Abstract
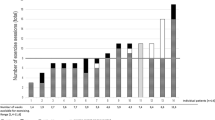
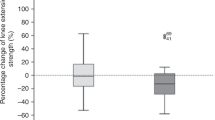
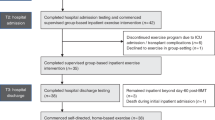
The aim of this randomized controlled trial was to investigate the effect of a 4- to 6-week multimodal program of exercise, relaxation and psychoeducation on physical capacity, functional performance and quality of life (QOL) in allogeneic hematopoietic cell transplantation (allo-HSCT) adult recipients. In all, 42 patients were randomized to a supervised multimodal intervention or to a control group receiving usual care. The primary end point was on aerobic capacity measured in VO2 max. Secondary end points were muscle strength, functional performance, physical activity level, QOL, fatigue, psychological well-being and clinical outcomes. The multimodal intervention had a significant effect on physical capacity: VO2 max (P<0.0001) and muscle strength: chest press (P<0.0001), leg extension (P=0.0003), right elbow flexor (P=0.0009), right knee extensor (P<0.0001) and functional performance (stair test) (0.0008). Moreover, the intervention group showed significantly better results for the severity of diarrhea (P=0.014) and fewer days of total parenteral nutrition (P=0.019). Longitudinal changes in QOL, fatigue and psychological well-being favored the intervention group, but did not reach statistical significance. Assignment of a multimodal intervention during allo-HSCT did not cause untoward events, sustained aerobic capacity and muscle strength and reduced loss of functional performance during hospitalization.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Gratwohl A, Baldomero H, Horisberger B, Schmid C, Passweg J, Urbano-Ispizua A . Current trends in hematopoietic stem cell transplantation in Europe. Blood 2002; 100: 2374–2386.
Devergie A . Graft vs host disease. In: Apperley J, Carrerras E, Gluckman E, Gratwohl A, Masszi T (eds). The EBMT Handbook 2004 revised edition: haemopoietic stem cell transplantation. European Group for Blood and Marrow Transplantation: Maastricht, 2004, pp 163–168.
Pallera AM, Schwartzberg LS . Managing the toxicity of hematopoietic stem cell transplant. J Support Oncol 2004; 2: 223.
Andrykowski MA, Brady MJ, Greiner CB, Altmaier EM, Burish TG, Antin JH et al. ‘Returning to normal’ following bone marrow transplantation: outcomes, expectations and informed consent. Bone Marrow Transplant 1995; 15: 581.
Courneya KS, Keats MR, Turner AR . Physical exercise and quality of life in cancer patients following high dose chemotherapy and autologous bone marrow transplantation. Psychooncology 2000; 9: 127–136.
Sherman RS, Cooke E, Grant M . Dialogue among survivors of hematopoietic cell transplantation support-group themes. J Psychosoc Oncol 2005; 23: 1–24.
Wiskemann J, Huber G . Physical exercise as adjuvant therapy for patients undergoing hematopoietic stem cell transplantation. Bone Marrow Transplant 2008; 41: 321–329.
Courneya KS, Friedenreich CM . Relationship between exercise pattern across the cancer experience and current quality of life in colorectal cancer survivors. J Altern Complement Med 1997; 3: 215–226.
Courneya KS, Friedenreich CM . Relationship between exercise during cancer treatment and current quality of life in survivors of breast cancer. J Psychosoc Oncol 1997; 15: 35–57.
Courneya KS, Keats MR, Turner AR . Physical exercise and quality of life in cancer patients following high dose chemotherapy and autologous bone marrow transplantation. Psychooncology 2000; 9: 127–136.
Hayes SC, Davies PS, Parker TW, Bashford J, Green A . Role of a mixed-type, moderate intensity exercise programme after peripheral blood stem cell transplantation. Br J Sports Med 2004; 38: 304–309.
Dimeo F, Schwartz S, Fietz T, Wanjura T, Boning D, Thiel E . Effects of endurance training on the physical performance of patients with hematological malignancies during chemotherapy. Support Care Cancer 2003; 11: 623–628.
Dimeo F, Bertz H, Finke J, Fetscher S, Mertelsmann R, Keul J . An aerobic exercise program for patients with haematological malignancies after bone marrow transplantation. Bone Marrow Transplant 1996; 18: 1157–1160.
