstein R, Lasset C, Pinkerton R, Zucker JM, Peters C, Burdach S et al. Impact of megatherapy in children with high-risk Ewing's tumours in complete remission: a report from the EBMT solid tumour registry. Bone Marrow Transplant 1995; 15: 697–705.
Atra A, Whelan JS, Calvagna V, Shankar AG, Ashley S, Shepherd V et al. High-dose busulphan/melphalan with autologous stem cell rescue in Ewing's sarcoma. Bone Marrow Transplant 1997; 20: 843–846.
Madero L, Munoz A, Sanchez de Toledo J, Díaz MA, Maldonado MS, Ortega JJ et al. Megatherapy in children with high-risk Ewing's sarcoma in first complete remission. Bone Marrow Transplant 1998; 21: 795–799.
Hawkins D, Barnett T, Bensinger W, Gooley T, Sanders J . Busulfan, melphalan, and thiotepa with or without total marrow irradiation with hematopoietic stem cell rescue for poor risk Ewing-sarcoma-family tumors. Med Pediatr Oncol 2000; 34: 328–337.
Oberlin O, Rey A, Desfachelles AS, Philip T, Plantaz D, Schmitt C et al. Impact of high dose busulfan plus melphalan as consolidation in metastatic Ewing tumors: a study by the Societè Francaise des cancers de l’enfant. J Clin Oncol 2006; 24: 3997–4002.
Al-Faris N, Al Harbi T, Goia C, Pappo A, Doyle J, Gassas A . Does consolidation with autologous stem cell transplantation improve the outcome of children with metastatic or relapsed Ewing sarcoma? Pediatr Blood Cancer 2007; 49: 190–195.
Marcus RB, Graham-Pole JR, Springfield DS, Fort JA, Gross S, Mendenhall NP et al. High risk Ewing's sarcoma: end-intensification using autologous bone marrow transplantation. Int J Radiat Oncol Biol Phys 1988; 15: 53–59.
Horowitz ME, Kinsella TJ, Wexler LH, Belasco J, Triche T, Tsokos M et al. Total-body irradiation and autologous bone marrow transplant in the treatment of high-risk Ewing's sarcoma and rhabdomyosarcoma. J Clin Oncol 1993; 11: 1911–1918.
Stewart DA, Gyonyor E, Paterson AHG, Paterson AH, Arthur K, Temple W et al. High-dose melphalan±total body irradiation and autologous hematopoietic stem cell rescue for adult patients with Ewing's sarcoma or peripheral neuroectodermal tumor. Bone Marrow Transplant 1996; 18: 315–318.
Meyers PA, Krailo MD, Ladanyi M, Chan KW, Sailer SL, Dickman PS et al. High-dose melphalan, etoposide, total-body irradiation, and autologous stem-cell reconstitution as consolidation therapy for high-risk Ewing's sarcoma does not improve prognosis. J Clin Oncol 2001; 19: 2812–2820.
Kushner BH, Meyers PA . How effective is dose-intensive/myeloablative therapy against Ewing's sarcoma/primitive neuroectodermal tumor metastatic to bone or bone marrow? The Memorial Sloan-Kettering experience and a literature review. J Clin Oncol 2001; 19: 870–880.
Bacci G, Ferrari S, Bertoni F, Rimondini S, Longhi A, Bacchini P et al. Prognostic factors in nonmetastatic Ewing's sarcoma of bone treated with adjuvant chemotherapy: analysis of 359 patients at the Istituto Ortopedico Rizzoli. J Clin Oncol 2000; 18: 4–11.
Seddon BM, Cassoni AM, Galloway MJ, Rees JH, Whelan JS . Fatal radiation myelopathy after high-dose busulfan and melphalan chemotherapy and radiotherapy for Ewing's sarcoma: a review of the literature and implications for practice. Clin Oncol (R Coll Radiol) 2005; 17: 385–390.
Lucidarme N, Valteau-Couanet D, Oberlin O, Couanet D, Kalifa C, Beaujean F et al. Phase II study of high-dose thiotepa and hematopoietic stem cell transplantation in children with solid tumors. Bone Marrow Transplant 1998; 22: 535–540.
Saarinen UM, Hovi L, Mäkipernaa A, Riikonen P . High-dose thiotepa with autologous bone marrow rescue in pediatric solid tumors. Bone Marrow Transplant 1991; 8: 369–376.
Weaver CH, Bensinger WI, Applelbaum FR, Lilleby K, Sandmaier B, Brunvand M et al. Phase I study of high dose busulfan, melphalan and thiotepa with autologous stem cell support in patients with refractory malignancies. Bone marrow Transplant 1994; 14: 813–819.
Schiffman KS, Bensinger WI, Applelbaum FR, Rowley S, Lilleby K, Clift RA et al. Phase II study of high dose busulfan, melphalan and thiotepa with autologous peripheral blood stem cell support in patients with malignant disease. Bone Marrow Transplant 1996; 17: 943–950.
Heideman RL, Cole DE, Balis F, Sato J, Reaman GH, Packer RJ et al. Phase I and pharmacokinetic evaluation of thiotepa in the cerebrospinal fluid and plasma of pediatric patients: evidence for dose-dependent plasma clearance of thiotepa. Cancer Res 1989; 49: 736–741.
Burdach S, Meyer-Bahlburg A, Laws HJ, Haase R, van Kaik B, Metzner B et al. High-dose therapy for patients with primary multifocal and early relapsed Ewing's tumors: results of two consecutive regimens assessing the role of total-body irradiation. J Clin Oncol 2003; 21: 3072–3078.
Burke MJ, Walterhouse DO, Jacobsohn DA, Duerst RE, Kletzel M . Tandem high-dose chemotherapy with autologous peripheral hematopoietic progenitor cell rescue as consolidation therapy for patients with high-risk Ewing family tumors. Pediatr Blood Cancer 2007; 49: 196–198.
Chan KW, Petropoulos M, Choroszy M, Herzog C, Jaffe N, Ater J et al. High-dose sequential chemotherapy and autologous stem cell reinfusion in advanced pediatric solid tumors. Bone Marrow Transplant 1997; 20: 1039–1043.
Ozkaynak MF, Sandoval C, Levendoglu-Tugal O, Jayabose S . A pilot trial of tandem autologous peripheral blood progenitor cell transplantation following high-dose thiotepa and carboplatin in children with poor-risk central nervous system tumors. Pediatr Hematol Oncol 2004; 21: 635–645.
Acknowledgements
We thank L. Ravà, for statistical support and ‘Comitato Girasole Onlus per l’Oncologia del Bambin Gesù’ for grant support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Ilari, I., De Ioris, M., Milano, G. et al. Toxicity of high-dose chemotherapy with etoposide, thiotepa and CY in treating poor-prognosis Ewing's sarcoma family tumors: the experience of the Bambino Gesù Children's Hospital. Bone Marrow Transplant 45, 1274–1280 (2010). https://doi.org/10.1038/bmt.2009.353
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2009.353