Abstract
Background:
The relationship between diet and survival after ovarian cancer diagnosis is unclear as a result of a limited number of studies and inconsistent findings.
Methods:
We examined the association between pre-diagnostic diet and overall survival in a population-based cohort (n=811) of Australian women diagnosed with invasive epithelial ovarian cancer between 2002 and 2005. Diet was measured by validated food frequency questionnaire. Deaths were ascertained up to 31 August 2014 via medical record review and Australian National Death Index linkage. We conducted Cox proportional hazards regression analysis, controlling for diagnosis age, tumour stage, grade and subtype, residual disease, smoking status, body mass index, physical activity, marital status, and energy intake.
Results:
We observed improved survival with highest compared with lowest quartile of fibre intake (hazard ratio (HR)=0.69, 95% CI: 0.53–0.90, P-trend=0.002). There was a suggestion of better survival for women with highest compared with lowest intake category of green leafy vegetables (HR=0.79, 95% CI: 0.62–0.99), fish (HR=0.74, 95% CI: 0.57–0.95), poly- to mono-unsaturated fat ratio (HR=0.76, 95% CI: 0.59–0.98), and worse survival with higher glycaemic index (HR=1.28, 95% CI: 1.01–1.65, P-trend=0.03).
Conclusions:
The associations we observed between healthy components of diet pre-diagnosis and ovarian cancer survival raise the possibility that dietary choices after diagnosis may improve survival.
Similar content being viewed by others
Main
Ovarian cancer has the highest mortality rate of all gynaecologic cancers (Hunn and Rodriguez, 2012), with the majority of women diagnosed at more advanced disease stages. Data from population-based registries have demonstrated that survival rates have not improved appreciably over time (Horner et al, 2009; Australian Institute of Health and Welfare, 2014). Lack of early screening tools and curative chemotherapy, as well as tumour molecular heterogeneity, are challenges to controlling the burden of disease (Hunn and Rodriguez, 2012). Observations that women with the same cancer characteristics given similar treatments can have different outcomes suggest there may be factors in addition to non-modifiable cancer characteristics that can influence survival (Bandera et al, 2009; Thomson and Alberts, 2010). Efforts to identify modifiable lifestyle factors that improve ovarian cancer survival have consequently gained momentum.
The mechanisms that drive ovarian carcinogenesis are not fully characterised, but may include elevated circulating gonadotropins, sex-steroid hormones, inflammatory cytokines, and altered glucose homeostasis (Ness and Modugno, 2006; Bandera et al, 2009; Wang and Sun, 2009; Yang et al, 2012). While diet has been shown to influence these mechanistic pathways and modify the carcinogenic process for other cancers (van Kruijsdijk et al, 2009; George et al, 2010), only five relatively large (N=244–636) observational studies to our knowledge have explored the effects of diet on survival after ovarian cancer diagnosis and findings have been heterogeneous (Nagle et al, 2003; Zhang et al, 2004; Sakauchi et al, 2007; Dolecek et al, 2010; Thomson et al, 2014). Nonetheless, limited data indicate that diet may be associated with survival among women diagnosed with ovarian cancer, with some evidence of possible benefit from greater fruit and vegetable intake (Nagle et al, 2003; Dolecek et al, 2010), vitamin E (Nagle et al, 2003), green tea (Zhang et al, 2004), and overall diet quality (Thomson et al, 2014).
We agnostically explored the associations between 34 dietary exposures representing usual food, beverage, nutrient, and dietary supplement intake before diagnosis and overall survival in 811 women with invasive ovarian cancer who participated in the Australian Ovarian Cancer Study (AOCS), a nationwide case–control study.
Materials and methods
Australian ovarian cancer study
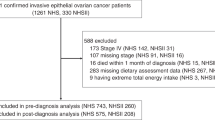
The AOCS has previously been described (Merritt et al, 2008). Briefly, women aged 18–79 years with a diagnosis of invasive or borderline epithelial ovarian, fallopian tube, or primary peritoneal cancer diagnosed between January 2002 and June 2006 were enrolled. The current analysis excluded women with borderline disease. Women were identified through gynaecologic oncology units and mandatory state-based cancer registries. Ethics approval and informed consent were obtained before enrolment. After exclusions (i.e., diagnosis not primary epithelial ovarian cancer or before study initiation, not an Australian resident), 1709 out of 2745 women agreed to participate; 1612 (94%) returned the main study questionnaire. Of these, 1132 women diagnosed with invasive disease before July 2005 also completed a food frequency questionnaire (FFQ). Women recruited in the final year of the study (n=242) were not asked to complete the FFQ. After excluding participants who omitted 14 (10%) or more FFQ line items, those with implausible caloric intakes (<700 or >4000 kcal per day), and women who reported changing their diet over the 6–12 months before diagnosis because they were asked to report their diet before it changed (n=309), dietary data were available for 811 women with invasive ovarian cancer.
Mortality ascertainment and follow-up
Vital status was determined through data abstracted from medical records every 6–12 months, and probabilistic record data linkage to the Australian National Death Index at the Australian Institute of Health and Welfare. All-cause mortality was the end point for follow-up, however, of those with cause of death information (90%, n=733), 95% of deaths were attributed to ovarian cancer. Participants were followed for a mean of 5.9±3.8 years. Survival time was defined as the interval between histologic diagnosis and date of death or censored at 31 August 2014. Left-truncated survival analyses were performed to account for the time elapsed between the date of diagnosis and study recruitment, in order to eliminate survivorship bias from excluding eligible women who died before study recruitment.
Covariates
Clinical data including International Federation of Obstetricians and Gynecologists stage (I/II and III/IV), tumour grade (well, moderately, or poorly differentiated, and missing), amount of residual tumour after surgery (none, ⩽1 cm, >1 cm, and missing), and histological subtype (serous, mucinous, endometrioid, clear cell, and other) were collected from histology reports and medical records. Socio-demographic and lifestyle information including age at diagnosis, education (school only, trade or technical qualification, and university), comorbid conditions including diabetes (none and ⩾1), smoking status (current, former, and never), height and weight (used to calculate body mass index (BMI): <18.5; 18.5–<25; 25–<30, and ⩾30 kg m−2), amount of physical activity (low, medium, and high physical activity index (PAI)), and reproductive and hormonal data were obtained via self-administered questionnaire at study enrolment. Inclusion of a variable for the time elapsed between diagnosis and dietary assessment did not alter the effect estimates of interest so this was not included in the final models.