Carlson LE, Smith D, Russell J, Fibich C, Whittaker T . Individualized exercise program for the treatment of severe fatigue in patients after allogeneic hematopoietic stem-cell transplant: a pilot study. Bone Marrow Transplant 2006; 37: 945–954.
Mello M, Tanaka C, Dulley FL . Effects of an exercise program on muscle performance in patients undergoing allogeneic bone marrow transplantation. Bone Marrow Transplant 2003; 32: 723–728.
Coleman EA, Coon S, Hall-Barrow J, Richards K, Gaylor D, Sterwart B . Feasibility of exercise during treatment for multiple myeloma. Cancer Nurs 2003; 26: 410–419.
Hayes SC, Davies PS, Parker TW, Bashford J . Total energy expenditure and body composition changes following peripheral blood stem cell transplantation and participation in an exercise programme. Bone Marrow Transplant 2003; 31: 331–338.
DeFor TE, Burns LJ, Gold EM, Weisdorf DJ . A randomized trial of the effect of a walking regimen on the functional status of 100 adult allogeneic donor hematopoietic cell transplant patients. Biol Blood Marrow Transplant 2007; 13: 948–955.
Kim SD, Kim HS . A series of bed exercises to improve lymphocyte count in allogeneic bone marrow transplantation patients. Eur J Cancer Care (Engl) 2006; 15: 453–457.
Dimeo F, Fetscher S, Lange W, Mertelsmann R, Keul J . Effects of aerobic exercise on the physical performance and incidence of treatment related complications after high dose chemotherapy. Blood 1997; 90: 3390–3394.
Hayes SC, Rowbottom D, Davies PS, Parker TW, Bashford J . Immunological changes after cancer treatment and participation in an exercise program. Med Sci Sports Exerc 2003; 35: 2–9.
Dimeo FC, Stieglitz RD, Novelli-Fischer U, Fetscher S, Keul J . Effects of physical activity on the fatigue and psychologic status of cancer patients during chemotherapy. Cancer 1999; 85: 2273–2277.
Wilson RW, Jacobsen PB, Fields KK . Pilot study of a home-based aerobic exercise program for sedentary cancer survivors treated with hematopoietic stem cell transplantation. Bone Marrow Transplant 2005; 35: 721–727.
Hayes S, Davies PS, Parker T, Bashford J, Newman B . Quality of life changes following peripheral blood stem cell transplantation and participation in a mixed-type, moderate-intensity, exercise program. Bone Marrow Transplant 2004; 33: 553–558.
Cunningham BA, Morris G, Cheney CL, Buergel N, Aker SN, Lenssen P . Effects of resistive exercise on skeletal muscle in marrow transplant recipients receiving total parenteral nutrition. J Parenter Enteral Nutr 1986; 10: 558–563.
Wilson RW, Taliaferro LA, Jacobsen PB . Pilot study of a self-administered stress management and exercise intervention during chemotherapy for cancer. Support Care Cancer 2006; 14: 928–935.
Adamsen L, Quist M, Midtgaard J, Andersen C, Møller, Knutsen L et al. The effect of a multidimensional exercise intervention on physical capacity, well-being and quality of life in cancer patients undergoing chemotherapy. Support Care Cancer 2006; 14: 116–1271.
Mock V, Burke MB, Sheehan PK, Creaton EM, Winningham ML, McKenney-Tedder S et al. A nursing rehabilitation program for women with breast cancer receiving adjuvant chemotherapy. Oncol Nurs Forum 1994; 21: 899–907.
Kim SD, Kim HS . Effects of a relaxation breathing exercise on fatigue in haemopoietic stem cell transplantation patients. J Clin Nurs 2005; 14: 51–55.
Carlson CR, Hoyle RH . Efficacy of abbreviated progressive muscle relaxation training: a quantitative review of behavioural medicine research. J Consult Clin Psychol 1993; 61: 1059–1067.
Fried R . Breathing as a clinical tool. In: Mostofsky DI, Barlow DH (eds). The Management of Stress and Anxiety in Medical Disorders. Allyn & Bacon: Boston, MA, 2000, pp 100–118.
Boesen E, Ross L, Frederiksen K, Thomsen BL, Dahlstrøm K, Schmidt G et al. Psychoeducational intervention for patients with cutaneous malignant melanoma: a replication study. J Clin Oncol 2005; 23: 1270–1277.
