Abstract
Background:
Foxp3+ regulatory T cells (Tregs) play a vital role in preventing autoimmunity, but also suppress antitumour immune responses. Tumour infiltration by Tregs has strong prognostic significance in colorectal cancer, and accumulating evidence suggests that chemotherapy and radiotherapy efficacy has an immune-mediated component. Whether Tregs play an inhibitory role in chemoradiotherapy (CRT) response in rectal cancer remains unknown.
Methods:
Foxp3+, CD3+, CD4+, CD8+ and IL-17+ cell density in post-CRT surgical samples from 128 patients with rectal cancer was assessed by immunohistochemistry. The relationship between T-cell subset densities and clinical outcome (tumour regression and survival) was evaluated.
Results:
Stromal Foxp3+ cell density was strongly associated with tumour regression grade (P=0.0006). A low stromal Foxp3+ cell density was observed in 84% of patients who had a pathologic complete response (pCR) compared with 41% of patients who did not (OR: 7.56, P=0.0005; OR: 5.27, P=0.006 after adjustment for presurgery clinical factors). Low stromal Foxp3+ cell density was also associated with improved recurrence-free survival (HR: 0.46, P=0.03), although not independent of tumour regression grade.
Conclusions:
Regulatory T cells in the tumour microenvironment may inhibit response to neoadjuvant CRT and may represent a therapeutic target in rectal cancer.
Similar content being viewed by others
Main
Neoadjuvant (preoperative) chemoradiotherapy (CRT) is used in locally advanced rectal cancer to reduce tumour volume, making it more amenable to resection, and to decrease the risk of local recurrence (Bosset et al, 2006; Gerard et al, 2006; Sauer et al, 2012). Although neoadjuvant CRT followed by surgery is now standard treatment in many centres around the world, response to CRT varies considerably between individuals. Pathological complete response (pCR), defined as no remaining viable tumour cells in the surgical specimen on histopathologic assessment, is obtained in ∼20% of patients, and is associated with a reduced risk of distant recurrence and improved long-term survival (de Campos-Lobato et al, 2011; Martin et al, 2012; Zorcolo et al, 2012). However, ∼40% of patients experience only mild to moderate tumour regression, or even no regression following neoadjuvant CRT (Huebner et al, 2012; Agarwal et al, 2013; Fokas et al, 2014), and factors associated with CRT response remain poorly understood.
The presence of tumour-infiltrating lymphocytes (TILs) is associated with improved clinical outcome in many cancers (Cho et al, 2003; Zhang et al, 2003; Hiraoka et al, 2006; Loi et al, 2013). In colorectal cancer, the density of tumour-infiltrating T cells is a strong prognostic indicator, even after adjustment for clinical and molecular risk factors (Pages et al, 2005; Galon et al, 2006; Nosho et al, 2010). The recently developed Immunoscore, which involves assessment of T-cell density in the central tumour and invasive margin based on expression of pairs of T-cell markers (CD3 and CD8, CD3 and CD45RO or CD8 and CD45RO) (Pages et al, 2009), may be more prognostic than histopathologic TNM stage for patients with stage I–III disease (Mlecnik et al, 2011), and is currently undergoing international validation with a view to its incorporation into routine histopathologic practice (Galon et al, 2012). In rectal cancer, the CD3/CD8 Immunoscore is independently prognostic in patients treated with surgery alone (Anitei et al, 2014).
The intratumoural or peritumoural density of total T cells and CD8 T cells has been almost universally associated with good prognosis, whereas tumour infiltration by Foxp3+ regulatory T cells (Tregs) is associated with poorer outcome in most solid cancers (reviewed in Fridman et al, 2012). This is consistent with their well-established role as suppressors of the antitumour immune response (reviewed in Zou 2006). However, in colorectal cancer, the presence of Foxp3+ cells in the local tumour microenvironment has been associated with good prognosis in several independent studies (Salama et al, 2009; Correale et al, 2010; Frey et al, 2010). One explanation for apparent paradox is that expansion of Tregs within the tumour occurs in response to the activation and expansion of tumour-specific CD8+ T cells, thereby representing an indirect measure of an antitumour immune response. The strong positive correlation between CD8+ and Foxp3+ TILs in colorectal cancer supports this theory (Michel et al, 2008; Frey et al, 2010; Yoon et al, 2012), as does the association between Foxp3+ TILs and good prognosis in oestrogen receptor-negative breast cancer, seen only in patients who also had a high density of CD8+ TILs (West et al, 2013). Alternatively, Tregs may limit the tumourigenic effects of Th17 cell-mediated inflammation (Liu et al, 2011; Tosolini et al, 2011).
There is now abundant evidence that both chemotherapy and radiotherapy can enhance antitumour immunity through a variety of mechanisms (Nowak et al, 2003; Ghiringhelli et al, 2007; Zhang et al, 2007; Ramakrishnan et al, 2010; Burnette et al, 2011; Gupta et al, 2012; Kim et al, 2014). Notably, 5-fluorouracil (5-FU), the radiosensitising agent currently used in neoadjuvant CRT for locally advanced rectal cancer, has been shown to induce immunogenic tumour cell death (Frey et al, 2012), and selective Treg depletion has been reported in patients with metastatic colorectal cancer following 5-FU-based chemotherapy (Maeda et al, 2011). Modulation of T-cell subsets may therefore play a significant role in response to neoadjuvant CRT. Indeed, high CD3 and CD8 expression in pretreatment rectal cancer biopsy samples is associated with tumour regression following CRT (Yasuda et al, 2011; Anitei et al, 2014), and radioresponsiveness has an immune-mediated component in animal models of colon, breast and other solid cancers (Apetoh et al, 2007; Liang et al, 2013; Gerber et al, 2014). In breast cancer, the disappearance of Tregs following neoadjuvant CRT is associated with complete pathological response (Ladoire et al, 2008). However, the impact of Tregs in the local tumour microenvironment during standard long-course CRT for locally advanced rectal cancer remains unknown.
In the current study we evaluated the density of Foxp3+, CD3+ CD8+, CD4+ and IL-17+ cells in surgical resection samples from 128 patients with locally advanced rectal cancer treated with long-course 5-FU-based CRT. Our a priori hypotheses were that response to CRT has an immune-mediated component and that Tregs play an inhibitory role.
