Abstract
Background:
Tumour biopsy for pharmacodynamic (PD) study is increasingly common in early-phase cancer trials. As they are non-diagnostic, the ethical justification for such procedures rests on their knowledge value. On the premise that knowledge value is related to reporting practices and outcome diversity, we assessed in a sample of recent invasive PD studies within cancer trials.
Methods:
We assessed reporting practices and outcomes for PD studies in a convenience sample of cancer trials published from 2000 to 2010 that employed invasive, non-diagnostic tissue procurement. Extracted data were used to measure outcome reporting in individual trials. Using a reporting scale we developed for exploratory purposes, we tested whether reporting varied with study characteristics, such as funding source or drug novelty.
Results:
Reporting varied widely within and across studies. Some practices were sporadically reported, including results of all planned tests (78% trials reporting), use of blinded histopathological assessment (43% trials reporting), biopsy dimensions (38% trials reporting), and description of patient flow through PD analysis (62%). Pharmacodynamic analysis as a primary end point and mandatory biopsy had statistically significant positive relationships with overall quality of reporting. A preponderance of positive results (61% of the studies described positive PD results) suggests possible publication bias.
Conclusion:
Our results highlight the need for PD-reporting guidelines, and suggest several avenues for improving the risk/benefit for studies involving invasive, non-diagnostic tissue procurement.
Similar content being viewed by others
Main
Biopsy for pharmacodynamic (PD) and biomarker analysis is increasingly common in early-phase cancer trials (Twelves, 2006; Goulart et al, 2007). In principle, PD end points can provide evidence of target effects for a drug, and support decision making for subsequent trials (Workman, 2003; Sarker et al, 2007; Sarker and Workman, 2007; Tan et al, 2009). However, many PD studies require invasive procedures like tumour biopsy.
Studies find that many patients are willing to undergo research biopsy (Seah et al, 2013) and that ethics review committees and oncologists may overestimate patient anxiety associated with biopsies (Agulnik et al, 2006). In one study, overall and major complication rates for tumour biopsies were 5.2% and 0.8%, respectively (Overman et al, 2012). However, the majority of patients describe their biopsies as being painful (Agulnik et al, 2006) and other studies indicate that 10% of patients receiving one common procedure – breast tumour biopsy – report moderate-to-severe pain (a more extended discussion of tumour biopsy risk and burden is available at Brown et al (2008); Hemmer et al (2008); Kimmelman et al (2012)). As biopsies often have no value for subjects in terms of clinical management, their ethical justification rests on an expectation that their performance will be redeemed by the value of the knowledge accrued (Olson et al, 2011).
Given that the burdens of such procedures are well understood, debates concerning their application revolve around conflicting views about the scientific utility of PD evidence. Some commentators question whether research biopsies return sufficient knowledge to justify their risks (Dowlati et al, 2001; Parulekar and Eisenhauer, 2004; Davis et al, 2005; Goulart et al, 2007; Ratain and Glassman, 2007). Such critics describe research biopsies as ‘taking without giving in return’ and an ‘expensive distraction’ (Helft and Daugherty, 2006; Ratain and Glassman, 2007; Olson et al, 2011). One critic argues, ‘given that biomarker support of mechanism, or lack thereof, has not contributed to go/no-go decisions in practice, sponsors should reconsider the value of including any biomarker evaluations in phase I oncological studies’ (Ratain and Glassman, 2007). Others insist that the procedures are safe and feasible, and stress the importance of gathering mechanistic evidence in drug development; defenders point to examples where enrichment trials involving biopsy enabled rapid translation of cancer strategies (Kelloff and Sigman, 2005; Agulnik et al, 2006; Cannistra, 2007; Brown et al, 2008; Peppercorn et al, 2010).
