Abstract
Background:
Three prospective studies have evaluated the association between dietary acrylamide intake and endometrial cancer (EC) risk with inconsistent results. The objective of this study was to evaluate the association between acrylamide intake and EC risk: for overall EC, for type-I EC, and in never smokers and never users of oral contraceptives (OCs). Smoking is a source of acrylamide, and OC use is a protective factor for EC risk.
Methods:
Cox regression was used to estimate hazard ratios (HRs) for the association between acrylamide intake and EC risk in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort. Acrylamide intake was estimated from the EU acrylamide monitoring database, which was matched with EPIC questionnaire-based food consumption data. Acrylamide intake was energy adjusted using the residual method.
Results:
No associations were observed between acrylamide intake and overall EC (n=1382) or type-I EC risk (n=627). We observed increasing relative risks for type-I EC with increasing acrylamide intake among women who both never smoked and were non-users of OCs (HRQ5vsQ1: 1.97, 95% CI: 1.08–3.62; likelihood ratio test (LRT) P-value: 0.01, n=203).
Conclusions:
Dietary intake of acrylamide was not associated with overall or type-I EC risk; however, positive associations with type I were observed in women who were both non-users of OCs and never smokers.
Similar content being viewed by others
Main
Acrylamide is a known neurotoxin in humans, and a carcinogen in animals (Friedman, 2003; LoPachin and Gavin, 2008; Hogervorst et al, 2010). In 1994, based on animals studies, as well as evidence found in humans, the International Agency for Research on Cancer (IARC) classified acrylamide as ‘probably carcinogenic’ to humans (IARC group 2A; IARC, 1994). In 2002, Swedish researchers discovered acrylamide in some heat-treated carbohydrate-rich foods (Tareke et al, 2002), and further research concluded that acrylamide is formed during common cooking procedures (predominantly through the Maillard reaction), such as frying, grilling, and baking (Friedman, 2003). In the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort, the main determinants of estimated dietary intake of acrylamide based on 24-h dietary recall (DR) were bread, crisp bread, rusks, coffee, fried potatoes, cakes, biscuits, and cookies (Freisling et al, 2013). Acrylamide is also a component of cigarette smoke, thus, smoking is an important source of exposure (Boettcher et al, 2005; Vesper et al, 2008).
Acrylamide is metabolised via the Cyp2e1 enzyme system to glycidamide, a chemically reactive epoxide and mutagen in animals (Doroshyenko et al, 2009; Hogervorst et al, 2010). After acrylamide administration, hormone-related (including uterine tumours) and other tumours (e.g., oral tissues) have been observed in rats (Johnson et al, 1986).
Endometrial cancer (EC) is the fourth most common cancer diagnosed in European women, but mortality is relatively low with a 5-year survival rate varying from 65 to 85% (Cook et al, 2006; Ferlay et al, 2013). There is considerable international variation in incidence as well as mortality, and both rates increase dramatically with age (Cook et al, 2006; Ferlay et al, 2013; Jamison et al, 2013). Established risks factors for EC are obesity, low physical activity, history of polycystic ovary syndrome, and greater lifetime exposure to estrogens (Kaaks et al, 2002; Cook et al, 2006). The use of oral contraceptives (OCs, containing both oestrogen and progestin in the formula) is well established to lower the risk of developing EC (Cook et al, 2006; Gierisch et al, 2013). There is evidence that tobacco smoking also reduces the risk of EC (Terry et al, 2004; Cook et al, 2006); however, an EPIC study reported an increased risk of EC in premenopausal women who smoked (Al-Zoughool et al, 2007). Endometrial cancer is generally classified into two types: type-I EC are mostly endometrioid adenocarcinomas and are associated with unopposed oestrogen exposure; and type-II EC tumours are mainly serous carcinomas, are believed to be oestrogen independent, and have poor prognosis (Amant et al, 2005; Setiawan et al, 2013).
Three prospective epidemiological studies have assessed the relationship between dietary intake of acrylamide and EC risk. The Netherlands Cohort Study (NLCS) observed a positive association between acrylamide intake and EC risk, especially in never smokers (Hogervorst et al, 2007). Likewise, the Nurses’ Health Study (NHS) reported an increased relative risk among women with the highest acrylamide intake (Wilson et al, 2010); however, no associations between acrylamide intake and EC were observed in the Swedish Mammography Cohort (SMC; Larsson et al, 2009).
The present study evaluated the association between questionnaire-based dietary intake of acrylamide and the risk of overall EC (type I, type II, and undefined) and type-I EC tumours, using data from 301 113 EPIC cohort participants. Subgroup analyses among never-smoking women and never users of OCs were performed with the aim to eliminate the influence of smoking (both a source of acrylamide and a protective factor) and the protective effect of OCs on EC risk.