Dietary exposure assessment
Dietary intake was measured at recruitment via a semi-quantitative, 135-item FFQ adapted from that developed by Willett et al (1985, 1987; Ashton, 1996) and validated in an Australian population (McNaughton et al, 2005; Marks et al, 2006; Ibiebele et al, 2009). Questionnaires were completed a median of 3.3 months after diagnosis and intake was assessed for the year before diagnosis. Participants reported usual frequency of intake over the previous 12 months for standard serving sizes of food items and beverages. Frequency categories included ‘<1’ or ‘1–3’ times per month; ‘1’, ‘2–4’, or ‘5–6’ times per week; and ‘1’, ‘2–3’, or ‘4+’ times per day. Nutrient intake was estimated based on the Australian Food Composition Tables (Food Standards Australia New Zealand, 2010) and energy-adjusted using the residual method (Willett et al, 1997). Participants also reported type, quantity, and frequency of intake of dietary supplements. Nutrient intake from supplements was calculated by multiplying intake frequency by nutrient content given in the Australian Register of Therapeutic Goods (Therapeutic Goods Administration, 2013) following a process reported by Ashton et al (1997). Food group variables were calculated by summing the number of standard serving sizes consumed per day of relevant items in that group (items listed in Supplementary Table S1). Food group serving size categorisation was based on the distribution of intakes in the study population. Vegetable intake excluded potatoes. Dietary supplement use was categorised as either (1) non-user and user; or (2) non-user, <50th percentile of intake among users, and >50th percentile of intake among users, where data were available. Glycaemic index (GI) and glycaemic load (GL) were calculated using the Australian GI database (FoodWorks 2007, Xyris Software, Professional Edition, Highgate Hill, QLD, Australia) supplemented by the International Tables of Glycaemic Index and Glycaemic Load values (2008) (Atkinson et al, 2008). Dietary GL was calculated by multiplying grams of carbohydrate per food serving by the GI of that food.
Statistical analysis
Crude survival probabilities were estimated using the Kaplan–Meier technique. Associations between dietary factors and overall survival were estimated using Cox proportional hazards regression; multivariate hazard ratios (HRs) and 95% CIs were generated using SAS (SAS, version 9.3; SAS Institute, Cary, NC, USA). The underlying time-metric was person-time (months) of follow-up. A formal test for proportionality was conducted by modelling the interaction between the natural logarithm of follow-up time and main dietary variables. Where the proportional hazards assumption was violated, a lag analysis was also conducted by stratifying the Cox proportional hazards models by different periods of follow-up time selected by inspecting the crude survival curves (Bellera et al, 2010). Effect estimates presented in tables are from models without the time interaction term.
A missing category was created for categorical variables with a large number of missing values. Variables associated with survival at P<0.10 in univariate models were evaluated in multivariate models. Stepwise backward selection was used to generate the most parsimonious models, retaining variables that improved model fit assessed by likelihood ratio test and remained statistically significant at P<0.05. Variables that were dropped from the model were added back in to multivariate models one at a time to confirm lack of effect on the estimates for each dietary exposure. The final multivariate models were adjusted for age at diagnosis, tumour stage and grade, amount of residual disease, subtype, smoking status, BMI, PAI, daily energy intake, and, where appropriate, marital status. Tests for linear trend were based on category median values. Finally, we conducted a sensitivity analysis, restricting to women diagnosed with high-grade (grade 3) serous tumours in order to determine diet–mortality associations within this homogenous subgroup.
Results
Clinical, socio-demographic, and lifestyle characteristics of the 811 women included in this analysis are presented in Table 1. A total of 547 (67%) women died during follow-up, 5-year survival was 59.9% and median survival was 59.6 months. Serous histological subtype, later-stage disease, moderately or poorly differentiated tumour, greater residual disease, tumour originating in the peritoneum or fallopian tube, parity, lower educational attainment, not having a current partner, and having ⩾1 comorbid conditions were statistically significantly associated with worse survival in this cohort in univariate analyses.
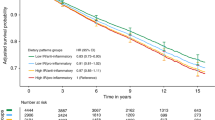
Table 2 presents the associations between diet exposures and overall mortality. There was a trend for lower mortality with higher fruit intake (P-trend=0.04). Higher intake of green leafy vegetables was inversely associated with mortality (HR=0.75, 95% CI: 0.61, 0.93; HR=0.79, 95% CI: 0.62, 0.99 for increasing intake) although a linear trend was not evident (P-trend=0.06). A similar pattern was seen for fish intake (HR for ⩾3 vs 0 servings per day=0.74, 95% CI: 0.57, 0.95, P-trend=0.17). Cruciferous vegetable and low-fat dairy intake did not meet the proportional hazard assumption. In the lag analysis considering stages of follow-up time, cruciferous vegetable and low-fat dairy intake was not associated with mortality when restricting to ⩽ or >3 years’ follow-up time. Other food groups (grains, oily fish (tuna and salmon), total meat and meat subgroups (poultry, red meat, and processed meat), and low- and high-fat dairy) were not associated with mortality. For beverages including black and green tea, and coffee, we did not observe an association with mortality.
Table 3 presents the associations between nutrient intakes (second, third, and fourth vs first quartile) and overall mortality. Improved survival was evident for highest compared with lowest quartile of fibre intake (HR=0.69, 95% CI: 0.53, 0.90, P-trend=0.002) and worse survival with higher GI (HR=1.28, 95% CI: 1.01, 1.65, P-trend=0.03). Polyunsaturated to monounsaturated fat ratio (PUFA:MUFA) was associated with lower mortality for the highest compared with lowest quartile of intake (HR=0.76, 95% CI: 0.59, 0.98, P-trend=0.06). There was a suggestive trend for increasing risk of mortality with higher intakes of saturated fat (P=0.07). No association was observed between total vitamin C, E, β-carotene, and retinol from diet plus supplements, total fat, mono or polyunsaturated fat alone, or GL and mortality.