Molassiotis A, Yung HP, Yam BM, Chan FY, Mok TS . The effectiveness of progressive muscle relaxation in managing chemotherapy, induced nausea and vomiting in Chinese breast cancer patients: a randomized controlled trial. Support Care Cancer 2002; 10: 237–246.
Jarden M, Hovgaard D, Boesen E, Adamsen L . Pilot study of a multimodal intervention: mixed-type exercise and psychoeducation in patients undergoing allogeneic stem cell transplantation. Bone Marrow Transplant 2007; 40: 793–800.
Thorhauge C . Registertilsynet åbner for følsomme data på Internet (Danish data protection agency open up for sensitive data on the Internet). 1999. Available from: http://www.computerworld.dk/art/4111?a=search&i=0 Last accessed July 4, 2008.
Nieman DC, Courneya KS . Immunological conditions. In: American College of Sports Medicine ACSM's Resource Manual for Guidelines for Exercise Testing and Prescription. Lippincott Williams & Wilkins: Philadelphia, 2006, pp 528–542.
Jacobsen E . Progressive Relaxation: A Physiological And Clinical Investigation of Muscular States and Their Significance in Psychology and Medical Practice. University of Chicago Press: Chicago, 1938.
Moorey S, Greer S . Cognitive Behaviour Therapy for People With Cancer. 2nd edn. Oxford University Press: Oxford, 2003.
Ainsworth BE, Haskell WL, Whitt MC, Irwin ML, Swartz AM, Strath SJ et al. Compendium of Physical Activities: an update of activity codes and MET intensities. Med Sci Sports Exerc 2000; 32: S498–S516.
Karvonen MJ, Kentala E, Mustala O . The effects of training heart rate: a longitudinal study. Ann Med Exp Biol Fenn 1957; 35: 307–315.
Borg G . Borg's Perceived Exertion and Pain scales. Human Kinetics: Champaign, IL, 1998.
Karnofsky DA, Burchenal JH . The clinical evaluation of chemotherapeutic agents in cancer. In: MacLeod CM (ed). Evaluation of Chemotherapeutic Agents. Columbia University Press: New York, 1949, pp 191–205.
Saltin B, Grimby G . Physiological analysis of middle-aged and old former athletes: comparison with still active athletes of the same ages. Circulation 1968; 38: 1104–1115.
Aastrand PO, Rodahl K, Dahl HA, Strømme SB . Evaluation of physical performance on the basis of tests. In: Textbook of Work Physiology: Physiological Bases of Exercise. 4th edn. Human Kinetics: Champaign, 2003, pp 273–297; 333–364.
Humphries B, Dugan EL, Doyle LA . Muscular fitness. In: American College of Sports Medicine ACSM's Resource Manual for Guidelines for Exercise Testing and Prescription. Lippincott Williams & Wilkins: Philadelphia, 2006, pp 206–224.
Harding VR, Williams AC, Richardson PH, Nicholas MK, Jackson JL, Richardson IH et al. The development of a battery of measures for assessing physical functioning of chronic pain patients. Pain 1994; 58: 367–375.
Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ et al. The European Organization for Research and Treatment of Cancer QLQ-C30: quality-og-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst 1993; 85: 365–376.
Grønvold M, Klee M, Sprangers MAG, Aaronson NK . Validation of the EORTC QLQ-C30 quality of life questionnaire through combined qualitative and quantitative assessment of patient–observer agreement. J Clin Epidemiol 1997; 50: 441–450.
Yellen SB, Cella DF, Webster K, Blendowski C, Kaplan E . Measuring fatigue and other anemia-related symptoms with the Functional assessment of Cancer Therapy (FACT) measurement system. J Pain Symptom Manage 1997; 13: 63–74.
Carroll BT, Kathol RG, Noyes R, Wald TG, Clamon GH . Screening for depression and anxiety in cancer patients using the Hospital Anxiety and Depression Scale. Gen Hosp Psychiatry 1993; 15: 69–74.
Johnston M, Pollard B, Hennessey P . Construct validation of the hospital anxiety and depression scale with clinical populations. J Psychosom Res 2000; 48: 579–584.
Zigmond AS, Snaith RP . The hospital anxiety and depression scale. Acta Psychiatr Scand 1983; 67: 361–370.