Materials and methods
Patients, treatment and follow-up
Patients who underwent neoadjuvant radiotherapy and surgery for rectal adenocarcinoma at our institution between August 2006 and December 2010, and who gave written informed consent for their biospecimens and health information to be used for research purposes, were identified from our prospectively maintained database. The consent rate for research participation at our institution is 98%. Clinical tumour and nodal stage (cT and cN), and the presence or absence of distant metastases (cM) was determined by computed tomography (CT) and magnetic resonance imaging (MRI) before commencement of CRT.
Neoadjuvant radiotherapy consisted of 50.4 Gy administered in 28 fractions over 5 weeks. Fluoropyrimidine-based chemotherapy was given concurrently via intravenous infusion 5-FU or oral capecitabine (Xeloda, Roche Products Pty Ltd, Dee Why, NSW, Australia). Surgical resection of the primary tumour was performed a minimum of 4 weeks after completing neoadjuvant treatment (6–8 weeks post CRT in most cases). Fluoropyrimidine-based adjuvant chemotherapy was offered to all patients (including those who attained a pCR), as per current Australian guidelines (ACN (Australian Cancer Network) Colorectal Cancer Guidelines Revision Committee, 2005), and was administered for up to 6 months postoperatively. Patients with distant metastases were progressively managed with neoadjuvant CRT, rectal surgery, potentially curative resection of metastatic lesions where possible and adjuvant chemotherapy.
Follow-up included 6-monthly visits, carcinoembryonic antigen (CEA) blood test and digital rectal examination. At 12-monthly intervals, patients had a repeat CT scan. A colonoscopy was performed at 12 months and then 4 years. Survival information was obtained from the Western Australian Cancer Registry every 6 months. Surviving patients were censored at the date of last survival update (1 May 2015). Cancer-specific survival (CSS) was defined as the time between the date of surgery and date of death from colorectal cancer. Recurrence-free survival (RFS) was defined as the time between the date of surgery and the date of first recurrence (local recurrence, development of first distant metastases or development of recurrent distant metastases after potentially curative resection) or date of death from colorectal cancer without prior documented recurrence (patients with unresected metastatic disease).
The study was approved by the St John of God Healthcare Human Research Ethics Committee and access to state cancer registry data was approved by the Department of Health Western Australia. The study was conducted in accordance with the Declaration of Helsinki.
Histopathologic assessment
Tumour regression grading was performed as part of routine pathological review using the Dworak system (Dworak et al, 1997), whereby grade 4 corresponds to pCR and grade 0 to no regression. Other variables obtained from the pathology report were pathologic T stage (ypT), pathologic N stage (ypN), resection margin status (R status), lymphovascular invasion (LVI), extramural venous invasion (EMVI) and perineural invasion (PNI).
Construction of tissue microarrays (TMAs)
Formalin-fixed, paraffin-embedded (FFPE) tumour samples and corresponding H&E slides were retrieved from the institutional pathology archive. A specialist gastrointestinal histopathologist (CH) reviewed the slides, verifying Dworak grade and selecting target areas of tumour and stroma. Tumour areas (Dworak 0–3 patients only) targeted central tumour, or residual malignant glands/tumour nests, depending on the degree of response. For the stromal cores, an area of nonepithelial tissue adjacent to the residual tumour was selected. In cases of pCR, areas within the fibrotic irradiated tumour bed, judged to most accurately represent the tumour stroma, were selected. Cylindrical tissue cores 1 mm in diameter were punched from the target area and inserted into a recipient paraffin block using an MTA-1 manual tissue arrayer (Beecher Instruments, Estigen, Tartu, Estonia). For each case, two tumour cores (where available), two stromal cores and one core of normal colonic epithelium (taken from a separate block of normal tissue) were included. Sections 4 μm in thickness were cut from the TMAs and mounted on positively charged slides for H&E staining and immunohistochemistry (IHC).
Immunohistochemistry
Staining for Foxp3, CD8 and IL-17 was performed manually. Manual staining protocols were carefully optimised with regard to antigen-retrieval, primary antibody concentration/incubation time and optimal detection system before staining the TMAs. Sections were dewaxed and rehydrated through graded xylene and alcohol washes. Antigen retrieval was performed in a DakoCytomation pressure cooker (Dako, Copenhagen, Denmark) at 121 °C for 6 min in Tris-EDTA buffer (pH 9.0). Peroxidase blocking solution (CINtec Histology Kit; Roche Australia, Castle Hill, NSW, Australia) was applied according to the manufacturer’s instructions. For Foxp3 and CD8 staining, sections were incubated with serum-free protein block (Dako) for 10 min, followed by the primary antibody (Foxp3 clone 236A/E7; Abcam, Melbourne, VIC, Australia, 1 : 100 dilution for 60 min or CD8 clone C8/144B; Dako, 1 : 100 dilution for 30 min). After washing in TBS-Tween, sections were incubated with a horseradish peroxidase (HRP)-conjugated secondary antibody for 30 min (CINtec Histology Kit), washed again in TBS-Tween and incubated with DAB solution (CINtec Histology Kit). The IL-17 staining was performed using a goat polyclonal anti-human IL-17 antibody (R&D Systems, Burlingame, MN, USA, 1 μg ml−1 diluted in blocking serum for 60 min) and an avidin/biotin system (Avidin/Biotin Blocking Kit, Vectastain Elite ABC kit and ImmPACT DAB; all from Vector Labs, Burlingame, CA, USA), according to the manufacturer’s instructions.
CD3 and CD4 staining was performed on an Autostainer Link 48 (Dako) using the EnVision FLEX+ high pH visualisation system (Dako). Rehydration and antigen-retrieval steps were performed in a PT Link pretreatment module (Dako). Primary antibodies used were a rabbit polyclonal anti-human CD3 and a mouse monoclonal anti-human CD4 clone 4B12 (both from Dako FLEX, ready-to-use).
Each TMA was stained on two separate occasions for each marker of interest. One staining run was selected as the primary run for analysis, based on overall staining quality. For cases where staining was judged to be inadequate (primarily because of detachment or folding of cores), the core from the alternative run was analysed where available.