Table 4 presents associations between dietary supplement use and overall mortality. The interaction of vitamin C intake from supplements with survival time was statistically significant. Above median intake of vitamin C (180 mg per day) from supplements was positively associated with mortality during the first 5 years after diagnosis (HR vs no use=1.36, 95% CI: 1.04, 1.78) but was not associated with mortality after 5 years. Among vitamin C supplement users, the range of intake was up to 2039 mg per day (IQR: 74–500).
We analysed 34 diet–mortality associations; adjusting for multiple comparisons would result in significant associations at a false discovery rate set at 0.2 of <0.006 (0.2/(34/1)) for the highest-ranked P-value. Thus, taking this into consideration, the inverse association between fibre and mortality remained statistically significant for this agnostic analysis.
Similar patterns were seen in sensitivity analyses restricted to women with high-grade serous tumours however the associations tended to be slightly stronger. As a result the inverse associations with total vegetable, oily fish, and green tea intake, and the positive association with total fat intake reached statistical significance (Supplementary Tables S2–S4). A significant inverse trend was also seen for cruciferous vegetables and a significant positive trend for coffee intake although none of the individual category estimates reached statistical significance.
Discussion
Our findings from a large, Australian population-based sample of women diagnosed with invasive epithelial ovarian cancer support a possible association between pre-diagnostic diet and survival after diagnosis. Overall, we observed a survival advantage for higher intakes of total and green leafy vegetables, fruit, fish, fibre intake, and PUFA:MUFA ratio, while higher GI was associated with worse survival. Higher vitamin C intake from supplements was inversely associated with survival up to 5 years after diagnosis but not after 5 years. A significant dose–response relationship (for survival advantage) was observed for both increasing fibre intake (better survival) and GI (worse survival). After controlling for multiple comparisons, the association with fibre remained statistically significant. Our findings were independent of tumour characteristics and, when we restricted to high-grade serous cases, the associations remained and we also observed a trend for reduced mortality risk with higher cruciferous vegetable and green tea intakes. These findings expand on a limited area of research as, to our knowledge, only five studies have been conducted on dietary exposures and survival after ovarian cancer diagnosis (Nagle et al, 2003; Zhang et al, 2004; Sakauchi et al, 2007; Yang et al, 2008; Dolecek et al, 2010; Thomson et al, 2014).
The World Cancer Research Fund Continuous Update Project Report on diet and risk of ovarian cancer judged the evidence that diet has a role as only limited/suggestive due to the scarcity and/or inconsistency of findings to date (World Cancer Research Fund/American Institute for Cancer Research, 2014). Nonetheless, emerging evidence supports some associations between diet and ovarian cancer survival. Findings from the Women’s Health Initiative (N=636 ovarian cancer cases) found significantly lower all-cause mortality for women reporting higher pre-diagnosis diet quality as measured by the Healthy Eating Index–2005, but no significant associations with the individual dietary components contributing to the dietary score (Thomson et al, 2014). It is possible that overall pattern of food consumption that accounts for correlated intakes and food/nutrient interactions is a more important exposure in the context of ovarian cancer as opposed to the role of single foods or nutrients. A large-scale prospective cohort study in Japan (N=64 327 women) assessed baseline frequency of intake of 32 food items and followed women for an average of 13 years; 77 women died of ovarian cancer. Salted fish and pickled Chinese cabbage were positively associated with ovarian cancer mortality but no other dietary associations were observed (Sakauchi et al, 2007). As survival was not the focus of this cohort, it is unclear how these dietary exposures were associated with ovarian cancer incidence and subsequent survival. A longitudinal study of 341women enrolled in a United States (US) case–control study between 1994 and 1998 found better survival for women with higher intake of fruits plus vegetables, total vegetables, and yellow and cruciferous vegetables pre-diagnosis (Dolecek et al, 2010). They also observed poorer survival with higher intakes of red and cured meats, and milk. Those findings were generally supported by another Australian study of 609 women diagnosed between 1990 and 1993, where better survival was observed with higher pre-diagnosis intakes of vegetables, cruciferous vegetables, and vitamin E, whereas lactose, dairy products, protein, red meat, and white meat were associated with worse survival (Nagle et al, 2003).
Some of these associations for individual foods were not observed in our current population; however, there are a number of differences between the studies. Compared with the current analysis, the US sample was slightly younger with earlier disease at diagnosis. Dietary intake was reported for the 3- to 5-year period before diagnosis as opposed to 12 months in this study, and the previous Australian study (Nagle et al, 2003; Dolecek et al, 2010). Similarly, participants in the first Australian study were younger with earlier-stage disease (Nagle et al, 2003). However, when we restricted analyses to participants aged <50 years, results were not altered significantly with regards to these dietary components. As cases in the current study were diagnosed 10 years after the first Australian study, changes in food consumption patterns could have influenced heterogeneity of exposure (Flood et al, 2010; Arabshahi et al, 2011), although intakes appeared similar to those in the previous study.
We observed an inverse association between green leafy vegetables (spinach and lettuces) and mortality, and a trend for reduced mortality risk with higher fruit intake. Spinach is rich in beta-carotene, lutein/zeaxanthin, and other vitamins and minerals. While two population-based case–control studies have reported reduced risks of ovarian cancer for the highest compared with lowest intakes of lutein/zeaxanthin (Bertone et al, 2001), two prospective cohorts found no association between green leafy vegetable intake and incident epithelial ovarian cancer (Larsson et al, 2004; Schulz et al, 2005). A previous analysis reported an inverse association between pre-diagnosis intake of fruit and vegetables combined and ovarian cancer survival (Dolecek et al, 2010), but analyses of diet and survival after diagnosis of ovarian cancer to date have grouped spinach/lettuce intake with total vegetable intake (Nagle et al, 2003; Dolecek et al, 2010). In addition to fibre, green leafy vegetables are also a good source of other nutrients shown to be implicated in cancer aetiology, including folate and flavonoids, although evidence for their roles in ovarian cancer survival is sparse (Zhang et al, 2004; Dixon et al, 2014).