Methods
Study population
The EPIC study was initiated between 1992 and 1998 in 23 centres from 10 European countries with the aim to investigate the relationships between nutrition and lifestyle factors, and cancer and other chronic diseases. All participants gave written informed consent. Ethical review boards from the IARC and local centres participating in EPIC approved the study. The EPIC methodology has been reported in detail by Riboli et al (2002).
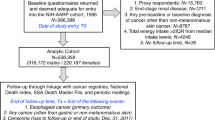
The EPIC study includes 521 330 participants, of which 367 903 are women. A total of 66 790 women were excluded from the current analyses because they were diagnosed with cancer before recruitment (n=19 853), had a hysterectomy (n=35 116), had incomplete follow-up data (n=2896), had no lifestyle or dietary information (n=2877), and no information on dietary intake of acrylamide at baseline (n=3), or had an extreme ratio of energy intake to energy required (n=6045); resulting in 301 113 participants for this analysis.
Identification of endometrial cancer cases
Information on cancer incidence was obtained through population cancer registries, or via a combination of methods: health insurance records, cancer and pathology registries, and active follow-up (France, Germany, Naples, and Greece). Subjects were followed until cancer diagnosis (except non-melanoma skin cancer), emigration, death, or until the end of follow-up (dates varied between centres, from December 2004 to June 2010).
Tumour morphology was specified for 664 (48%) cases, of which 627 (93%) were classified as type I (endometrioid adenocarcinomas), and 37 (7%) as type II (serous, or clear cell, or squamous adenocarcinomas; Tavassoli and Devilee, 2003). Overall EC comprises type I, type II, and cases that were undefined for histology. Tumours were classified as C54 according to the International Classification of Diseases, 10th revision.
Dietary and acrylamide intake assessment
Information on diet was assessed at baseline (with timeframe referring to the previous 12 months) through country-specific, validated dietary questionnaires (DQ; Riboli et al, 2002). The development of the acrylamide database in EPIC has been previously described (Freisling et al, 2013; Obon-Santacana et al, 2013). To summarise, the EPIC acrylamide database is a compilation of the information acquired to a large extent from the European Community Institute for Reference Materials and Measurements (IRMM). The average acrylamide levels for specific foods in the IRMM database were obtained through a combination of methods based on either liquid or gas chromatography coupled to mass spectrometry. All food items with acrylamide data derived from the IRMM database were classified according to EPIC-Soft food classification (Voss et al, 1998; Slimani et al, 2000). The reported foods on the DQ and, when available, their relevant description (e.g., baked potatoes) were matched with the corresponding foods in the acrylamide database. Information on cooking methods for acrylamide sources was available for potatoes (except in Italy), bread, and breaded meats. If an exact match was not possible, the food was linked to the mean of all foods of the respective food group in the acrylamide database (Freisling et al, 2013; Obon-Santacana et al, 2013).
Lifestyle and reproductive information assessment
At baseline, questionnaires were used to collect data on tobacco smoking, education, physical activity, and menstrual and reproductive factors (i.e., age at first menstrual period, ever use of OCs, ever use of hormone replacement therapy (HRT)). Baseline menopausal status was self-reported for each woman in most centres, and in case of incomplete data, an algorithm was developed based on the age at recruitment: women were classified as premenopausal if their baseline ages were <46 years, or reported having menstrual cycles the year before recruitment; perimenopausal if their ages were between 46 and 55 years, or had irregular menses the year before recruitment; and postmenopausal if their ages were >56 years, or had bilateral ovariectomy (surgical menopause), or had <4 menstrual cycles in the past year before recruitment (Riboli et al, 2002).
Height, weight, and waist or hip circumference were measured at baseline by trained personnel for all EPIC participants, except for most participants in France, Norway and Oxford cohorts, where height and weight were self-reported. Umeå and Norway did not record data on waist or hip circumference, and only some participants from France have information on waist (29%) and hip circumference (29%; Riboli et al, 2002).
Statistical analysis
Proportional hazards models (Cox regression) were used to estimate hazards ratio (HR) and 95% confidence intervals (95% CI) for overall EC risk in relation to dietary intake of acrylamide. Analyses were also performed separately for risk of type-I EC. Analyses for type-II EC cases were not carried out due to small sample sizes (n=37). All multivariate models had age as the time scale and were stratified by study centre to control for centre effects (i.e., questionnaire design and follow-up procedures), and by age at recruitment (in 1-year categories) as the primary time variable.
All estimates of acrylamide intake in these analyses were energy adjusted using the residual method (Willett, 1998; Ferrari et al, 2013). One continuous variable and one categorical variable for dietary intake of acrylamide were evaluated in Cox models: average daily intake in 10 μg increments (10 μg per day), and quintiles of intake (μg per day) based on the distribution in the full EPIC cohort of women.