For triple staining, an alkaline phosphatase (AP) and HRP multiplex detection system was used according to the manufacturer’s instructions (Biocare Medical, Concord, CA, USA). Following our standard antigen-retrieval process described above, sections were incubated for 15 min with a universal blocking reagent (Background Sniper), then for 30 min with a CD4/CD8 ready-to-use multiplex cocktail (Biocare Medical). Endogenous peroxidase was then blocked using Peroxidazed 1 and antibody detection was performed using the MACH 2 Doublestain 2 system with Betazoid DAB and Warp Red (Biocare Medical). Sections were then washed and incubated for 60 min with the primary Foxp3 antibody (clone 236A/E7; Abcam). Detection was performed using MACH 2 mouse HRP polymer and Vina Green. All triple stain reagents other than the Foxp3 antibody were purchased from Biocare Medical.
All sections were lightly counterstained in Mayers haematoxylin (Hurst Scientific, Perth, WA, Australia) before dehydration and mounting. Sections of human tonsil were included as positive and negative controls in every staining run. An isotype control was used in place of the primary antibody on negative control sections.
Assessment of T-cell density
Slides were scanned using a high-resolution digital scanner (Aperio Scanscope XT; Leica Biosystems, North Ryde, NSW, Australia) at × 40 magnification. Evaluation of T-cell density (cells per mm2 tissue) was performed using image analysis software (Aperio Imagescope version 11; Leica Biosystems) after careful manual annotation of the cores to ensure that the area of analysis matched the histology type to which the core was assigned, and to exclude areas of damaged or missing tissue. Data presented represent the average of two cores where possible.
Statistical analysis
Statistical analyses were performed using SAS version 9.3 (SAS Institute Inc., Cary, NC, USA) and GraphPad Prism version 6.0 (GraphPad software Inc., San Diego, CA, USA). Differences in T-cell density between response groups were assessed using general linear models. Associations between clinical/immunological variables and pCR were assessed using logistic regression. Time-to-event outcomes (CSS and RFS) were estimated using the Kaplan–Meier method with curves compared using the log-rank test. Hazard ratios (HRs) were determined using Cox proportional hazards regression (Cox, 1972). Multivariate analyses were performed using backwards stepwise selection of variables P<0.2 in univariate analysis. Differences and associations were considered statistically significant where P<0.05.
Results
Patient characteristics and treatment response
A total of 135 eligible patients were identified. Samples from 128 of these patients were included in the TMAs. Reasons for exclusion included concurrent malignancy (one patient), prior polypectomy of the primary tumour (one patient), minimal neoadjuvant treatment received (one patient) and insufficient or unavailable material (four patients). Characteristics of the 128 patients are shown in Table 1. Mean age at the time of surgery was 63 years and 72% were male. The majority of patients (81%) were clinical T stage 3 and 74% were assessed as having nodal involvement on pretreatment CT scan and MRI. Eleven patients (9%) had metastatic disease at diagnosis. This included lesions in the liver (eight patients), lung (one patient), liver and lung (one patient) and peritoneal disease (one patient). The median interval between the end of CRT and surgery was 7 weeks. One elderly patient (93 years of age) elected to have neoadjuvant long-course radiotherapy (RT) without concurrent chemotherapy. The most commonly performed surgical procedures performed were ultra-low anterior resection (67%) and abdominoperineal resection (27%). Other procedures included low anterior resection (three patients), Hartmann’s procedure (two patients) and total colectomy (two patients). Of the 11 patients with distant metastases, 5 underwent resection of liver/lung lesions and 1 patient with liver metastases received selective internal radiation therapy (SIRT).
In all, 26 patients (20%) achieved a pCR (Dworak grade 4) following neoadjuvant CRT (Table 1). In addition, 46 patients (36%) were assigned Dworak grade 3, 47 patients (37%) Dworak grade 2 and 9 patients (7%) Dworak grade 1. No patient was classed as Dworak grade 0.
T-cell subsets in the tumour microenvironment after CRT
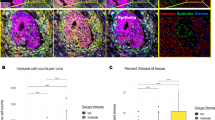
Foxp3+, CD3+, CD8+, CD4+ and IL-17+ cells were identified in the tumour, the stroma and the normal epithelium cores following CRT (Figure 1). The density of Foxp3+ cells was highest in the tumour cores, followed by the normal cores and then the stroma (median 125 vs 88 vs 28 cells per mm2 respectively; Figure 1B). We observed a similar distribution of CD8+ and CD4+ T cells, with IL-17+ cells most abundant in or around the normal epithelium (median 12 vs 11 vs 29 in tumour vs stroma vs normal cores; Figure 1B). To further characterise Foxp3+ cells, we performed concurrent staining for CD4, CD8 and Foxp3 using a multiplex IHC system. Manual scoring of a subset of 25 stromal cores, selected to include a range of Foxp3+ cell densities, demonstrated, as expected, that 99% of Foxp3+ cells with visible surface staining were CD4+ (Figure 1A, and data not shown).
Identification of T-cell subsets. (A) Representative immunohistochemical staining of stromal cores for Foxp3 (top left), CD3 (top centre), CD4 (top right), CD8 (bottom left), IL-17 (bottom centre) and multiplex detection of Foxp3 (green), CD4 (brown) and CD8 (pink) (bottom right). Arrows indicate CD4+Foxp3+ cells. Scale bar, 50 μm. (B) Frequencies of Foxp3+, CD4+, CD8+ and IL-17+ cells in tumour (Dworak 1–3 patients only), stroma and normal cores. Line at median. *P<0.05, **P<0.01, ***P<0.001.
Low stromal Foxp3+ T-cell density is associated with response to CRT
Analysis of T-cell subset density by Dworak grade revealed a strong inverse correlation between Foxp3+ cell density in the stroma and tumour regression (P=0.0006; Figure 2A). Of patients who had a pCR, 84% had a stromal Foxp3+ cell density lower than the median value as compared with 41% of noncomplete responders (OR: 7.56, P=0.0005; Figure 2B and Table 2). Stromal Foxp3+ cell density was strongly correlated with stromal CD3+ and CD4+ T-cell density, consistent with the co-expression of these molecules on Tregs (Supplementary Figure 1). A significant positive correlation was also observed between Foxp3+ and CD8+ cells in the stroma, the tumour and the normal cores. Foxp3+ and IL-17+ cell density positively correlated in the normal mucosa, but not in the tumour or stroma, where no significant correlation was seen (Supplementary Figure 1, and data not shown).