Such debates are hampered by a paucity of systematic evidence concerning the knowledge value of PD studies. In part, this reflects the fact that there are no widely accepted measures of knowledge value. In this report, we sought to highlight measures that could improve the risk/knowledge value of tumour biopsies and associated PD analyses. In particular, we measured two objective proxies of knowledge value: reporting practices and outcome diversity. In order for ‘knowledge value’ to accrue, scientific findings must be reported in sufficient detail to permit readers to form or update beliefs. They must also enable others to reproduce findings in studies addressing similar questions. We measured the extent to which publications reported on study elements that were viewed as important in similar studies – those involving tumour prognostic biomarkers. The second proxy builds on the premise that a population of studies is more informative when it reflects a diversity of outcomes for tested hypotheses. Pharmacodynamic studies generally set out to test well-formulated hypotheses about specified target effects. Finding that in a population of PD studies, hypothesised target effects are almost always confirmed suggests either publication bias, limited information gain (as outcomes were predicted in advance of the PD study), or both. Our studies highlight the potential value of reporting standards for PD studies in cancer.
Materials and methods
Our primary objective was to describe the reporting practices in a convenience sample of recent invasive PD studies embedded within cancer trials. Our secondary objectives were to measure diversity of study outcomes and to identify characteristics of studies that correlate with better reporting.
Sample
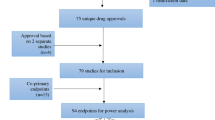
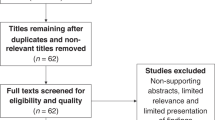
Our study utilised a convenience sample of studies involving tumour biopsy. To capture a sample of studies that involved PD analyses and invasive tissue procurement while excluding the very large volume of studies involving minimally invasive collection (for example, venipuncture), we devised a search strategy that was highly specific. Briefly, we used keywords like ‘biopsy’ and ‘pharmacodynamic’ to search PubMed for articles published from 2000 to 2010 (inclusive) reporting on the use of invasive, non-diagnostic tissue procurement in cancer trials. We excluded articles where (a) non-diagnostic status of tissue procurement was ambiguous; (b) biopsy was not performed; (c) trials did not involve cancer patients; or (d) tissue procurement was minimally invasive (for example, venipuncture). Our search methods are described in greater detail elsewhere (Freeman and Kimmelman, 2012). After an initial screening by title and abstract, eligibility was confirmed using the full report.
Extraction elements
We developed a data extraction form for assessing study reporting and outcomes. Our form (Appendix 1) covered three domains: (1) study characteristics (for example, the year of publication, phase of trial, drug identity); (2) PD study practices and reporting (for example, description of assays, patient flow through study, use of blinded analysis); and (3) study outcomes (for example, confirmation status of PD hypotheses, author conclusions).
Elements within the second domain were adapted from REMARK criteria and supplemented with items described in Eisenhauer et al (2006); McShane et al (2005). Extraction elements and coding conventions were initially developed by JK, and then discussed, refined, and approved by JD and JGM. After piloting extraction against 15 studies, we refined our form and coding criteria.
Extraction
All articles were extracted using paper forms by two reviewers (GF and JK) blinded to the other’s extractions (but not author identities). We interpreted the absence of an affirmative practice statement as the absence of that practice (that is, studies not reporting blinded assessment were coded as not having implemented blinded outcome assessment). Studies were classified as implementing mandatory biopsy when explicitly stated in the report or when tissue samples were collected from all subjects. Data from extractions were entered into an Excel spreadsheet for analysis. Cohen’s κ-inter-rater agreement was calculated; values exceeded 0.8, which we considered ‘good agreement’ (Fleiss, 1981; Toulmonde et al, 2011). Disagreements were resolved through discussion.
Reporting score
We developed a reporting score (RS) in order to explore the range of reporting quality, and to enable a series of tests concerning relationships between study characteristics and reporting. Our score was modelled after those used for prognostic tumour biomarker studies and randomized trials (Lai et al, 2006; Kyzas et al, 2007; Rios et al, 2008; Toulmonde et al, 2011) and was developed through discussions with all authors. It consisted of eight reporting domains: (1) goal and hypothesis; (2) subject eligibility; (3) specimen characteristics; (4) assay protocol; (5) statistics; (6) subject flow; (7) results; (8) discussion. Domains contained one or more evenly weighted reporting variables. Reporting on any item within a domain would result in a fractional score and each domain had a potential score of one. Scores in each domain were summed to calculate an overall RS for each study.