The following variables were included as known risk factors or potential confounders in these analyses: body mass index (BMI, kg m−2), smoking status (never smokers, current pipe or cigar or occasional smokers, current cigarette smokers: 1–15, 16–25, or ⩾26 cigarettes per day, former cigarette smokers who quit >20 years, 11–20 years, or ⩽10 years before recruitment), history of diabetes (no, yes), OC use (never, ever), HRT use (never, ever), baseline menopause status combined with age at menopause (premenopausal, perimenopausal, postmenopausal with: <45, 45–49, 50–52, 53–55, and ⩾56 years, surgical menopause, postmenopausal women with missing age at menopause), parity (nulliparous, 1, 2, ⩾3, parous but with missing number of full-term pregnancies), and age at menarche (<12, 12, 13, 14, and ⩾15 years). Variables for education level (none, primary, technical/professional, secondary, and higher education), physical activity using the Cambridge index (Wareham et al, 2003), alcohol intake (non-drinkers, drinkers of 0–6, >6–12, >12–24, and >24 g per day), total fat (g per day), total fibre (g per day), vegetables (g per day), and fruits, nuts and seeds consumption (g per day) were evaluated, but were not included in final models because they did not change effect estimates >10%. Missing values for specific variables were categorised as ‘unknown’ and were included in the analyses. All statistical models presented in this study were further adjusted for total energy intake (per 1000 kcal per day).
Analyses of effect-measure modification were carried out by known EC risk factors (BMI, menopausal status, and HRT use), by known protective factors (OC use, and smoking status), by geographical region, and by factors that may affect the activity of Cyp2e1 (alcohol intake, and BMI; Wilson et al, 2009; Freisling et al, 2013). The following subgroups were examined: BMI (<25 kg m−2, ⩾25 kg m−2), OC use (never, ever), HRT use (never, ever), baseline menopausal status (premenopausal, perimenopausal, and postmenopausal), smoking status (never, current, or former smokers), and alcohol intake (never, ever drinkers). For region-specific analyses, countries were classified as northern (France, UK, The Netherlands, Germany, Sweden, Denmark, and Norway) and southern (Italy, Spain, and Greece); and by median acrylamide-intake level (‘high’ ⩾21 μg per day and ‘low’ <21 μg per day) in the EPIC cohort.
Sensitivity analyses were additionally performed excluding all cases diagnosed during the first 2 years of follow-up, with the aim to avoid possible influences of preclinical disease on dietary habits including intakes of acrylamide.
To evaluate dose-response trends, the median value for each acrylamide quintile was estimated and included in a score test. Statistical significance of effect-measure modification was evaluated using a LRT and based on the continuous acrylamide intake variable. The proportional hazards (PHs) assumption was tested in STATA (College Station, Texas, USA) using Schoenfeld residuals (Schoenfeld, 1982), and it was met for type-I EC analyses; however, it was violated for overall EC analyses. Variables responsible for the PH violation were: OC use, HRT use, and smoking status; thus, stratified analyses by these variables were also performed for overall EC risk, and the PH assumption was subsequently met. All analyses were performed using SAS v. 9.1 (Cary, NC, USA).
Results
Basic information on cohorts members
The average acrylamide intake in the EPIC subcohort of women was 24±13 μg per day (0.4±0.2 μg per kg body weight per day), and the 10th–90th percentile range was 10–41 μg per day (0.2–0.6 μg per kg body weight per day). Denmark, followed by the UK and The Netherlands, had the highest mean and median dietary acrylamide intakes, while Italy had the lowest acrylamide intake (Table 1). In total, after 11 years of follow-up there were 1382 first primary EC cases, of which 627 were classified as type-I EC, 37 type-II EC, and 718 cases that were not specified with regard to histology (Table 1).
Women with the highest acrylamide-intake levels tended to have the highest intakes of energy, total fats, total carbohydrates, vegetables, and coffee. Women with the highest intake levels tended to be premenopausal, have a higher proportion of OC use and with longer duration, and were more often current smokers or former smokers at baseline (Table 2). In contrast, women classified in the lower quintiles tended to be postmenopausal, non-consumers of alcohol and tobacco, and to have lower levels of physical activity (Table 2). There were few differences across acrylamide intake quintiles by age, age at first menstrual period, age at menopause, BMI, or waist-to-hip ratio (Table 2).
Overall EC risk and type-I EC risk
No association was observed between acrylamide intake and overall EC (Table 3) or type-I EC risk (Table 4). Similar results were found when we restricted the analyses to cases diagnosed 2 years after recruitment (Tables 3 and 4), or when known type-I and type-II EC were combined in the same analysis (data not shown). Further, an analysis among EC cases that could not be classified into type-I or type-II EC was also carried out, but no associations were observed (data not shown). Most of the stratified analyses performed with overall EC (type I, type II, and undefined) cases indicated no heterogeneity between subgroups (Table 3). When stratified analyses by OC use, and by OC use and smoking were performed, statistically significant LRT P-values were observed; however, neither the continuous nor the categorical acrylamide variable suggested an association with disease risk (Table 3).