Low stromal Foxp3+ cell density is associated with response to CRT. (A) Stromal Foxp3+ cell density by Dworak grade (P=0.0006; general linear model). Circles represent outliers using Tukey’s method (> the 75th percentile plus 1.5 times the interquartile range). (B) Low vs high stromal Foxp3+ cell density (split at the median value) by pCR (P=0.0005; logistic regression). ***P<0.001.
Stromal Foxp3+ cell density was the only variable independently associated with pCR in multivariate analysis (OR: 5.27, P=0.0058; Table 2). Although low total T-cell and CD4+ T-cell densities were also associated with pCR in univariate analysis, these associations were no longer significant after adjustment for Foxp3+ T-cell density and presurgery clinical and demographic factors (Table 2). The ratio between Foxp3+ Tregs and other T-cell subsets was also investigated, but no correlation with CRT response was stronger than for stromal Foxp3+ cell density alone.
No association between tumour regression grade and the density of Foxp3+ Tregs, or other T-cell subsets, in the tumour (available for Dworak 1–3 patients only) or normal cores was found (Supplementary Figure 2A and B, and data not shown). However, there was no significant correlation between the density of Foxp3+ Tregs in the stroma and that in the tumour or normal cores (Supplementary Figure 2C and D).
Foxp3+ T-cell density as a prognostic marker in neoadjuvantly treated rectal cancer
A total of 104 patients (81%) were alive on the censor date and median follow-up was 76 months from the date of surgery. There were 4 deaths from causes other than colorectal cancer during the follow-up period (1 in each of Dworak 1, 2, 3 and 4 groups). Of these patients, 92 (72%) remained alive without disease recurrence. Only 3 patients (2.3%) had a local recurrence (2 in the Dworak grade 2 and 1 in the Dworak grade 3 groups), and in 1 case this was preceded by development of liver metastases. Median time to disease recurrence was 15.2 months (range 1.9–53 months). Distant recurrence events included development of new metastases (26 patients) and recurrence of previously resected metastatic disease (2 patients).
There were no deaths from rectal cancer in the Dworak 4 group (pCR). Dworak grade was significantly associated with both CSS and RFS (Figure 3A and B and Table 3). Stromal Foxp3+ cell density was also significantly associated with survival; the probability of 5-year CSS and RFS being 92% vs 81% and 80% vs 63% in the Foxp3 low vs high groups respectively (Figure 3C and D and Table 3). In multivariate analysis, adjusting for clinical and pathologic variables, cM status, Dworak grade and the presence or absence of PNI were significantly associated with RFS (Table 3). When the analysis was limited to patients without distant metastases at time of primary surgery, Dworak grade and resection margin status were the only independent prognostic factors (data not shown). No statistically significant interactions between variables included in the multivariate analyses were observed.
Discussion
This study investigated the relationship between Foxp3+ Treg density in the local tumour microenvironment following neoadjuvant CRT and treatment response in locally advanced rectal cancer. Given the increasing evidence for an immune-mediated component to chemotherapy and radiotherapy efficacy (Apetoh et al, 2007; Ladoire et al, 2008; Yasuda et al, 2011; Liang et al, 2013; Anitei et al, 2014; Gerber et al, 2014), and the well-established role of Tregs as suppressors of antitumour immunity, we hypothesised that Tregs inhibit the response of rectal carcinomas to neoadjuvant CRT.
We report a strong inverse correlation between stromal Foxp3+ cell density and tumour regression following CRT. Patients with a low stromal Foxp3+ cell density were over 5 times more likely to have a pCR, after adjustment for pretreatment clinical and demographic factors. Stromal Foxp3+ density was also significantly associated with improved RFS in univariate analysis. Although stromal Foxp3+ cell density was not independently prognostic in multivariate analysis, Dworak grade remained significantly associated with RFS, after adjustment for clinical and pathological variables, highlighting the clinical significance of response to CRT, even in the context of potentially curative resection.
In a recent study, Shinto et al (2014) found post-CRT stromal CD8+ T-cell density to be associated with improved RFS and CSS in a cohort of 93 rectal cancer patients who received short-course CRT (20 Gy given over 5 days with concurrent uracil over 7 days) and surgery 30 days later. They also assessed Foxp3+ cell density in resection samples, and found no significant association with survival. However, CD8/Foxp3 ratio in pre-CRT biopsies was predictive of improved tumour regression. An important difference between this and the current study is treatment schedule. Complete response is less common after short-course CRT (just 3% of the cohort studied by Shinto et al (2014) were classed as Dworak grade 4), and tumour regression can continue for up to 12 weeks post CRT (Glimelius, 2014). As such, although highlighting the potential importance of local antitumour immunity in rectal cancer response to CRT, these results may not be relevant to long-course CRT.
The negative association between the presence of Foxp3+ Tregs and clinical outcome observed in this study appears contrary to several studies in colorectal cancer, including our own previous work (Salama et al, 2009; Correale et al, 2010; Frey et al, 2010) that have demonstrated a positive association between Foxp3+ TILS and prognosis. However, the majority of samples assessed in these previous studies were from chemo/radiotherapy-naive colon cancer patients. Multiple preclinical studies have now demonstrated that radiotherapy efficacy requires an intact immune system and that tumour radioresponsiveness is associated with dendritic cell activation and CD8-driven antitumour immunity (Apetoh et al, 2007; Gupta et al, 2012; Liang et al, 2013; Gerber et al, 2014). Similar to many other chemotherapy drugs, 5-FU induces HMGB1 release from tumour cells, one of the major hallmarks of immunogenic cell death (Frey et al, 2012), and may preferentially deplete Tregs over other T-cell subsets (Maeda et al, 2011). If tumour regression following CRT in rectal cancer is, at least in part, due to CRT-induced antitumour immunity, it is rational that Tregs may play an antagonistic role.