For total fish intake, we observed a protective association with survival, but no dose–response relationship. In two Australian case–control studies, higher fish intake was associated with lower risk (Kolahdooz et al, 2010) but a meta-analysis of 15 case–control and cohort studies found that total fish consumption was not associated with ovarian cancer risk overall, although it was protective in Australian/European but not in Asian/North American studies (Jiang et al, 2014). Mechanisms proposed to explain the protective effect of fish intake include the rich source of omega-3 fatty acids and their anti-apoptotic and anti-inflammatory properties (Fernandez et al, 1999; Sharma et al, 2005). Fish has not previously been analysed independently of other white meats in relation to ovarian cancer survival (Nagle et al, 2003; Dolecek et al, 2010).
We observed a beneficial effect of dietary fibre, which lowers circulating oestrogens by inhibiting bile reabsorption and increasing faecal excretion (Ferrari et al, 2013), on survival. In the previous Australian analysis, fibre was not associated with ovarian cancer survival (Nagle et al, 2003), although the median of the highest-intake category was lower than the current analysis (35 vs 43 g per day). We found a similarly null association for fibre intake of 35 g per day, suggesting a protective effect only for particularly high levels of fibre intake. To date, no other studies have explored this association. Findings for the association between fibre intake and ovarian cancer risk have been mixed (Silvera et al, 2007a; Hedelin et al, 2011), although fibre intake has been associated with lower breast cancer mortality (Belle et al, 2011; Buck et al, 2011). A high-fibre, low-fat dietary intervention was shown to reduce bioavailable oestrogen among women diagnosed with breast cancer, a plausible mechanism in the context of improving ovarian cancer survival (Rock et al, 2004). Other potential mechanisms for fibre’s inverse association with mortality include its influence on inflammation (Ma et al, 2008; Villasenor et al, 2011), and metabolic regulation including effects on GI (Lattimer and Haub, 2010).
We found suggestive evidence for a trend towards poorer survival with increasing saturated fat intake, which has not been observed in prior studies (Nagle et al, 2003; Dolecek et al, 2010). High dietary fat intake has been proposed to increase circulating progesterone and oestrogens, promoting tumour development (Pyragius et al, 2013). Previous investigations highlight the inconsistency of this association (Genkinger et al, 2006; Blank et al, 2012).
The ratio of PUFA:MUFA was inversely associated with mortality. PUFA, largely found in nuts, seeds, fish, and leafy green vegetables, includes both omega-3 (n-3) and omega-6 (n-6) subtypes; avocado, canola, and olive oils are good sources of MUFA. A large population-based case–control study (n=1872 cases and 1978 controls) found a lower risk of epithelial ovarian cancer for the highest compared with lowest intakes of overall PUFA, n-3 and n-6 PUFA, but no association with MUFA (Merritt et al, 2014). The ratio of PUFA:MUFA was not associated with risk for developing ovarian cancer in a second study (Ibiebele et al, 2012). Unsaturated fatty acids have a role in cell membrane integrity and can affect cell function through eicosanoid synthesis, gene expression regulation, and effects on apoptosis and cell proliferation (Abel et al, 2014). Previous studies have shown that n-3 FA inhibit ovarian cancer cell proliferation (Sharma et al, 2005; Sharma et al, 2009), but the importance of n-6:n-3 and PUFA:MUFA in the context of carcinogenesis remains unclear (Abel et al, 2014). Prior studies of diet and ovarian cancer survival have not assessed unsaturated fatty acid ratio (Nagle et al, 2003; Zhang et al, 2004; Sakauchi et al, 2007; Yang et al, 2008; Dolecek et al, 2010).
GI showed a positive linear relationship with mortality. Previous case–control and prospective cohort studies have reported mixed findings for the association between GI and ovarian cancer risk (Augustin et al, 2003; Silvera et al, 2007b; George et al, 2009; Nagle et al, 2011). The GI quantitatively assesses the effect of food on post-consumption blood glucose levels and is purported to influence carcinogenesis by increasing circulating insulin-like growth factors and oestrogens, and promoting tumour progression through insulin-related cell signalling (George et al, 2009). Studies on ovarian cancer survival are lacking.
Vitamin C supplement intake was positively associated with mortality for the earlier period of follow-up (up to 5 years) but we found no association with vitamin C from diet alone. Previous investigations on ovarian cancer survival have not reported on supplemental vitamin C and findings for the association with ovarian cancer risk have been mixed (Fleischauer et al, 2001; Chang et al, 2008; Crane et al, 2014; Koushik et al, 2015). A null association between vitamin C from food and ovarian cancer risk but a harmful effect for vitamin C supplement use has also been reported (Gifkins et al, 2012). Findings from the WHI among vitamin C replete postmenopausal women also showed that women who went on to develop ovarian cancer had significantly higher vitamin C supplement intake than controls. Proposed mechanisms included vitamin C pro-oxidant activity and enhanced iron absorption with iron-associated oxidative stress and effects on malignant transformation (Thomson et al, 2008). Future studies powered to explore dietary supplement intake and ovarian cancer survival with characterisation of dose, formulation, and duration are warranted.