Outcome reporting
Studies were assessed along three outcome categories. The first was results of hypothesis tests. Results were coded as positive where a treatment caused hypothesised changes in targets (that is, an increase in apoptosis assessed by TUNEL staining with a proapoptotic drug) and negative where hypotheses failed confirmation (but were not necessarily disconfirmed). As most studies tested many markers, we coded each report according to whether some, all, or no tested hypotheses were positive. The second outcome category was discussion of results in light of hypotheses. Studies were scored as ‘positive’ when discussions indicated that PD results were consistent with the predicted molecular effects of the agent. Discussions were coded as ambiguous where they gave no clear indication as to whether PD supported the predicted effect of the agent, and were coded as negative where they suggested PD did not support the predicted molecular effects. The third outcome assessed was discussion of results in light of future study planning. Studies were coded as informative where PD results (whether themselves positive or negative) were said to inform planning of future studies. Discussions were coded as uninformative where they gave no clear indication of how PD results related to future investigations.
In a post hoc analysis, we studied the effect of industry funding on PD outcome reporting, focusing on the proportion of positive assay results and the discussion of those results in light of hypotheses and planning for future studies. Fisher’s exact test of independence was used to calculate significance (McDonald, 2009).
Statistics
As this was an exploratory study, we used a convenience sample of PD studies rather than a prospectively determined sample size. We tested a priori-formulated hypotheses of correlation between RS and the following seven variables: (1) the year of publication, (2) public funding, (3) journal impact factor, (4) separate publication for PD results, (5) use of a non-novel test drug, (6) mandatory biopsy; and (7) author assessment of the trial outcome (negative outcome defined as studies recommending that further trials of the investigational agent should not be undertaken). Significance of relationships was tested using one-way ANOVA with SPSS software. We defined significance as P⩽0.05. We did not correct for multiple comparisons.
Results
Sample
Our PubMed search produced a sample of 68 eligible articles reporting results from early-phase cancer trials utilising non-diagnostic biopsy for PD analysis (flow of articles is described in Figure 1; see Appendix 2 for an inventory of studies). Table 1 displays the characteristics of the trials in our sample; Table 2 reports biopsy characteristics within our sample. Ten studies in our sample (15%) actively reported safety events related to biopsy; of these, one reported a single adverse event at or above grade 3.
Our sample captured a total of 2644 patients receiving invasive non-diagnostic biopsies. Although reporting of patient flow through PD studies was poor, we recorded author explanations for discrepancies between patients approached for biopsy, samples collected, and samples analysed. The most common reason for discrepancy was insufficient quality or quantity of sample for analysis (84%), followed by patient refusal (19%) and medical contraindication for biopsy (19%). Missed samples (3%) were because of patient death.
Reporting score
We calculated the RS for each article in our sample. The RS range had a score centred around 5.5 (Figure 2). Some variables, like description of causal pathway and biopsy location, were consistently reported (Table 3). However, there was broad variation within specific domains in the RS. A fifth of articles did not report results for all PD analyses performed; 57% did not report the status of blinding for pathological analysis and 62% did not provide information about the dimensions of the biopsy sample.
Reporting predictors
The use of a non-novel study drug showed positive but non-significant trend towards a higher RS (5.6 vs 5.2, P=0.219). Pharmacodynamics as a primary end point showed a significant positive relationship with RS (5.8 vs 5.1, P=0.04), as did the use of mandatory biopsy (5.9 vs 5.1, P=0.023). We found no relationship between RS and the year of publication, journal impact factor, funding source, or author assessment of the trial outcome.
Pharmacodynamic study outcome
The majority of articles (66%) reported some negative PD results and 10% of the articles reported all negative PD results. Fifty-six percent of studies reported at least one positive PD parameter. The majority of studies (61%) described their PD results as ‘positive’ in discussions (for example, PD results provided evidence of the investigational agent having intended effects on molecular targets).
A large majority of articles (78%) contained a discussion of PD results in relation to the direction of future studies. Among these, 72% discussed possible amendments to the conduct or direction of future studies based on the PD findings of the current study.