The density of tumour-infiltrating T cells, and more specifically, CD8+ T cells, is a strong indicator of improved survival in colorectal cancer (Pages et al, 2009; Nosho et al, 2010; Mlecnik et al, 2011), and in rectal cancer biopsies, CD8 T-cell density predicts the likelihood of tumour regression following CRT (Yasuda et al, 2011; Anitei et al, 2014). Therefore, the association between low stromal CD3+ (and CD4+) cell density and pCR found in the current study seems counterintuitive. It is possible that a higher density of lymphocytes in the local tumour microenvironment after CRT in poor responders simply correlates with residual tumour burden. However, only Foxp3+ cell density was independently associated with treatment response in this study, indicating that the presence of Tregs is specifically important.
Given the strong association between tumour regression and stromal Foxp3+ Treg density observed, it is perhaps surprising that we found no association between tumour regression and Treg density in the tumour cores. Selection of truly representative target areas is a challenge common to all studies using TMAs, and can be particularly difficult when using tissue from pretreated patients with significant variation in treatment response. Although target sites were carefully selected by an experienced specialist gastrointestinal pathologist, we cannot exclude the possibility that the lack of correlation between Treg density in the stromal and tumour cores (for Dworak grade 1–3 patients) may be a function of target site selection. However, ours is not the first study to find T-cell subsets in the stromal compartment to be most strongly associated with outcome (Tougeron et al, 2013; Vayrynen et al, 2013; Mei et al, 2014), and importantly, the strongest association observed in the current study was between low stromal Treg density and pCR, the patient group for whom tumour cores were not available.
Our data support the hypothesis that response to CRT in rectal cancer is partially immune-mediated and inhibited by the presence of Tregs. It is possible that Tregs are selectively depleted in responding patients during CRT, providing a window of opportunity for the initiation of an effective antitumour immune response. Alternatively, fewer Tregs in the local tumour environment before treatment may allow for effective CRT-induced antitumour immunity. Which of these scenarios is most likely cannot be deduced from our data. We plan to answer this question by evaluating Treg density in pre-CRT biopsies in conjunction with subsequent surgical specimens.
If Tregs do limit tumour regression following CRT in rectal cancer, response rates may potentially be improved by combining current neoadjuvant regimens with an immune-modulating therapy. Although there is as yet no drug available that selectively depletes Tregs in humans effectively, ‘checkpoint blockade’, an approach that aims to stimulate an effective antitumour immune response through blocking inhibitory signals in T cells, has shown major promise in other solid cancers (Postow et al, 2015) and may be useful in this setting. It is of particular interest in the context of the current study that the anticytotoxic T lymphocyte-associated protein 4 (CTLA-4) monoclonal antibody, ipilimumab, exerts its immune-stimulating effect, not only through blocking inhibitory signals in effector T cells, but also through inhibiting Treg function (Peggs et al, 2009; Qureshi et al, 2011). However, given the important role played by Tregs in maintaining gut homeostasis (Barnes and Powrie, 2009), the timing and mode of delivery of any immune-modulating agent will likely be critical. Indeed, in a murine model of colitis-associated colon cancer (CAC), transient depletion of Tregs during the acute inflammation phase leads to uncontrolled inflammation and death, whereas depletion during the late phase results in enhanced CD8+ T cell-driven antitumour immunity and reduced tumour formation (Pastille et al, 2014). Clinical trials in this field will therefore require careful design.
In summary, we show that low stromal Foxp3+ Treg density post CRT is associated with pCR and improved long-term outcome in locally advanced rectal cancer. Our data suggest that Tregs in the local microenvironment may inhibit the response of rectal carcinomas to neoadjuvant CRT and may represent a therapeutic target in rectal cancer.
Change history
22 December 2015
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
ACN (Australian Cancer Network) Colorectal Cancer Guidelines Revision Committee (2005) Guidelines for the Prevention, Early Detection and Management of Colorectal Cancer. The Cancer Council Australia and Australian Cancer Network: Sydney.
Agarwal A, Chang GJ, Hu CY, Taggart M, Rashid A, Park IJ, You YN, Das P, Krishnan S, Crane CH, Rodriguez-Bigas M, Skibber J, Ellis L, Eng C, Kopetz S, Maru DM (2013) Quantified pathologic response assessed as residual tumor burden is a predictor of recurrence-free survival in patients with rectal cancer who undergo resection after neoadjuvant chemoradiotherapy. Cancer 119 (24): 4231–4241.
Anitei MG, Zeitoun G, Mlecnik B, Marliot F, Haicheur N, Todosi AM, Kirilovsky A, Lagorce C, Bindea G, Ferariu D, Danciu M, Bruneval P, Scripcariu V, Chevallier JM, Zinzindohoue F, Berger A, Galon J, Pages F (2014) Prognostic and predictive values of the immunoscore in patients with rectal cancer. Clin Cancer Res 20 (7): 1891–1899.
Apetoh L, Ghiringhelli F, Tesniere A, Obeid M, Ortiz C, Criollo A, Mignot G, Maiuri MC, Ullrich E, Saulnier P, Yang H, Amigorena S, Ryffel B, Barrat FJ, Saftig P, Levi F, Lidereau R, Nogues C, Mira JP, Chompret A, Joulin V, Clavel-Chapelon F, Bourhis J, Andre F, Delaloge S, Tursz T, Kroemer G, Zitvogel L (2007) Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat Med 13 (9): 1050–1059.
Barnes MJ, Powrie F (2009) Regulatory T cells reinforce intestinal homeostasis. Immunity 31 (3): 401–411.
Bosset JF, Collette L, Calais G, Mineur L, Maingon P, Radosevic-Jelic L, Daban A, Bardet E, Beny A, Ollier JC (2006) Chemotherapy with preoperative radiotherapy in rectal cancer. N Engl J Med 355 (11): 1114–1123.
Burnette BC, Liang H, Lee Y, Chlewicki L, Khodarev NN, Weichselbaum RR, Fu YX, Auh SL (2011) The efficacy of radiotherapy relies upon induction of type i interferon-dependent innate and adaptive immunity. Cancer Res 71 (7): 2488–2496.