Our study has several strengths, including its large sample size, long duration and complete follow-up, population-based case selection and detailed collection of information on tumour characteristics, and lifestyle factors, allowing adjustment for known prognostic factors. There was comprehensive measurement of food, nutrient, and dietary supplement exposures. Some limitations should be addressed. Diet was measured pre-diagnosis and may not reflect intake after diagnosis. Nevertheless, although some women may change their diet after cancer diagnosis, overall, pre-diagnosis diet is likely to be correlated with post-diagnosis diet. Studies of breast cancer survivors suggest dietary changes after diagnosis are modest (Wayne et al, 2004), and limited post-diagnosis dietary data available for 289 women in our cohort showed moderate correlations for fruit, vegetables, and meat (intraclass correlations 0.46, 0.32, and 0.42, respectively). Furthermore, we restricted the current analysis to women who reported no change in their diet for the 6–12 months before diagnosis. There is the potential for selection bias if the association between diet and survival differs by characteristics of participants compared with non-participants, given that enrolment was limited to those well enough to complete dietary assessment. However, this cohort included women who diagnosed primarily with later-stage disease, reflecting the general population of ovarian cancer survivors. Information or recall bias is a possible limitation as diagnosis of late-stage disease with reduced wellbeing may have influenced dietary recall, although our results were adjusted for stage and grade of disease, and we observed similar associations by disease stage. Any measurement error, inherent in self-report measures for dietary assessment, is likely to have attenuated real associations and thus cannot explain the associations seen. Given the observational design, it is possible that observed associations are due to confounding by unknown or unmeasured confounding factors although we adjusted for the key factors known to influence ovarian cancer survival. Results may be due to chance; after controlling for multiple comparisons, only fibre remained statistically significantly inversely associated with mortality.
In summary, in one of few dietary analyses within a large, observational study of women diagnosed with invasive ovarian cancer, we observed improved survival with higher intakes of dietary fibre. There was a suggestion of improved survival with higher consumption of green leafy vegetables, fish, and PUFA:MUFA ratio, and poorer survival with higher GI that require replication in future large-scale prospective studies. These observations that healthy components of a pre-diagnosis diet are associated with ovarian cancer survival raise the possibility that healthful dietary choices after diagnosis may improve ovarian cancer survival. The role of diet in ovarian cancer survival would be further clarified by analyses of post-diagnosis diet, evidence for which is currently being gathered in a randomised, controlled (Thomson et al, 2016).
Change history
06 June 2017
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Abel S, Riedel S, Gelderblom WC (2014) Dietary PUFA and cancer. Proc Nutr Soc 73 (3): 361–367.
Arabshahi S, Lahmann PH, Williams GM, Marks GC, van der Pols JC (2011) Longitudinal change in diet quality in Australian adults varies by demographic, socio-economic, and lifestyle characteristics. J Nutr 141 (10): 1871–1879.
Ashton B (1996) Under–reporting of energy intake in two methods of dietary assessment in the Nambour Trial. Aust J Nutr Diet 53: 53–60.
Ashton BA, Ambrosini GL, Marks GC, Harvey PW, Bain C (1997) Development of a dietary supplement database. Aust N Z J Public Health 21 (7): 699–702.
Atkinson FS, Foster-Powell K, Brand-Miller JC (2008) International tables of glycemic index and glycemic load values: 2008. Diabetes Care 31 (12): 2281–2283.
Augustin LS, Polesel J, Bosetti C, Kendall CW, La Vecchia C, Parpinel M, Conti E, Montella M, Franceschi S, Jenkins DJ, Dal Maso L (2003) Dietary glycemic index, glycemic load and ovarian cancer risk: a case-control study in Italy. Ann Oncol 14 (1): 78–84.
Australian Institute of Health and Welfare (2014) Cancer in Australia: An Overview 2014 Cancer Series No 90. Catalogue No CAN88 AIHW: Canberra, Australia.
Bandera EV, Kushi LH, Rodriguez-Rodriguez L (2009) Nutritional factors in ovarian cancer survival. Nutr Cancer 61 (5): 580–586.
Belle FN, Kampman E, McTiernan A, Bernstein L, Baumgartner K, Baumgartner R, Ambs A, Ballard-Barbash R, Neuhouser ML (2011) Dietary fiber, carbohydrates, glycemic index, and glycemic load in relation to breast cancer prognosis in the HEAL cohort. Cancer Epidemiol Biomarkers Prev 20 (5): 890–899.
Bellera CA, MacGrogan G, Debled M, de Lara CT, Brouste V, Mathoulin-Pelissier S (2010) Variables with time-varying effects and the Cox model: some statistical concepts illustrated with a prognostic factor study in breast cancer. BMC Med Res Methodol 10: 20.
Bertone ER, Hankinson SE, Newcomb PA, Rosner B, Willet WC, Stampfer MJ, Egan KM (2001) A population-based case-control study of carotenoid and vitamin A intake and ovarian cancer (United States). Cancer Causes Control 12 (1): 83–90.
Blank MM, Wentzensen N, Murphy MA, Hollenbeck A, Park Y (2012) Dietary fat intake and risk of ovarian cancer in the NIH-AARP Diet and Health Study. Br J Cancer 106 (3): 596–602.
Buck K, Zaineddin AK, Vrieling A, Heinz J, Linseisen J, Flesch-Janys D, Chang-Claude J (2011) Estimated enterolignans, lignan-rich foods, and fibre in relation to survival after postmenopausal breast cancer. Br J Cancer 105 (8): 1151–1157.
Chang ET, Lee VS, Canchola AJ, Dalvi TB, Clarke CA, Reynolds P, Purdie DM, Stram DO, West DW, Ziogas A, Bernstein L, Horn-Ross PL (2008) Dietary patterns and risk of ovarian cancer in the California Teachers Study cohort. Nutr Cancer 60 (3): 285–291.
Crane TE, Khulpateea BR, Alberts DS, Basen-Engquist K, Thomson CA (2014) Dietary intake and ovarian cancer risk: a systematic review. Cancer Epidemiol Biomarkers Prev 23 (2): 255–273.
Dixon SC, Ibiebele TI, Protani MM, Beesley J, deFazio A, Crandon AJ, Gard GB, Rome RM, Webb PM, Nagle CM Australian Ovarian Cancer Study G (2014) Dietary folate and related micronutrients, folate-metabolising genes, and ovarian cancer survival. Gynecol Oncol 132 (3): 566–572.
Dolecek TA, McCarthy BJ, Joslin CE, Peterson CE, Kim S, Freels SA, Davis FG (2010) Prediagnosis food patterns are associated with length of survival from epithelial ovarian cancer. J Am Diet Assoc 110 (3): 369–382.