Kalo J, Agergård J, Aadahl M, Visby R, Halkjær Kristensen J, Jacobsen N . Fatigue in hematological patients treated with allogeneic stem cell transplantation. J Danish Med Assoc 2007; 169/38: 3194–3197.
Saltin B, Blomqvist G, Mitchell JH, Johnson RL, Wildenthal K, Chapman CB . Response to exercise after bed rest and after training. A longitudinal study of adaptive changes in oxygen transport and body composition. Circulation 1968; 38 (Suppl 5): VII-1–VII-78.
Bloomfield SA . Changes in musculoskeletal structure and function with prolonged bed rest. Med Sci Sports Exerc 1997; 29: 197–206.
Bolton JW, Hornung CA, Olsen GN . Determinants of achievement in stair climbing as an exercise test. Mil Med 1994; 159: 644–646.
Convertino VA . Cardiovasular consequences consequences of bed rest: effect on maximal oxygen uptake. Med Sci Sports Exerc 1997; 29: 191–196.
Patrick DL, Ferketich SL, Frame PS, Harris JJ, Hendricks CB, Levin B et al. National Institutes of Health State-of-the-Science Conference Statement: symptom management in cancer: pain, depression, and fatigue, July 15–17, 2002. J Natl Cancer Inst 2003; 95: 1110–1117.
Schmitz KH, Holtzmann J, Courneya K, Masse LC, Duval S, Kane R . Controlled physical activity trials in cancer survivors: a systematic review and meta-analysis. Cancer Epidemiol Biomarkers Prev 2005; 14: 1588–1595.
Courneya KS, Segal RJ, Mackey JR, Gelmon K, Reid RD, Friedenreich CM et al. Effects of aerobic and resistance exercise in breast cancer patients receiving adjuvant chemotherapy: a multicenter randomized controlled trial. J Clin Oncol 2007; 25: 4396–4404.
Decker WA, Turner-McGlade J, Fehir KM . Psychosocial aspects and the physiological effects of a cardiopulmonary exercise program in patients undergoing bone marrow transplantation for acute leukemia. Transplant Proc 1989; 21: 3068–3069.
Hjermstad MJ, Loge JH, Evensen SA, Kvaløy SO, Fayers PM, Kaasa S . The course of anxiety and depression during the first year after allogeneic or autologuous stem cell transplantation. Bone Marrow Transplant 1999; 24: 1219–1228.
Szeulga DJ, Stuart RK, Brookmeyer R, Utermohlen V, Santos GW . Nutritional support of bone marrow parenteral nutrition to an enteral feeding program. Cancer Res 1987; 47: 3309–3316.
Asher RAJ . The dangers of going to bed. BMJ 1947; 2: 967–968.
Acknowledgements
The authors gratefully acknowledge the study participants, the staff and administration at the Department of Hematology and The University Hospitals Center for Nursing and Care Research, University Hospital of Copenhagen, Denmark. This research was supported by grants from The Lundbeck Foundation, The Novo Nordic Foundation, The Danish Cancer Society, The Copenhagen Hospital Corporation and The Danish Nursing Society.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jarden, M., Baadsgaard, M., Hovgaard, D. et al. A randomized trial on the effect of a multimodal intervention on physical capacity, functional performance and quality of life in adult patients undergoing allogeneic SCT. Bone Marrow Transplant 43, 725–737 (2009). https://doi.org/10.1038/bmt.2009.27
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2009.27
Keywords
This article is cited by
-
Effects of pulmonary rehabilitation in hematopoietic stem cell transplantation recipients: a randomized controlled study
Supportive Care in Cancer (2024)
-
Fascia training in patients undergoing allogeneic hematopoietic cell transplantation—a pilot study
Supportive Care in Cancer (2023)
-
Potential benefits of a virtual, home-based combined exercise and mindfulness training program for HSC transplant survivors: a single-arm pilot study
BMC Sports Science, Medicine and Rehabilitation (2022)
-
Impact of Resistance Exercise and Nutritional Endorsement on physical performance in patients with GvHD (IRENE-G study) – design and rational of a randomized controlled trial
BMC Cancer (2022)
-
Preventing the adverse cardiovascular consequences of allogeneic stem cell transplantation with a multi-faceted exercise intervention: the ALLO-Active trial protocol
BMC Cancer (2022)