Effect-measure modification by OC use and smoking in type-I EC
Subgroup analyses for known type-I EC were also stratified by smoking status, OC use, menopausal status, HRT use, BMI, and geographical region. None of the HRs in never smokers or ever smokers indicated associations between dietary acrylamide intake and type-I EC risk; however, statistically significant evidence for heterogeneity was observed (LRT P-value: 0.01; Table 4).
Inverse associations were observed for the highest versus the lowest quintile of acrylamide intake (HRQ5vsQ1: 0.57, 95% CI: 0.34–0.96; P-value for trend: 0.01), as well as a continuous variable (HR: 0.83, 95% CI: 0.71–0.95; Table 4). Regarding the HRs obtained in the subgroup of non-OC users, none of them were statistically significant (HR10 μg per day: 1.10, 95% CI: 0.99–1.23; Table 4).
Moreover, the OC-use model was additionally adjusted by duration of OC use (per 2 years of OC use), and the results were similar to those presented without adjustment for this variable (data not shown).
There were some differences in non-dietary variables between OC users and non-users. OC users with the highest acrylamide intake tended to have a higher proportion of former or current smokers, and these women tended to smoke more cigarettes per day than non-users. Further, non-OC users were older than OC users, but with similar age at menopause. With regard to dietary factors, there were no major differences between OC users and non-users (data not shown).
The association between acrylamide intake and type-I EC risk among OC users and non-users was also evaluated by smoking status. Women who at baseline reported being never smokers and non-users of OCs (including 203 type-I EC cases) were at the highest risk of developing type-I EC, when acrylamide was evaluated both as a continuous variable and in quintiles (HR10 μg per day: 1.17, 95% CI: 1.02–1.34; HRQ5vsQ1: 1.97, 95% CI: 1.08–3.62; P-value for trend: 0.01; Table 4). Otherwise, associations between dietary acrylamide intake and type-I EC were below the null value in ever smokers (current and former smokers) and OC users (HR10 μg per day: 0.75, 95% CI: 0.60–0.94; Table 4). The LRT P-value of the contrast between ‘never smokers/non-OC users’, ‘ever smokers/non-OC users’, ‘never smokers/OC users’, and ‘ever smokers/OC users’ for the continuous acrylamide intake variable was 0.01 (Table 4).
Other effect-measure modifications in type-I EC
There was no evidence for effect-measure modification by BMI (Table 4), HRT use, or by geographical region (all LRT P-values >0.12, data not shown); however, evidence for effect-measure modification was found when the analyses were stratified by baseline menopausal status (LRT P-value: 0.01; Table 4), but none of the individual HRs were statistically significant. Likewise, effect-measure modification was observed by alcohol intake (LRT P-value: 0.01), but only the continuous variable in never drinkers showed a statistically significant positive association (HR10 μg per day: 1.23, 95% CI: 1.02–1.47; Table 4).
Discussion
No overall association was observed between dietary intake of acrylamide and overall EC or type-I EC risk; nevertheless, elevated relative risks, as well as P-values for linear trend were observed for the association between dietary intake of acrylamide and type-I EC among women who both never smoked and never used OCs. Statistically significant inverse associations between type-I EC risk and acrylamide intake were observed in OC users, and among OC users and ever smokers.
It is widely published that use of OCs (containing oestrogen and progestin) is protective against EC risk, and this effect is maintained for years (Amant et al, 2005; Cook et al, 2006; Cibula et al, 2010; Gierisch et al, 2013). Likewise, cigarette smoking tends to lower the risk of developing EC, and it is thought to be more pronounced in recent smokers (Cook et al, 2006). All the relative risk estimates for type-I EC risk observed among OC users and ever smokers were below the null value; however, because OC use, duration of OC use, and smoking are associated with higher acrylamide intake in EPIC, and are also associated with lower EC risk, residual confounding by these variables may play a role in the observed inverse associations (in OC users and smokers). In addition, OC users, compared to non-OC users, tended to smoke more cigarettes per day and reported less time since having quit smoking. Thus, these baseline characteristics may have partially influenced the results obtained in this subgroup of women. Moreover, it has been hypothesised that acrylamide may have hormonal effects, and the results in non-OC users for type I are potentially compatible with this hypothesis, since type-I EC is considered to be oestrogen driven (Amant et al, 2005); nevertheless, this hypothesis has not been substantiated, and other mechanisms (i.e., genotoxicity caused by glycidamide) may be compatible with the results (Hogervorst et al, 2010, 2013).
The relation between dietary intake of acrylamide and EC risk has been previously published in three prospective cohort studies. Both the NLCS and NHS studies found statistically significantly increased relative risks: the NLCS among never-smoking women, and the NHS in the entire cohort (Hogervorst et al, 2007; Wilson et al, 2010). Although the NLCS and NHS studies did not evaluate the association between acrylamide intake and type-I EC specifically, about 80% of EC cases are thought to be type-I endometrioid tumours (Amant et al, 2005); thus, the majority of the cases in the previous publications were likely type-I EC cases. Only the SMC study observed no associations between acrylamide intake and EC risk (Larsson et al, 2009), and this could be due to the smaller baseline ranges of acrylamide intake in that study. The median acrylamide intake for the reference group in the SMC was 16.9 μg per day, and for the highest intake category was 32.5 μg per day, whereas in EPIC, the median for the reference group was 9.3 μg per day, and for the highest intake category was 44.0 μg per day. All three previous studies presented statistical models adjusted for OC use, but none reported analyses stratified by OC use.