Cho Y, Miyamoto M, Kato K, Fukunaga A, Shichinohe T, Kawarada Y, Hida Y, Oshikiri T, Kurokawa T, Suzuoki M, Nakakubo Y, Hiraoka K, Murakami S, Shinohara T, Itoh T, Okushiba S, Kondo S, Katoh H (2003) CD4+ and CD8+ T cells cooperate to improve prognosis of patients with esophageal squamous cell carcinoma. Cancer Res 63 (7): 1555–1559.
Correale P, Rotundo MS, Del Vecchio MT, Remondo C, Migali C, Ginanneschi C, Tsang KY, Licchetta A, Mannucci S, Loiacono L, Tassone P, Francini G, Tagliaferri P (2010) Regulatory (FoxP3+) T-cell tumor infiltration is a favorable prognostic factor in advanced colon cancer patients undergoing chemo or chemoimmunotherapy. J Immunother 33 (4): 435–441.
Cox DR (1972) Regression models and life-tables. J R Statist Soc B 34 (2): 187–220.
De Campos-Lobato LF, Stocchi L, da Luz Moreira A, Geisler D, Dietz DW, Lavery IC, Fazio VW, Kalady MF (2011) Pathologic complete response after neoadjuvant treatment for rectal cancer decreases distant recurrence and could eradicate local recurrence. Ann Surg Oncol 18 (6): 1590–1598.
Dworak O, Keilholz L, Hoffmann A (1997) Pathological features of rectal cancer after preoperative radiochemotherapy. Int J Colorectal Dis 12 (1): 19–23.
Fokas E, Liersch T, Fietkau R, Hohenberger W, Beissbarth T, Hess C, Becker H, Ghadimi M, Mrak K, Merkel S, Raab HR, Sauer R, Wittekind C, Rodel C (2014) Tumor regression grading after preoperative chemoradiotherapy for locally advanced rectal carcinoma revisited: updated results of the CAO/ARO/AIO-94 trial. J Clin Oncol 32 (15): 1554–1562.
Frey B, Stache C, Rubner Y, Werthmoller N, Schulz K, Sieber R, Semrau S, Rodel F, Fietkau R, Gaipl US (2012) Combined treatment of human colorectal tumor cell lines with chemotherapeutic agents and ionizing irradiation can in vitro induce tumor cell death forms with immunogenic potential. J Immunotoxicol 9 (3): 301–313.
Frey DM, Droeser RA, Viehl CT, Zlobec I, Lugli A, Zingg U, Oertli D, Kettelhack C, Terracciano L, Tornillo L (2010) High frequency of tumor-infiltrating FOXP3(+) regulatory T cells predicts improved survival in mismatch repair-proficient colorectal cancer patients. Int J Cancer 126 (11): 2635–2643.
Fridman WH, Pages F, Sautes-Fridman C, Galon J (2012) The immune contexture in human tumours: impact on clinical outcome. Nat Rev Cancer 12 (4): 298–306.
Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pages C, Tosolini M, Camus M, Berger A, Wind P, Zinzindohoue F, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Pages F (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313 (5795): 1960–1964.
Galon J, Pages F, Marincola FM, Angell HK, Thurin M, Lugli A, Zlobec I, Berger A, Bifulco C, Botti G, Tatangelo F, Britten CM, Kreiter S, Chouchane L, Delrio P, Arndt H, Asslaber M, Maio M, Masucci GV, Mihm M, Vidal-Vanaclocha F, Allison JP, Gnjatic S, Hakansson L, Huber C, Singh-Jasuja H, Ottensmeier C, Zwierzina H, Laghi L, Grizzi F, Ohashi PS, Shaw PA, Clarke BA, Wouters BG, Kawakami Y, Hazama S, Okuno K, Wang E, O'Donnell-Tormey J, Lagorce C, Pawelec G, Nishimura MI, Hawkins R, Lapointe R, Lundqvist A, Khleif SN, Ogino S, Gibbs P, Waring P, Sato N, Torigoe T, Itoh K, Patel PS, Shukla SN, Palmqvist R, Nagtegaal ID, Wang Y, D'Arrigo C, Kopetz S, Sinicrope FA, Trinchieri G, Gajewski TF, Ascierto PA, Fox BA (2012) Cancer classification using the Immunoscore: a worldwide task force. J Transl Med 10: 205.
Gerard JP, Conroy T, Bonnetain F, Bouche O, Chapet O, Closon-Dejardin MT, Untereiner M, Leduc B, Francois E, Maurel J, Seitz JF, Buecher B, Mackiewicz R, Ducreux M, Bedenne L (2006) Preoperative radiotherapy with or without concurrent fluorouracil and leucovorin in T3-4 rectal cancers: results of FFCD 9203. J Clin Oncol 24 (28): 4620–4625.
Gerber SA, Lim JY, Connolly KA, Sedlacek AL, Barlow ML, Murphy SP, Egilmez NK, Lord EM (2014) Radio-responsive tumors exhibit greater intratumoral immune activity than nonresponsive tumors. Int J Cancer 134 (10): 2383–2392.
Ghiringhelli F, Menard C, Puig PE, Ladoire S, Roux S, Martin F, Solary E, Le Cesne A, Zitvogel L, Chauffert B (2007) Metronomic cyclophosphamide regimen selectively depletes CD4+CD25+ regulatory T cells and restores T and NK effector functions in end stage cancer patients. Cancer Immunol Immunother 56 (5): 641–648.
Glimelius B (2014) Optimal time intervals between pre-operative radiotherapy or chemoradiotherapy and surgery in rectal cancer? Front Oncol 4: 50.
Gupta A, Probst HC, Vuong V, Landshammer A, Muth S, Yagita H, Schwendener R, Pruschy M, Knuth A, van den Broek M (2012) Radiotherapy promotes tumor-specific effector CD8+ T cells via dendritic cell activation. J Immunol 189 (2): 558–566.
Hiraoka K, Miyamoto M, Cho Y, Suzuoki M, Oshikiri T, Nakakubo Y, Itoh T, Ohbuchi T, Kondo S, Katoh H (2006) Concurrent infiltration by CD8+ T cells and CD4+ T cells is a favourable prognostic factor in non-small-cell lung carcinoma. Br J Cancer 94 (2): 275–280.