Industry funding vs results positivity
Industry-funded trials were more likely to report all or some positive PD results than non-industry-funded studies. No industry-funded trial reported all negative results for PD parameters tested. Trials with industry funding trended towards greater positivity in discussion both in terms of support for the predicted method of action of the drug (75% vs 53%, P=0.11) and planning for future studies (80% vs 67%, P=0.359).
Discussion
Biopsies for PD in anticancer drug trials are often burdensome and entail non-trivial costs. Justification of procedures rests on a favourable gain of scientific knowledge (Weijer and Miller, 2004). Poor PD reporting does not adequately redeem burdens and can produce biased findings that lead to unsuccessful clinical development (Tan et al, 2009). At present, there is little systematic evidence to inform the planning, implementation, and ethical evaluation of PD studies involving invasive tissue procurement.
Our study explored two relatively objective proxies of knowledge value in a convenience sample of PD studies using research biopsies. Encouragingly, a large fraction of studies reported tissue location, procurement method, and discussion of PD results. However, many important items were reported sporadically, including results of all planned tests, use of blinded histopathological assessment, biopsy dimensions, and description of patient flow through the PD portion of the trial. Previous studies of prognostic marker research reporting showed that over 90% of studies reported ‘positive’ outcomes (Kyzas et al, 2005; Kyzas et al, 2007). Disproportionate reporting of positive results was also observed in genetic association studies (Ioannidis et al, 2001). We entered this study expecting near-uniform positivity among PD reports. Instead, we found that two-thirds of articles contained negative outcomes, and a similar proportion described PD analysis as affirming hypotheses in discussion. This is evidence that PD is not characterised by overwhelming publication bias, and that results are not overdetermined at study inception. Nevertheless, that the fraction of studies reporting uniform positivity (34%) vs those reporting uniform negativity (10%) suggests, in our view, the presence of some bias. Whether this bias pertains to publication bias, or enhanced pre-test probability, we are unable to say. Analysis of positivity would be greatly aided if studies declared their primary hypothesis; the only instance where this occurred was in studies that reported only a single PD marker analysis. We further take the fact that a large fraction of PD studies were described as informing decisions for future studies as support for invasive PD evaluation. Future studies should investigate the fraction of PD findings that motivate actual new investigations.
Our study has several limitations. First, some might question the premises guiding our proxy indicators of knowledge value. Poorly reported studies can still hold value, and uniformly positive results can convert modest degrees of belief in drug effects into higher degrees of belief. Still, uniform confirmation would seem a modest gain of information for considerable burden. Second, some items in the RS, similar to blinded outcome assessment, straddle ‘good reporting’ and ‘good methodological practice,’ and high quality reporting can mask poor methodological practice (Huwiler-Muntener et al, 2002; Toulmonde et al, 2011). Third, in line with the exploratory orientation, our study did not capture a comprehensive sample of studies involving research biopsies. A larger sample might have produced different findings and our sample may have been underpowered to detect relationships between study characteristics and reporting quality. Fourth, although our article points to ways that reporting of PD might improve, nothing in our premises, data, or analysis provides a clear basis for deciding whether current research biopsy and PD study practices meet an adequate threshold of knowledge value. Last, our RS scale should be interpreted with caution. It was not the result of a consensus building process (unlike CONSORT and REMARK) (Harris, 2005; McShane et al, 2005; Lai et al, 2006; ‘How CONSORT began’, 2008; Rios et al, 2008; Toulmonde et al, 2011). Furthermore, it gave uniform weighting for each criterion, which may not be appropriate, given that some items probably matter more than others with respect to valid study interpretation. Nevertheless, our scale was at least modelled on validated criteria and we believe its application is justified in the context of this exploratory exercise. Finally, although this study identifies deficiencies in current reporting practices and may aid in the development of consensus guidelines, it must be noted that delay to publication means that current study practices may not be accurately represented in our study.