Some evidence for an inverse association between the highest and lowest acrylamide quintiles and type-I EC risk was observed among women with a BMI <25 kg m−2; however, neither the continuous variable for acrylamide intake (per 10 μg per day) nor the LRT P-value were statistically significant. A suggestive increased risk for type-I EC was observed in women who reported never drinking alcohol at baseline when the continuous acrylamide variable was evaluated; nevertheless, this result was based on 103 type-I EC cases. Further, suggestive evidence for heterogeneity of the association between dietary acrylamide intake and type-I EC risk was also indicated by smoking status, and by menopausal status at baseline; nevertheless no dose-response trend was observed.
The strengths of our study are that EPIC is one of the largest prospective cohort studies on diet and cancer, and recall bias is unlikely because exposure and diet information were collected years before cancer diagnoses. The present study had more cases than the other three previously published studies (n=1382), and this allowed us to evaluate known type-I EC separately (n=627). The SMC study analysed 687 EC cases (Larsson et al, 2009), the NHS study analysed 484 EC cases (Wilson et al, 2010), and the NLCS study evaluated 221 (Hogervorst et al, 2007).
The present study had the following limitations: some food preparation techniques (e.g., cooking method) that could have contributed to the variability of total acrylamide intake were not assessed in all EPIC centres. In addition, the correlation coefficient between a single 24-h DR in EPIC, and acrylamide intake derived from food intake questionnaires was low: 0.17 (Ferrari et al, 2013). This could indicate that a single 24-h DR may not be enough to accurately estimate the average acrylamide intake. Further, the EPIC acrylamide estimates might have been influenced by measurement error; however, all the analyses were adjusted for energy intake since in EPIC and in other populations, it has been observed that the validity of acrylamide estimates improved after energy intake adjustment (Ferrari et al, 2013). Another limitation of our study is that 718 EC cases were not classified in any of the EC subtypes; however, as has been previously mentioned, a large proportion (≈80%) of endometrial carcinomas are thought to be type I (Amant et al, 2005). Finally, it should to be kept in mind that several subgroups have been examined in this study; thus, some of the observed results might be due to chance.
In conclusion, the results of the present study indicate that there were no associations between dietary intake of acrylamide and risk of overall EC or type-I EC; nevertheless, women with elevated acrylamide intake (upper quintile median, 44 μg per day) who both never smoked and never used OCs at baseline, were at higher risk of developing type-I EC relative to women with the lowest intakes. Additional studies with biomarkers of internal dose of acrylamide exposure are needed in order to better understand the associations observed.
Change history
26 August 2014
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Al-Zoughool M, Dossus L, Kaaks R, Clavel-Chapelon F, Tjonneland A, Olsen A, Overvad K, Boutron-Ruault MC, Gauthier E, Linseisen J, Chang-Claude J, Boeing H, Schulz M, Trichopoulou A, Chryssa T, Trichopoulos D, Berrino F, Palli D, Mattiello A, Tumino R, Sacerdote C, Bueno-de-Mesquita HB, Boshuizen HC, Peeters PH, Gram IT, Braaten T, Lund E, Chirlaque MD, Ardanaz E, Agudo A, Larranaga N, Quiros JR, Berglund G, Manjer J, Lundin E, Hallmans G, Khaw KT, Bingham S, Allen N, Key T, Jenab M, Cust AE, Rinaldi S, Riboli E (2007) Risk of endometrial cancer in relationship to cigarette smoking: results from the EPIC study. Int J Cancer 121 (12): 2741–2747.
Amant F, Moerman P, Neven P, Timmerman D, Van LE, Vergote I (2005) Endometrial cancer. Lancet 366 (9484): 491–505.
Boettcher MI, Schettgen T, Kutting B, Pischetsrieder M, Angerer J (2005) Mercapturic acids of acrylamide and glycidamide as biomarkers of the internal exposure to acrylamide in the general population. Mutat Res 580 (1-2): 167–176.
Cibula D, Gompel A, Mueck AO, La VC, Hannaford PC, Skouby SO, Zikan M, Dusek L (2010) Hormonal contraception and risk of cancer. Hum Reprod Update 16 (6): 631–650.
Cook LS, Weiss NS, Doherty JA, Chen C (2006) Endometrial Cancer. In Cancer Epidemiology and Prevention, Schottenfeld D, Fraumeni Jr JF (eds) pp 1027–1043. Oxford University Press: New York.