Huebner M, Wolff BG, Smyrk TC, Aakre J, Larson DW (2012) Partial pathologic response and nodal status as most significant prognostic factors for advanced rectal cancer treated with preoperative chemoradiotherapy. World J Surg 36 (3): 675–683.
Kim S, Ramakrishnan R, Lavilla-Alonso S, Chinnaiyan P, Rao N, Fowler E, Heine J, Gabrilovich DI (2014) Radiation-induced autophagy potentiates immunotherapy of cancer via up-regulation of mannose 6-phosphate receptor on tumor cells in mice. Cancer Immunol Immunother 63 (10): 1009–1021.
Ladoire S, Arnould L, Apetoh L, Coudert B, Martin F, Chauffert B, Fumoleau P, Ghiringhelli F (2008) Pathologic complete response to neoadjuvant chemotherapy of breast carcinoma is associated with the disappearance of tumor-infiltrating foxp3+ regulatory T cells. Clin Cancer Res 14 (8): 2413–2420.
Liang H, Deng L, Chmura S, Burnette B, Liadis N, Darga T, Beckett MA, Lingen MW, Witt M, Weichselbaum RR, Fu YX (2013) Radiation-induced equilibrium is a balance between tumor cell proliferation and T cell-mediated killing. J Immunol 190 (11): 5874–5881.
Liu J, Duan Y, Cheng X, Chen X, Xie W, Long H, Lin Z, Zhu B (2011) IL-17 is associated with poor prognosis and promotes angiogenesis via stimulating VEGF production of cancer cells in colorectal carcinoma. Biochem Biophys Res Commun 407 (2): 348–354.
Loi S, Sirtaine N, Piette F, Salgado R, Viale G, Van Eenoo F, Rouas G, Francis P, Crown JP, Hitre E, de Azambuja E, Quinaux E, Di Leo A, Michiels S, Piccart MJ, Sotiriou C (2013) Prognostic and predictive value of tumor-infiltrating lymphocytes in a phase III randomized adjuvant breast cancer trial in node-positive breast cancer comparing the addition of docetaxel to doxorubicin with doxorubicin-based chemotherapy: BIG 02-98. J Clin Oncol 31 (7): 860–867.
Maeda K, Hazama S, Tokuno K, Kan S, Maeda Y, Watanabe Y, Kamei R, Shindo Y, Maeda N, Yoshimura K, Yoshino S, Oka M (2011) Impact of chemotherapy for colorectal cancer on regulatory T-cells and tumor immunity. Anticancer Res 31 (12): 4569–4574.
Martin ST, Heneghan HM, Winter DC (2012) Systematic review and meta-analysis of outcomes following pathological complete response to neoadjuvant chemoradiotherapy for rectal cancer. Br J Surg 99 (7): 918–928.
Mei Z, Liu Y, Liu C, Cui A, Liang Z, Wang G, Peng H, Cui L, Li C (2014) Tumour-infiltrating inflammation and prognosis in colorectal cancer: systematic review and meta-analysis. Br J Cancer 110 (6): 1595–1605.
Michel S, Benner A, Tariverdian M, Wentzensen N, Hoefler P, Pommerencke T, Grabe N, von Knebel Doeberitz M, Kloor M (2008) High density of FOXP3-positive T cells infiltrating colorectal cancers with microsatellite instability. Br J Cancer 99 (11): 1867–1873.
Mlecnik B, Tosolini M, Kirilovsky A, Berger A, Bindea G, Meatchi T, Bruneval P, Trajanoski Z, Fridman WH, Pages F, Galon J (2011) Histopathologic-based prognostic factors of colorectal cancers are associated with the state of the local immune reaction. J Clin Oncol 29 (6): 610–618.
Nosho K, Baba Y, Tanaka N, Shima K, Hayashi M, Meyerhardt JA, Giovannucci E, Dranoff G, Fuchs CS, Ogino S (2010) Tumour-infiltrating T-cell subsets, molecular changes in colorectal cancer, and prognosis: cohort study and literature review. J Pathol 222 (4): 350–366.
Nowak AK, Lake RA, Marzo AL, Scott B, Heath WR, Collins EJ, Frelinger JA, Robinson BW (2003) Induction of tumor cell apoptosis in vivo increases tumor antigen cross-presentation, cross-priming rather than cross-tolerizing host tumor-specific CD8 T cells. J Immunol 170 (10): 4905–4913.
Pages F, Berger A, Camus M, Sanchez-Cabo F, Costes A, Molidor R, Mlecnik B, Kirilovsky A, Nilsson M, Damotte D, Meatchi T, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Galon J (2005) Effector memory T cells, early metastasis, and survival in colorectal cancer. N Engl J Med 353 (25): 2654–2666.
Pages F, Kirilovsky A, Mlecnik B, Asslaber M, Tosolini M, Bindea G, Lagorce C, Wind P, Marliot F, Bruneval P, Zatloukal K, Trajanoski Z, Berger A, Fridman WH, Galon J (2009) In situ cytotoxic and memory T cells predict outcome in patients with early-stage colorectal cancer. J Clin Oncol 27 (35): 5944–5951.
Pastille E, Bardini K, Fleissner D, Adamczyk A, Frede A, Wadwa M, von Smolinski D, Kasper S, Sparwasser T, Gruber AD, Schuler M, Sakaguchi S, Roers A, Muller W, Hansen W, Buer J, Westendorf AM (2014) Transient ablation of regulatory T cells improves antitumor immunity in colitis-associated colon cancer. Cancer Res 74 (16): 4258–4269.
Peggs KS, Quezada SA, Chambers CA, Korman AJ, Allison JP (2009) Blockade of CTLA-4 on both effector and regulatory T cell compartments contributes to the antitumor activity of anti-CTLA-4 antibodies. J Exp Med 206 (8): 1717–1725.
Postow MA, Callahan MK, Wolchok JD (2015) Immune checkpoint blockade in cancer therapy. J Clin Oncol 33 (17): 1974–1982.