Our study suggests several avenues investigators, funders, or IRBs might consider for improving the risk–benefit balance of PD studies. First, we recommend the research community develop formalized reporting guidelines similar to REMARK and CONSORT. Second, given our observation that separate PD-reporting trends towards higher quality, and that reporting quality for PD studies may be constrained by word counts at journals, we encourage investigators to consider separate PD publication, using standard methods described in a reference or reporting methods in supplementary materials (Toulmonde et al, 2011). Journal editors may have a role in limiting ‘text limitation bias.’ Third, given that PD components might not be registered in http://www.clinicaltrials.gov, IRBs might have a more active role in promoting reporting and publication by asking investigators to provide a detailed reporting plan for PD studies. A recent article recommended the creation of an online biomarker study registry similar to http://www.clinicaltrials.gov (Andre et al, 2011). We support extending this initiative to PD.
Together with a previous study by our team, our results offer a complex picture of the quality of reporting for PD studies involving non-diagnostic biopsy. A preponderance of positive results, coupled with a finding that 63% of PD studies go unreported suggests biases. Low perceived quality of reports, and low reporting of basic factors like patient flow, suggests considerable room for improvement. On the other hand, some studies demonstrate careful reporting, many negative results are reported, and a large fraction of studies report that PD findings will help guide future investigations. In the end, we conclude that the evidence gathered above provides ammunition for proponents as well as opponents of research biopsies in cancer. In any event, our findings and analysis provide grounds for developing and disseminating PD-reporting standards.
Change history
20 August 2013
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Agulnik M, Oza A, Pond G, Siu L (2006) Impact and perceptions of mandatory tumor biopsies for correlative studies in clinical trials of novel anticancer agents. J Clin Oncol 24: 4801–4807.
Andre F, McShane LM, Michiels S, Ransohoff DF, Altman DG, Reis-Filho JS, Pusztai L (2011) Biomarker studies: a call for a comprehensive biomarker study registry. Nat Rev Clin Oncol 8 (3): 171–176.
Brown A, Wendler D, Camphausen K, Miller F, Citrin D (2008) Performing nondiagnostic research biopsies in irradiated tissue: a review of scientific, clinical, and ethical considerations. J Clin Oncol 26: 3987–3994.
Cannistra S (2007) Performance of biopsies in clinical research. J Clin Oncol 25: 1454–1455.
Davis DW, Takamori R, Raut CP, Xiong HQ, Herbst RS, Stadler WM, McConkey DJ (2005) Pharmacodynamic analysis of target inhibition and endothelial cell death in tumors treated with the vascular endothelial growth factor receptor antagonists SU5416 or SU6668. Clin Cancer Res 11 (2 Pt 1): 678–689.
Dowlati A, Haaga J, Remick SC, Spiro TP, Gerson SL, Liu L, Willson JK (2001) Sequential tumor biopsies in early phase clinical trials of anticancer agents for pharmacodynamic evaluation. Clin Cancer Res 7 (10): 2971–2976.
Eisenhauer E, Twelves C, Buyse M (2006) Phase 1 Cancer Clinical Trials: A Practical Guide. Oxford University Press: New York, NY, USA.
Fleiss JL (1981) Statistical Methods For Rates And Proportions 2nd edn. Wiley: New York, NY, USA.
Freeman GA, Kimmelman J (2012) Publication and reporting conduct for pharmacodynamic analyses of tumor tissue in early phase oncology trials. Clin Cancer Res 18: 6478–6484.
Goulart BH, Clark JW, Pien HH, Roberts TG, Finkelstein SN, Chabner BA (2007) Trends in the use and role of biomarkers in phase I oncology trials. Clin Cancer Res 13 (22 Pt 1): 6719–6726.
Harris AL (2005) REporting recommendations for tumour MARKer prognostic studies (REMARK). Br J Cancer 93 (4): 385–386.
Helft P, Daugherty C (2006) Are we taking without giving in return? The ethics of research-related biopsies and the benefits of clinical trial participation. J Clin Oncol 24: 4793–4795.
Hemmer JM, Kelder JC, van Heesewijk HP (2008) Stereotactic large-core needle breast biopsy: analysis of pain and discomfort related to the biopsy procedure. Eur Radiol 18 (2): 351–354.
How CONSORT began (2008) Available from http://www.consort-statement.org/about-consort/history/how-consort-began/.