Fernandez E, Chatenoud L, La Vecchia C, Negri E, Franceschi S (1999) Fish consumption and cancer risk. Am J Clin Nutr 70 (1): 85–90.
Ferrari P, Rinaldi S, Jenab M, Lukanova A, Olsen A, Tjonneland A, Overvad K, Clavel-Chapelon F, Fagherazzi G, Touillaud M, Kaaks R, von Rusten A, Boeing H, Trichopoulou A, Lagiou P, Benetou V, Grioni S, Panico S, Masala G, Tumino R, Polidoro S, Bakker MF, van Gils CH, Ros MM, Bueno-de-Mesquita HB, Krum-Hansen S, Engeset D, Skeie G, Pilar A, Sanchez MJ, Buckland G, Ardanaz E, Chirlaque D, Rodriguez L, Travis R, Key T, Khaw KT, Wareham NJ, Sund M, Lenner P, Slimani N, Norat T, Aune D, Riboli E, Romieu I (2013) Dietary fiber intake and risk of hormonal receptor-defined breast cancer in the European Prospective Investigation into Cancer and Nutrition study. Am J Clin Nutr 97 (2): 344–353.
Fleischauer AT, Olson SH, Mignone L, Simonsen N, Caputo TA, Harlap S (2001) Dietary antioxidants, supplements, and risk of epithelial ovarian cancer. Nutr Cancer 40 (2): 92–98.
Flood VM, Burlutsky G, Webb KL, Wang JJ, Smith WT, Mitchell P (2010) Food and nutrient consumption trends in older Australians: a 10-year cohort study. Eur J Clin Nutr 64 (6): 603–613.
Food Standards Australia New Zealand (2010) NUTTAB 2010 – Australian Food Composition Tables. Food Standards Australia New Zealand: Canberra, Australia, Available at http://www.foodstandards.gov.au/science/monitoringnutrients/nutrientables/nuttab/Pages/default.aspx.
Genkinger JM, Hunter DJ, Spiegelman D, Anderson KE, Beeson WL, Buring JE, Colditz GA, Fraser GE, Freudenheim JL, Goldbohm RA, Hankinson SE, Koenig KL, Larsson SC, Leitzmann M, McCullough ML, Miller AB, Rodriguez C, Rohan TE, Ross JA, Schatzkin A, Schouten LJ, Smit E, Willett WC, Wolk A, Zeleniuch-Jacquotte A, Zhang SM, Smith-Warner SA (2006) A pooled analysis of 12 cohort studies of dietary fat, cholesterol and egg intake and ovarian cancer. Cancer Causes Control 17 (3): 273–285.
George SM, Mayne ST, Leitzmann MF, Park Y, Schatzkin A, Flood A, Hollenbeck A, Subar AF (2009) Dietary glycemic index, glycemic load, and risk of cancer: a prospective cohort study. Am J Epidemiol 169 (4): 462–472.
George SM, Neuhouser ML, Mayne ST, Irwin ML, Albanes D, Gail MH, Alfano CM, Bernstein L, McTiernan A, Reedy J, Smith AW, Ulrich CM, Ballard-Barbash R (2010) Postdiagnosis diet quality is inversely related to a biomarker of inflammation among breast cancer survivors. Cancer Epidemiol Biomarkers Prev 19 (9): 2220–2228.
Gifkins D, Olson SH, Paddock L, King M, Demissie K, Lu SE, Kong AN, Rodriguez-Rodriguez L, Bandera EV (2012) Total and individual antioxidant intake and risk of epithelial ovarian cancer. BMC Cancer 12: 211.
Hedelin M, Lof M, Andersson TM, Adlercreutz H, Weiderpass E (2011) Dietary phytoestrogens and the risk of ovarian cancer in the women's lifestyle and health cohort study. Cancer Epidemiol Biomarkers Prev 20 (2): 308–317.
Horner MJ, Ries LAG, Krapcho M, Neyman N, Aminou R, Howlader N, Altekruse SF, Feuer EJ, Huang L, Mariotto A, Miller BA, Lewis DR, Eisner MP, Stinchcomb DG, Edwards BK (eds). (2009) SEER Cancer Statistics Review, 1975–2006. National Cancer Institute: Bethesda, MD, USA. Based on November 2008 SEER data submission, posted to the SEER web site..
Hunn J, Rodriguez GC (2012) Ovarian cancer: etiology, risk factors, and epidemiology. Clin Obstet Gynecol 55 (1): 3–23.
Ibiebele TI, Nagle CM, Bain CJ, Webb PM (2012) Intake of omega-3 and omega-6 fatty acids and risk of ovarian cancer. Cancer Causes Control 23 (11): 1775–1783.
Ibiebele TI, Parekh S, Mallitt KA, Hughes MC, O'Rourke PK, Webb PM Australian Ovarian Cancer Study G the Australian Cancer S (2009) Reproducibility of food and nutrient intake estimates using a semi-quantitative FFQ in Australian adults. Public Health Nutr 12 (12): 2359–2365.
Jiang PY, Jiang ZB, Shen KX, Yue Y (2014) Fish intake and ovarian cancer risk: a meta-analysis of 15 case-control and cohort studies. PLoS One 9 (4): e94601.
Kolahdooz F, van der Pols JC, Bain CJ, Marks GC, Hughes MC, Whiteman DC, Webb PM Australian Cancer S the Australian Ovarian Cancer Study G (2010) Meat, fish, and ovarian cancer risk: results from 2 Australian case-control studies, a systematic review, and meta-analysis. Am J Clin Nutr 91 (6): 1752–1763.
Koushik A, Wang M, Anderson KE, van den Brandt P, Clendenen TV, Eliassen AH, Freudenheim JL, Genkinger JM, Hakansson N, Marshall JR, McCullough ML, Miller AB, Robien K, Rohan TE, Schairer C, Schouten LJ, Tworoger SS, Wang Y, Wolk A, Zeleniuch-Jacquotte A, Smith-Warner SA (2015) Intake of vitamins A, C, and E and folate and the risk of ovarian cancer in a pooled analysis of 10 cohort studies. Cancer Causes Control 26 (9): 1315–1327.