Doroshyenko O, Fuhr U, Kunz D, Frank D, Kinzig M, Jetter A, Reith Y, Lazar A, Taubert D, Kirchheiner J, Baum M, Eisenbrand G, Berger FI, Bertow D, Berkessel A, Sorgel F, Schomig E, Tomalik-Scharte D (2009) In vivo role of cytochrome P450 2E1 and glutathione-S-transferase activity for acrylamide toxicokinetics in humans. Cancer Epidemiol Biomarkers Prev 18 (2): 433–443.
Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J, Rosso S, Coebergh JWW, Comber H, Forman D, Bray F (2013) Cancer incidence and mortality patterns in Europe: estimates for 40 countries in 2012. Eur J Cancer 49 (6): 1374–1403.
Ferrari P, Freisling H, Duell EJ, Kaaks R, Lujan-Barroso L, Clavel-Chapelon F, Boutron-Ruault MC, Nailler L, Polidoro S, Mattiello A, Palli D, Tumino R, Grioni S, Knuppel S, Tjonneland A, Olsen A, Overvad K, Orfanos P, Katsoulis M, Trichopoulou A, Quiros JR, Ardanaz E, Huerta JM, Etxezarreta PA, Sanchez MJ, Crowe F, Khaw KT, Wareham NJ, Ocke M, Bueno-De-Mesquita B, Peeters PH, Ericson U, Wirfalt E, Hallmans G, Johansson I, Engeset D, Nicolas G, Gallo V, Norat T, Riboli E, Slimani N (2013) Challenges in estimating the validity of dietary acrylamide measurements. Eur J Nutr 52 (5): 1503–1512.
Freisling H, Moskal A, Ferrari P, Nicolas G, Knaze V, Clavel-Chapelon F, Boutron-Ruault MC, Nailler L, Teucher B, Grote VA, Boeing H, Clemens M, Tjonneland A, Olsen A, Overvad K, Quiros JR, Duell EJ, Sanchez MJ, Amiano P, Chirlaque MD, Barricarte A, Khaw KT, Wareham NJ, Crowe FL, Gallo V, Oikonomou E, Naska A, Trichopoulou A, Palli D, Agnoli C, Tumino R, Polidoro S, Mattiello A, Bueno-de-Mesquita HB, Ocke MC, Peeters PH, Wirfalt E, Ericson U, Bergdahl IA, Johansson I, Hjartaker A, Engeset D, Skeie G, Riboli E, Slimani N (2013) Dietary acrylamide intake of adults in the European Prospective Investigation into Cancer and Nutrition differs greatly according to geographical region. Eur J Nutr 52 (4): 1369–1380.
Friedman M (2003) Chemistry, biochemistry, and safety of acrylamide. A review. J Agric Food Chem 51 (16): 4504–4526.
Gierisch JM, Coeytaux RR, Peragallo UR, Havrilesky LJ, Moorman PG, Lowery WJ, Dinan M, McBroom AJ, Hasselblad V, Sanders GD, Myers ER (2013) Oral contraceptive use and risk of breast, cervical, colorectal, and endometrial cancers: a systematic review. Cancer Epidemiol Biomarkers Prev 22 (11): 1931–1943.
Hogervorst JG, Baars BJ, Schouten LJ, Konings EJ, Goldbohm RA, van den Brandt PA (2010) The carcinogenicity of dietary acrylamide intake: a comparative discussion of epidemiological and experimental animal research. Crit Rev Toxicol 40 (6): 485–512.
Hogervorst JG, Fortner RT, Mucci LA, Tworoger SS, Eliassen AH, Hankinson SE, Wilson KM (2013) Associations between dietary acrylamide intake and plasma sex hormone levels. Cancer Epidemiol Biomarkers Prev 22 (11): 2024–2036.
Hogervorst JG, Schouten LJ, Konings EJ, Goldbohm RA, van den Brandt PA (2007) A prospective study of dietary acrylamide intake and the risk of endometrial, ovarian, and breast cancer. Cancer Epidemiol Biomarkers Prev 16 (11): 2304–2313.
IARC (1994) IARC working group on the evaluation of carcinogenic risks to humans: some industrial chemicals. Lyon, 15-22 February 1994. IARC Monogr Eval Carcinog Risks Hum 60: 1–560.
Jamison PM, Noone AM, Ries LA, Lee NC, Edwards BK (2013) Trends in endometrial cancer incidence by race and histology with a correction for the prevalence of hysterectomy, SEER 1992 to 2008. Cancer Epidemiol Biomarkers Prev 22 (2): 233–241.
Johnson KA, Gorzinski SJ, Bodner KM, Campbell RA, Wolf CH, Friedman MA, Mast RW (1986) Chronic toxicity and oncogenicity study on acrylamide incorporated in the drinking water of Fischer 344 rats. Toxicol Appl Pharmacol 85 (2): 154–168.
Kaaks R, Lukanova A, Kurzer MS (2002) Obesity, endogenous hormones, and endometrial cancer risk: a synthetic review. Cancer Epidemiol Biomarkers Prev 11 (12): 1531–1543.