Huwiler-Muntener K, Juni P, Junker C, Egger M (2002) Quality of reporting of randomized trials as a measure of methodologic quality. JAMA 287 (21): 2801–2804.
Ioannidis JP, Ntzani EE, Trikalinos TA, Contopoulos-Ioannidis DG (2001) Replication validity of genetic association studies. Nat Genet 29 (3): 306–309.
Kelloff GJ, Sigman CC (2005) New science-based endpoints to accelerate oncology drug development. (Review). Eur J Cancer 41 (4): 491–501.
Kimmelman J, Lemmens T, Kim SY (2012) Analysis of consent validity for invasive, nondiagnostic research procedures. (Research Support, Non-U.S. Gov′t). IRB 34 (5): 1–7.
Kyzas PA, Denaxa-Kyza D, Ioannidis JPA (2007) Almost all articles on cancer prognostic markers report statistically significant results. Eur J Cancer 43 (17): 2559–2579.
Kyzas PA, Loizou KT, Ioannidis JPA (2005) Selective reporting biases in cancer prognostic factor studies. J Natl Cancer Inst 97 (14): 1043–1055.
Lai R, Chu R, Fraumeni M, Thabane L (2006) Quality of randomized controlled trials reporting in the primary treatment of brain tumors. J Clin Oncol 24 (7): 1136–1144.
McDonald J. H (2009) Handbook of Biological Statistics 2nd edn. Sparky House Publishing: Baltimore, MD, USA.
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM (2005) Reporting recommendations for tumor marker prognostic studies (REMARK). J Natl Cancer Inst 97 (16): 1180–1184.
Olson EM, Lin NU, Krop IE, Winer EP (2011) The ethical use of mandatory research biopsies. Nat Rev Clin Oncol 8 (10): 620–625.
Overman MJ, Modak J, Kopetz S, Murthy R, Yao JC, Hicks ME, Tam AL (2012) Use of research biopsies in clinical trials: are risks and benefits adequately discussed? J Clin Oncol 31 (1): 17–22.
Parulekar WR, Eisenhauer EA (2004) Phase I trial design for solid tumor studies of targeted, non-cytotoxic agents: theory and practice. J Natl Cancer Inst 96 (13): 990–997.
Peppercorn J, Shapira I, Collyar D, Deshields T, Lin N, Krop I, Bertagnolli MM (2010) Ethics of mandatory research biopsy for correlative end points within clinical trials in oncology. J Clin Oncol 28 (15): 2635–2640.
Ratain MJ, Glassman RH (2007) Biomarkers in phase I oncology trials: signal, noise, or expensive distraction? Clin Cancer Res 13 (22 Pt 1): 6545–6548.
Rios LP, Odueyungbo A, Moitri MO, Rahman MO, Thabane L (2008) Quality of reporting of randomized controlled trials in general endocrinology literature. J Clin Endocrinol Metab 93 (10): 3810–3816.
Sarker D, Pacey S, Workman P (2007) Use of pharmacokinetic/pharmacodynamic biomarkers to support rational cancer drug development. Biomark Med 1 (3): 399–417.
Sarker D, Workman P (2007) Pharmacodynamic biomarkers for molecular cancer therapeutics. Adv Cancer Res 96: 213–268.
Seah DS, Scott SM, Najita J, Openshaw T, Krag K, Frank E, Sohl J, Stadler ZK, Garrett M, Silverman SG, Peppercorn J, Winer EP, Come SE, Lin NU (2013) Attitudes of patients with metastatic breast cancer toward research biopsies. Ann Oncol 24 (7): 1853–1859.
Tan DS, Thomas GV, Garrett MD, Banerji U, De Bono JS, Kaye SB, Workman P (2009) Biomarker-driven early clinical trials in oncology: a paradigm shift in drug development. Cancer J 15 (5): 406–420.
Toulmonde M, Bellera C, Mathoulin-Pelissier S, Debled M, Bui B, Italiano A (2011) Quality of randomized controlled trials reporting in the treatment of sarcomas. J Clin Oncol 29 (9): 1204–1209.
Twelves C (2006) Practical aspects of pharmacokinetics and pharmacodynamics. In Eisenhauer EA, Twelves C, Buyse M (eds) Phase 1 Cancer Clinical Trials: A Practical Guide pp 209–243. Oxford University Press: New York, NY, USA.