Larsson SC, Holmberg L, Wolk A (2004) Fruit and vegetable consumption in relation to ovarian cancer incidence: the Swedish Mammography Cohort. Br J Cancer 90 (11): 2167–2170.
Lattimer JM, Haub MD (2010) Effects of dietary fiber and its components on metabolic health. Nutrients 2 (12): 1266–1289.
Ma Y, Hebert JR, Li W, Bertone-Johnson ER, Olendzki B, Pagoto SL, Tinker L, Rosal MC, Ockene IS, Ockene JK, Griffith JA, Liu S (2008) Association between dietary fiber and markers of systemic inflammation in the Women’s Health Initiative Observational Study. Nutrition 24 (10): 941–949.
Marks GC, Hughes MC, van der Pols JC (2006) The effect of personal characteristics on the validity of nutrient intake estimates using a food-frequency questionnaire. Public Health Nutr 9 (3): 394–402.
McNaughton SA, Marks GC, Gaffney P, Williams G, Green A (2005) Validation of a food-frequency questionnaire assessment of carotenoid and vitamin E intake using weighed food records and plasma biomarkers: the method of triads model. Eur J Clin Nutr 59 (2): 211–218.
Merritt MA, Cramer DW, Missmer SA, Vitonis AF, Titus LJ, Terry KL (2014) Dietary fat intake and risk of epithelial ovarian cancer by tumour histology. Br J Cancer 110 (5): 1392–1401.
Merritt MA, Green AC, Nagle CM, Webb PM (2008) Talcum powder, chronic pelvic inflammation and NSAIDs in relation to risk of epithelial ovarian cancer. Int J Cancer 122 (1): 170–176.
Nagle CM, Kolahdooz F, Ibiebele TI, Olsen CM, Lahmann PH, Green AC, Webb PM Australian Cancer S the Australian Ovarian Cancer Study G (2011) Carbohydrate intake, glycemic load, glycemic index, and risk of ovarian cancer. Ann Oncol 22 (6): 1332–1338.
Nagle CM, Purdie DM, Webb PM, Green A, Harvey PW, Bain CJ (2003) Dietary influences on survival after ovarian cancer. Int J Cancer 106 (2): 264–269.
Ness RB, Modugno F (2006) Endometriosis as a model for inflammation-hormone interactions in ovarian and breast cancers. Eur J Cancer 42 (6): 691–703.
Pyragius CE, Fuller M, Ricciardelli C, Oehler MK (2013) Aberrant lipid metabolism: an emerging diagnostic and therapeutic target in ovarian cancer. Int J Mol Sci 14 (4): 7742–7756.
Rock CL, Flatt SW, Thomson CA, Stefanick ML, Newman VA, Jones LA, Natarajan L, Ritenbaugh C, Hollenbach KA, Pierce JP, Chang RJ (2004) Effects of a high-fiber, low-fat diet intervention on serum concentrations of reproductive steroid hormones in women with a history of breast cancer. J Clin Oncol 22 (12): 2379–2387.
Sakauchi F, Khan MM, Mori M, Kubo T, Fujino Y, Suzuki S, Tokudome S, Tamakoshi A Group JS (2007) Dietary habits and risk of ovarian cancer death in a large-scale cohort study (JACC study) in Japan. Nutr Cancer 57 (2): 138–145.
Schulz M, Lahmann PH, Boeing H, Hoffmann K, Allen N, Key TJ, Bingham S, Wirfalt E, Berglund G, Lundin E, Hallmans G, Lukanova A, Martinez Garcia C, Gonzalez CA, Tormo MJ, Quiros JR, Ardanaz E, Larranaga N, Lund E, Gram IT, Skeie G, Peeters PH, van Gils CH, Bueno-de-Mesquita HB, Buchner FL, Pasanisi P, Galasso R, Palli D, Tumino R, Vineis P, Trichopoulou A, Kalapothaki V, Trichopoulos D, Chang-Claude J, Linseisen J, Boutron-Ruault MC, Touillaud M, Clavel-Chapelon F, Olsen A, Tjonneland A, Overvad K, Tetsche M, Jenab M, Norat T, Kaaks R, Riboli E (2005) Fruit and vegetable consumption and risk of epithelial ovarian cancer: the European Prospective Investigation into Cancer and Nutrition. Cancer Epidemiol Biomarkers Prev 14 (11 Pt 1): 2531–2535.
Sharma A, Belna J, Espat J, Rodriguez G, Cannon VT, Hurteau JA (2009) Effects of omega-3 fatty acids on components of the transforming growth factor beta-1 pathway: implication for dietary modification and prevention in ovarian cancer. Am J Obstet Gynecol 200 (5): 516 e1–516 e6.
Sharma A, Belna J, Logan J, Espat J, Hurteau JA (2005) The effects of omega-3 fatty acids on growth regulation of epithelial ovarian cancer cell lines. Gynecol Oncol 99 (1): 58–64.
Silvera SA, Jain M, Howe GR, Miller AB, Rohan TE (2007a) Dietary fiber intake and ovarian cancer risk: a prospective cohort study. Cancer Causes Control 18 (3): 335–341.
Silvera SA, Jain M, Howe GR, Miller AB, Rohan TE (2007b) Glycaemic index, glycaemic load and ovarian cancer risk: a prospective cohort study. Public Health Nutr 10 (10): 1076–1081.
Therapeutic Goods Administration (2013) Australian Register of Therapeutic Goods (ARTG) Vol. 2013 Available at https://www.tga.gov.au/searching-australian-register-therapeutic-goods-artg/.
Thomson CA, Alberts DS (2010) Diet and survival after ovarian cancer: where are we and what's next? J Am Diet Assoc 110 (3): 366–368.
Thomson CA, Crane TE, Miller A, Garcia DO, Basen-Engquist K, Alberts DS (2016) A randomized trial of diet and physical activity in women treated for stage II-IV ovarian cancer: rationale and design of the Lifestyle Intervention for Ovarian Cancer Enhanced Survival (LIVES): an NRG Oncology/Gynecologic Oncology Group (GOG-225) Study. Contemp Clin Trials 49: 181–189.