Larsson SC, Hakansson N, Akesson A, Wolk A (2009) Long-term dietary acrylamide intake and risk of endometrial cancer in a prospective cohort of Swedish women. Int J Cancer 124 (5): 1196–1199.
LoPachin RM, Gavin T (2008) Acrylamide-induced nerve terminal damage: relevance to neurotoxic and neurodegenerative mechanisms. J Agric Food Chem 56 (15): 5994–6003.
Obon-Santacana M, Slimani N, Lujan-Barroso L, Travier N, Hallmans G, Freisling H, Ferrari P, Boutron-Ruault MC, Racine A, Clavel F, Saieva C, Pala V, Tumino R, Mattiello A, Vineis P, Arguelles M, Ardanaz E, Amiano P, Navarro C, Sanchez MJ, Molina ME, Key T, Khaw KT, Wareham N, Peeters PH, Trichopoulou A, Bamia C, Trichopoulos D, Boeing H, Kaaks R, Katzke V, Ye W, Sund M, Ericson U, Wirfalt E, Overvad K, Tjonneland A, Olsen A, Skeie G, Asli LA, Weiderpass E, Riboli E, Bueno-de-Mesquita HB, Duell EJ (2013) Dietary intake of acrylamide and pancreatic cancer risk in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort. Ann Oncol 24 (10): 2645–2651.
Riboli E, Hunt KJ, Slimani N, Ferrari P, Norat T, Fahey M, Charrondiere UR, Hemon B, Casagrande C, Vignat J, Overvad K, Tjonneland A, Clavel-Chapelon F, Thiebaut A, Wahrendorf J, Boeing H, Trichopoulos D, Trichopoulou A, Vineis P, Palli D, Bueno-de-Mesquita HB, Peeters PH, Lund E, Engeset D, Gonzalez CA, Barricarte A, Berglund G, Hallmans G, Day NE, Key TJ, Kaaks R, Saracci R (2002) European Prospective Investigation into Cancer and Nutrition (EPIC): study populations and data collection. Public Health Nutr 5 (6B): 1113–1124.
Schoenfeld D (1982) Partial residuals for the proportional hazards regression model. Biometrica 69 (1): 239–241.
Setiawan VW, Yang HP, Pike MC, McCann SE, Yu H, Xiang YB, Wolk A, Wentzensen N, Weiss NS, Webb PM, van den Brandt PA, van de Vijver K, Thompson PJ, Strom BL, Spurdle AB, Soslow RA, Shu XO, Schairer C, Sacerdote C, Rohan TE, Robien K, Risch HA, Ricceri F, Rebbeck TR, Rastogi R, Prescott J, Polidoro S, Park Y, Olson SH, Moysich KB, Miller AB, McCullough ML, Matsuno RK, Magliocco AM, Lurie G, Lu L, Lissowska J, Liang X, Lacey JV Jr., Kolonel LN, Henderson BE, Hankinson SE, Hakansson N, Goodman MT, Gaudet MM, Garcia-Closas M, Friedenreich CM, Freudenheim JL, Doherty J, De Vivo I, Courneya KS, Cook LS, Chen C, Cerhan JR, Cai H, Brinton LA, Bernstein L, Anderson KE, Anton-Culver H, Schouten LJ, Horn-Ross PL (2013) Type I and II endometrial cancers: have they different risk factors? J Clin Oncol 31 (20): 2607–2618.
Slimani N, Ferrari P, Ocke M, Welch A, Boeing H, Liere M, Pala V, Amiano P, Lagiou A, Mattisson I, Stripp C, Engeset D, Charrondiere R, Buzzard M, Staveren W, Riboli E (2000) Standardization of the 24-hour diet recall calibration method used in the European Prospective Investigation into Cancer and Nutrition (EPIC): general concepts and preliminary results. Eur J Clin Nutr 54 (12): 900–917.
Tareke E, Rydberg P, Karlsson P, Eriksson S, Tornqvist M (2002) Analysis of acrylamide, a carcinogen formed in heated foodstuffs. J Agric Food Chem 50 (17): 4998–5006.
Tavassoli FA, Devilee P (2003) Tumours of the Uterine Corpus. In World Health Organization Classification Tumours. Pathology and Genetics of Tumours of the Breast and Female Genital Organs, Tavassoli FA, Devilee P (eds) pp 218–258. IARC Press: Lyon.
Terry PD, Rohan TE, Franceschi S, Weiderpass E (2004) Endometrial Cancer. In Tobacco and Public Health: Science and Policy, Boyle P, Gray N, Henningfield J, Seffrin J, Zatonski W (eds) pp 523–545. Oxford University Press: New York.