Weijer C, Miller PB (2004) When are research risks reasonable in relation to anticipated benefits? Nat Med 10 (6): 570–573.
Workman P (2003) How much gets there and what does it do?: the need for better pharmacokinetic and pharmacodynamic endpoints in contemporary drug discovery and development. Curr Pharm Des 9 (11): 891–902.
Acknowledgements
This work was funded by the Canadian Institutes of Health Research (EOG 102824).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
Appendices
Appendix 1




Appendix 2
Inventory of studies included in sample
Adjei AA, RB Cohen, WFranklin, CMorris, DWilson, JRMolina, LJHanson, L Gore, L Chow, S Leong, L Maloney, G Gordon, H Simmons, A Marlow, K Litwiler, S Brown, G Poch, K Kane, J Haney, SG Eckhardt (2008) Phase I pharmacokinetic and pharmacodynamic study of the oral, small-molecule mitogen-activated protein kinase kinase 1/2 inhibitor Azd6244 (Arry-142886) in patients with advanced cancers. J Clin Oncol 26(13): 2139–2146.
Agulnik M, EW Cohen, RB Cohen, EX Chen, EE Vokes, SJ Hotte, EWinquist, S Laurie, DN Hayes, JE Dancey, S Brown, GR Pond, I Lorimer, M Daneshmand, J Ho, MS Tsao, LL Siu (2007) Phase Ii study of lapatinib in recurrent or metastatic epidermal growth factor receptor and/or Erbb2 expressing adenoid cystic carcinoma and non adenoid cystic carcinoma malignant tumors of the salivary glands. J Clin Oncol 25(25): 3978–3984.
Agulnik M, G da Cunha Santos, D Hedley, T Nicklee, PP Dos Reis, J Ho, GR Pond, H Chen, S Chen, Y Shyr, E Winquist, D Soulieres, EX Chen, JA Squire, P Marrano, S Kamel-Reid, J Dancey, LL Siu, MS Tsao (2007) Predictive and pharmacodynamic biomarker studies in tumor and skin tissue samples of patients with recurrent or metastatic squamous cell carcinoma of the head and neck treated with erlotinib. J Clin Oncol 25(16): 2184–2190.
Albanell J, F Rojo, S Averbuch, A Feyereislova, JM Mascaro, R Herbst, P LoRusso, D Rischin, S Sauleda, J Gee, RI Nicholson, J Baselga (2002) Pharmacodynamic studies of the epidermal growth factor receptor inhibitor zd1839 in skin from cancer patients: histopathologic and molecular consequences of receptor inhibition. J Clin Oncol 20(1): 110–124.
Annunziata CM, AJ Walker, L Minasian, M Yu, H Kotz, BJ Wood, K Calvo, P Choyke, D Kimm, SM Steinberg, EC Kohn (2010) Vandetanib, designed to inhibit Vegfr2 and Egfr signaling, had no clinical activity as monotherapy for recurrent ovarian cancer and no detectable modulation of Vegfr2. Clin Cancer Res 16(2): 664–672.
Badros A, AM Burger, S Philip, R Niesvizky, SS Kolla, O Goloubeva, C Harris, J Zwiebel, JJ Wright, I Espinoza-Delgado, MR Baer, JL Holleran, MJ Egorin, S Grant (2009) Phase I study of vorinostat in combination with bortezomib for relapsed and refractory multiple myeloma. Clin Cancer Res 15(16): 5250–5257.
Baselga J, J Albanell, A Ruiz, A Lluch, P Gascon, V Guillem, S Gonzalez, S Sauleda, I Marimon, JM Tabernero, MT Koehler, F Rojo (2005) Phase Ii and tumor pharmacodynamic study of gefitinib in patients with advanced breast cancer. J Clin Oncol 23(23): 5323–5333.