Thomson CA, Neuhouser ML, Shikany JM, Caan BJ, Monk BJ, Mossavar-Rahmani Y, Sarto G, Parker LM, Modugno F, Anderson GL (2008) The role of antioxidants and vitamin A in ovarian cancer: results from the Women's Health Initiative. Nutr Cancer 60 (6): 710–719.
Thomson CA, E Crane T, Wertheim BC, Neuhouser ML, Li W, Snetselaar LG, Basen-Engquist KM, Zhou Y, Irwin ML (2014) Diet quality and survival after ovarian cancer: results from the Women’s Health Initiative. J Natl Cancer Inst 106 (11): dju314.
van Kruijsdijk RC, van der Wall E, Visseren FL (2009) Obesity and cancer: the role of dysfunctional adipose tissue. Cancer Epidemiol Biomarkers Prev 18 (10): 2569–2578.
Villasenor A, Ambs A, Ballard-Barbash R, Baumgartner KB, McTiernan A, Ulrich CM, Neuhouser ML (2011) Dietary fiber is associated with circulating concentrations of C-reactive protein in breast cancer survivors: the HEAL study. Breast Cancer Res Treat 129 (2): 485–494.
Wang CS, Sun CF (2009) C-reactive protein and malignancy: clinico-pathological association and therapeutic implication. Chang Gung Med J 32 (5): 471–482.
Wayne SJ, Lopez ST, Butler LM, Baumgartner KB, Baumgartner RN, Ballard-Barbash R (2004) Changes in dietary intake after diagnosis of breast cancer. J Am Diet Assoc 104 (10): 1561–1568.
Willett WC, Howe GR, Kushi LH (1997) Adjustment for total energy intake in epidemiologic studies. Am J Clin Nutr 65 (4 Suppl): 1220S–1228S, discussion 1229S–1231S.
Willett WC, Reynolds RD, Cottrell-Hoehner S, Sampson L, Browne ML (1987) Validation of a semi-quantitative food frequency questionnaire: comparison with a 1-year diet record. J Am Diet Assoc 87 (1): 43–47.
Willett WC, Sampson L, Stampfer MJ, Rosner B, Bain C, Witschi J, Hennekens CH, Speizer FE (1985) Reproducibility and validity of a semiquantitative food frequency questionnaire. Am J Epidemiol 122 (1): 51–65.
World Cancer Research Fund/American Institute for Cancer Research (2014) Continuous Update Project Report. Food, Nutrition, Physical Activity, and the Prevention of Ovarian Cancer 2014 Available at http://www.dietandcancerreport.org/cup/cup_resources.php.
Yang HP, Trabert B, Murphy MA, Sherman ME, Sampson JN, Brinton LA, Hartge P, Hollenbeck A, Park Y, Wentzensen N (2012) Ovarian cancer risk factors by histologic subtypes in the NIH-AARP Diet and Health Study. Int J Cancer 131 (4): 938–948.
Yang L, Klint A, Lambe M, Bellocco R, Riman T, Bergfeldt K, Persson I, Weiderpass E (2008) Predictors of ovarian cancer survival: a population-based prospective study in Sweden. Int J Cancer 123 (3): 672–679.
Zhang M, Lee AH, Binns CW, Xie X (2004) Green tea consumption enhances survival of epithelial ovarian cancer. Int J Cancer 112 (3): 465–469.
Acknowledgements
We acknowledge the members of the AOCS Group, including all the clinical and scientific collaborators, participating institutions, study nurses, and research assistants (see http://www.aocstudy.org/ for a complete listing), and thank all of the women who participated in the study. We also acknowledge staff at the Australian Institute of Health and Welfare for conducting the linkage to the Australian National Death Index and Maria Celia Hughes from the QIMR Berghofer Medical Research Institute for preparation of the dietary data. The Australian Ovarian Cancer Study was supported by the U.S. Army Medical Research and Materiel Command (DAMD17-01-1-0729), the National Health and Medical Research Council (NHMRC) of Australia (Grants 400281, 400413, and 199600), the Cancer Councils of Victoria, Queensland, New South Wales, South Australia, and Tasmania, and the Cancer Foundation of Western Australia. MCP was supported by National Cancer Institute Cancer Prevention Training Grant T32 CA105666, MMP by an Australian Postgraduate Award, CMN and TII by NHMRC Program Grant #552429, and PMW by a fellowship from the NHMRC. Funders had no role in the design, analysis, or writing of this article. The opinions and conclusions expressed in this article are solely the views of the authors and do not necessarily reflect those of the Food and Drug Administration (STM).
Author contributions
MCP conducted statistical analysis and wrote the paper; CMN contributed to study design and statistical analysis; TII was responsible for the dietary data and estimation of nutrient intakes; MMP conducted the linkage to the National Death Index; JC, SH, DN, and JN contributed to participant recruitment and data collection; LMF and STM contributed to statistical analysis; and PMW designed the study and has primary responsibility for the final content. All authors reviewed and approved the final manuscript.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Supplementary Information accompanies this paper on British Journal of Cancer website
Supplementary information
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Playdon, M., Nagle, C., Ibiebele, T. et al. Pre-diagnosis diet and survival after a diagnosis of ovarian cancer. Br J Cancer 116, 1627–1637 (2017). https://doi.org/10.1038/bjc.2017.120
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2017.120
Keywords
This article is cited by
-
Pre-diagnosis tea and coffee consumption and survival after a diagnosis of ovarian cancer: results from the Ovarian Cancer Association Consortium
British Journal of Cancer (2024)
-
Pre-diagnosis and post-diagnosis dietary patterns and survival in women with ovarian cancer
British Journal of Cancer (2022)
-
Association between dietary intake and risk of ovarian cancer: a systematic review and meta-analysis
European Journal of Nutrition (2021)
-
Carbohydrate Nutrition and the Risk of Cancer
Current Nutrition Reports (2019)
-
Integrative Medizin bei Patientinnen mit Ovarialkarzinomen
Der Onkologe (2019)