Qureshi OS, Zheng Y, Nakamura K, Attridge K, Manzotti C, Schmidt EM, Baker J, Jeffery LE, Kaur S, Briggs Z, Hou TZ, Futter CE, Anderson G, Walker LS, Sansom DM (2011) Trans-endocytosis of CD80 and CD86: a molecular basis for the cell-extrinsic function of CTLA-4. Science 332 (6029): 600–603.
Ramakrishnan R, Assudani D, Nagaraj S, Hunter T, Cho HI, Antonia S, Altiok S, Celis E, Gabrilovich DI (2010) Chemotherapy enhances tumor cell susceptibility to CTL-mediated killing during cancer immunotherapy in mice. J Clin Invest 120 (4): 1111–1124.
Salama P, Phillips M, Grieu F, Morris M, Zeps N, Joseph D, Platell C, Iacopetta B (2009) Tumor-infiltrating FOXP3+ T regulatory cells show strong prognostic significance in colorectal cancer. J Clin Oncol 27 (2): 186–192.
Sauer R, Liersch T, Merkel S, Fietkau R, Hohenberger W, Hess C, Becker H, Raab HR, Villanueva MT, Witzigmann H, Wittekind C, Beissbarth T, Rodel C (2012) Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. J Clin Oncol 30 (16): 1926–1933.
Shinto E, Hase K, Hashiguchi Y, Sekizawa A, Ueno H, Shikina A, Kajiwara Y, Kobayashi H, Ishiguro M, Yamamoto J (2014) CD8+ and FOXP3+ tumor-infiltrating T cells before and after chemoradiotherapy for rectal cancer. Ann Surg Oncol 21 (Suppl 3): S414–S421.
Tosolini M, Kirilovsky A, Mlecnik B, Fredriksen T, Mauger S, Bindea G, Berger A, Bruneval P, Fridman WH, Pages F, Galon J (2011) Clinical impact of different classes of infiltrating T cytotoxic and helper cells (Th1, th2, treg, th17) in patients with colorectal cancer. Cancer Res 71 (4): 1263–1271.
Tougeron D, Maby P, Elie N, Fauquembergue E, Le Pessot F, Cornic M, Sabourin JC, Michel P, Frebourg T, Latouche JB (2013) Regulatory T lymphocytes are associated with less aggressive histologic features in microsatellite-unstable colorectal cancers. PLoS One 8 (4): e61001.
Vayrynen JP, Tuomisto A, Klintrup K, Makela J, Karttunen TJ, Makinen MJ (2013) Detailed analysis of inflammatory cell infiltration in colorectal cancer. Br J Cancer 109 (7): 1839–1847.
West NR, Kost SE, Martin SD, Milne K, Deleeuw RJ, Nelson BH, Watson PH (2013) Tumour-infiltrating FOXP3(+) lymphocytes are associated with cytotoxic immune responses and good clinical outcome in oestrogen receptor-negative breast cancer. Br J Cancer 108 (1): 155–162.
Yasuda K, Nirei T, Sunami E, Nagawa H, Kitayama J (2011) Density of CD4(+) and CD8(+) T lymphocytes in biopsy samples can be a predictor of pathological response to chemoradiotherapy (CRT) for rectal cancer. Radiat Oncol 6: 49.
Yoon HH, Orrock JM, Foster NR, Sargent DJ, Smyrk TC, Sinicrope FA (2012) Prognostic impact of FoxP3+ regulatory T cells in relation to CD8+ T lymphocyte density in human colon carcinomas. PLoS One 7 (8): e42274.
Zhang B, Bowerman NA, Salama JK, Schmidt H, Spiotto MT, Schietinger A, Yu P, Fu YX, Weichselbaum RR, Rowley DA, Kranz DM, Schreiber H (2007) Induced sensitization of tumor stroma leads to eradication of established cancer by T cells. J Exp Med 204 (1): 49–55.
Zhang L, Conejo-Garcia JR, Katsaros D, Gimotty PA, Massobrio M, Regnani G, Makrigiannakis A, Gray H, Schlienger K, Liebman MN, Rubin SC, Coukos G (2003) Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N Engl J Med 348 (3): 203–213.
Zorcolo L, Rosman AS, Restivo A, Pisano M, Nigri GR, Fancellu A, Melis M (2012) Complete pathologic response after combined modality treatment for rectal cancer and long-term survival: a meta-analysis. Ann Surg Oncol 19 (9): 2822–2832.
Zou W (2006) Regulatory T cells, tumour immunity and immunotherapy. Nat Rev Immunol 6 (4): 295–307.
Acknowledgements
This study was funded by the Cancer Council Western Australia (ID 1048279), the Raine Medical Research Foundation, the St John of God Foundation, the Colorectal Surgical Society of Australia and New Zealand (CSSANZ), the Tonkinson Foundation for Colorectal Cancer Research and the McCusker Charitable Foundation. We thank Dr Tim Threlfall of the Western Australian Cancer Registry and Cheryl Penter of St John of God Subiaco Hospital for assistance with the data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Supplementary Information accompanies this paper on British Journal of Cancer website
Supplementary information
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
McCoy, M., Hemmings, C., Miller, T. et al. Low stromal Foxp3+ regulatory T-cell density is associated with complete response to neoadjuvant chemoradiotherapy in rectal cancer. Br J Cancer 113, 1677–1686 (2015). https://doi.org/10.1038/bjc.2015.427
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2015.427
Keywords
This article is cited by
-
Wnt/β-catenin signalling activates IMPDH2-mediated purine metabolism to facilitate oxaliplatin resistance by inhibiting caspase-dependent apoptosis in colorectal cancer
Journal of Translational Medicine (2024)
-
Use of patient-derived explants as a preclinical model for precision medicine in colorectal cancer: A scoping review
Langenbeck's Archives of Surgery (2023)
-
Relationship between stromal regulatory T cells and the response to neoadjuvant chemotherapy for locally advanced rectal cancer
Surgery Today (2022)
-
Biomarkers and cell-based models to predict the outcome of neoadjuvant therapy for rectal cancer patients
Biomarker Research (2021)
-
Tumor Microenvironment Mediators CD8+- and FOXP3+-Labeled T Lymphocytes Are Prospective Prognosticators in Curatively Treated Rectal Cancer Patients
Journal of Gastrointestinal Cancer (2021)