Vesper HW, Slimani N, Hallmans G, Tjonneland A, Agudo A, Benetou V, Bingham S, Boeing H, Boutron-Ruault MC, Bueno-de-Mesquita HB, Chirlaque D, Clavel-Chapelon F, Crowe F, Drogan D, Ferrari P, Johansson I, Kaaks R, Linseisen J, Lund E, Manjer J, Mattiello A, Palli D, Peeters PH, Rinaldi S, Skeie G, Trichopoulou A, Vineis P, Wirfalt E, Overvad K, Stromberg U (2008) Cross-sectional study on acrylamide hemoglobin adducts in subpopulations from the European Prospective Investigation into Cancer and Nutrition (EPIC) Study. J Agric Food Chem 56 (15): 6046–6053.
Voss S, Charrondiere UR, Slimani N, Kroke A, Riboli E, Wahrendorf J, Boeing H (1998) [EPIC-SOFT a European computer program for 24-hour dietary protocols]. Z Ernahrungswiss 37 (3): 227–233.
Wareham NJ, Jakes RW, Rennie KL, Schuit J, Mitchell J, Hennings S, Day NE (2003) Validity and repeatability of a simple index derived from the short physical activity questionnaire used in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Public Health Nutr 6 (4): 407–413.
Willett WC (1998) Nutritional Epidemiology. Oxford University Press: New York.
Wilson KM, Balter K, Adami HO, Gronberg H, Vikstrom AC, Paulsson B, Tornqvist M, Mucci LA (2009) Acrylamide exposure measured by food frequency questionnaire and hemoglobin adduct levels and prostate cancer risk in the Cancer of the Prostate in Sweden Study. Int J Cancer 124 (10): 2384–2390.
Wilson KM, Mucci LA, Rosner BA, Willett WC (2010) A prospective study on dietary acrylamide intake and the risk for breast, endometrial, and ovarian cancers. Cancer Epidemiol Biomarkers Prev 19 (10): 2503–2515.
Acknowledgements
The author’s responsibilities were as follows: ER, RK, NS, LL-B, HF, PF, LD, NC-B, LB, RTF, HB, A Tjønneland, AO, VM, EM-M, NL, M-DC, EA, K-TK, NW, RCT, YL, MAM, A Trichopoulou, VB, DT, CS, SS, RT, CS, RG, HBB-d-M, E Wirfält, UE, AI, NO, GS, ITG, E Weiderpass, and NCO-M: designed and conducted the multicenter EPIC cohort study. NS, EJD, RK, and MO-S: conducted the research. MO-S: analysed the data. MO-S and EJD: wrote the manuscript. MO-S and EJD: had primary responsibility for the final content. MO-S is affiliated with the University of Barcelona. All authors read and approved the final version of the manuscript. This work was partially supported by the Wereld Kanker Onderzoek Fonds (WCRF NL) (grant WCRF 2011/442) and by the Health Research Fund (FIS) of the Spanish Ministry of Health (Exp PI11/01473). The coordination of EPIC is financially supported by the European Commission (DG-SANCO) and the International Agency for Research on Cancer. The national cohorts are supported by the Health Research Fund (FIS) of the Spanish Ministry of Health, Regional Governments of Andalucía, Asturias, Basque Country, Murcia (no. 6236), Navarra and the Catalan Institute of Oncology, La Caixa (BM 06-130), Red Temática de Investigación Cooperativa en Cáncer (RD12/0036/0018; RD06/0020/0091) (Spain); Danish Cancer Society (Denmark); Ligue contre le Cancer, Institut Gustave Roussy, Mutuelle Générale de l’Education Nationale, Institut National de la Santé et de la Recherche Médicale (INSERM, France); Deutsche Krebshilfe, Deutsches Krebsforschungszentrum (DKFZ) and Federal Ministry of Education and Research (Germany); the Hellenic Health Foundation (Greece); Associazione Italiana per la Ricerca sul Cancro (AIRC) and National Research Council (Italy); Dutch Ministry of Public Health, Welfare and Sports (VWS), Netherlands Cancer Registry (NKR), LK Research Funds, Dutch Prevention Funds, Dutch ZON (Zorg Onderzoek Nederland), World Cancer Research Fund (WCRF) and Statistics Netherlands (The Netherlands); Nordic Center of Excellence in Food, Nutrition and Health-Helga (Norway); Swedish Cancer Society, Swedish Scientific Council and Regional Government of Skåne and Västerbotten (Sweden); Cancer Research UK, Medical Research Council (United Kingdom).
Disclaimer
None of the funding agencies had a role in the design, implementation, analysis or interpretation of study results.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Obón-Santacana, M., Kaaks, R., Slimani, N. et al. Dietary intake of acrylamide and endometrial cancer risk in the European Prospective Investigation into Cancer and Nutrition cohort. Br J Cancer 111, 987–997 (2014). https://doi.org/10.1038/bjc.2014.328
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2014.328
Keywords
This article is cited by
-
Association between CYP2E1 polymorphisms and risk of differentiated thyroid carcinoma
Archives of Toxicology (2016)
-
Dietary acrylamide intake and risk of endometrial cancer in prospective cohort studies
Archives of Gynecology and Obstetrics (2015)