Baselga J, D Rischin, M Ranson, H Calvert, E Raymond, DG Kieback, SB Kaye, L Gianni, A Harris, T Bjork, SD Averbuch, A Feyereislova, H Swaisland, F Rojo, J Albanell (2002) Phase I safety, pharmacokinetic, and pharmacodynamic trial of zd1839, a selective oral epidermal growth factor receptor tyrosine kinase inhibitor, in patients with five selected solid tumor types. J Clin Oncol 20(21): 4292–4302.
Baselga J, V Semiglazov, P van Dam, A Manikhas, M Bellet, J Mayordomo, M Campone, E Kubista, R Greil, G Bianchi, J Steinseifer, B Molloy, E Tokaji, H Gardner, P Phillips, M Stumm, HA Lane, JM Dixon, W Jonat, HS Rugo (2009) Phase Ii randomized study of neoadjuvant everolimus plus letrozole compared with placebo plus letrozole in patients with estrogen receptor-positive breast cancer. J Clin Oncol 27(16): 2630–2637.
Borghaei H, K Alpaugh, G Hedlund, G Forsberg, C Langer, A Rogatko, R Hawkins, S Dueland, U Lassen, RB Cohen (2009) Phase I dose escalation, pharmacokinetic and pharmacodynamic study of naptumomab estafenatox alone in patients with advanced cancer and with docetaxel in patients with advanced non-small-cell lung cancer. J Clin Oncol 27(25): 4116–4123.
Boss DS, GK Schwartz, MR Middleton, DD Amakye, H Swaisland, RS Midgley, M Ranson, S Danson, H Calvert, R Plummer, C Morris, RD Carvajal, LR Chirieac, JH Schellens, GI Shapiro (2010) Safety, tolerability, pharmacokinetics and pharmacodynamics of the oral cyclin-dependent kinase inhibitor azd5438 when administered at intermittent and continuous dosing schedules in patients with advanced solid tumours. Ann Oncol 21(4): 884–894.
Busam KJ, P Capodieci, R Motzer, T Kiehn, D Phelan, AC Halpern (2001) Cutaneous side-effects in cancer patients treated with the antiepidermal growth factor receptor antibody C225. Br J Dermatol 144(6): 1169–1176.
Byrd JC, G Marcucci, MR Parthun, JJ Xiao, RB Klisovic, M Moran, TS Lin, S Liu, AR Sklenar, ME Davis, DM Lucas, B Fischer, R Shank, SL Tejaswi, P Binkley, J Wright, KK Chan, MR Grever (2005) A phase 1 and pharmacodynamic study of depsipeptide (Fk228) in chronic lymphocytic leukemia and acute myeloid leukemia. Blood 105(3): 959–967.
Camidge DR, MN Pemberton, JW Growcott, D Johnstone, PJ Laud, JR Foster, KJ Randall, AM Hughes (2005) Assessing proliferation, cell-cycle arrest and apoptotic end points in human buccal punch biopsies for use as pharmacodynamic biomarkers in drug development. Br J Cancer 93(2): 208–215.
Decensi A, R Torrisi, S Bruno, M Costantini, A Curotto, G Nicolo, B Malcangi, L Baglietto, GP Bruttini, B Gatteschi, G Rondanina, M Varaldo, M Perloff, WF Malone, P Bruzzi (2000) Randomized trial of fenretinide in superficial bladder cancer using DNA flow cytometry as an intermediate end point. Cancer Epidemiol Biomarkers Prev 9(10): 1071–1078.
Dowlati A, K Robertson, T Radivoyevitch, J Waas, NP Ziats, P Hartman, FW Abdul-Karim, JK Wasman, J Jesberger, J Lewin, K McCrae, P Ivy, SC Remick (2005) Novel phase i dose de-escalation design trial to determine the biological modulatory dose of the antiangiogenic agent Su5416. Clin Cancer Res 11(21): 7938–7944.
Duran I, J Kortmansky, D Singh, H Hirte, W Kocha, G Goss, L Le, A Oza, T Nicklee, J Ho, D Birle, GR Pond, D Arboine, J Dancey, S Aviel-Ronen, MS Tsao, D Hedley, LL Siu (2006) A phase Ii clinical and pharmacodynamic study of temsirolimus in advanced neuroendocrine carcinomas. Br J Cancer 95(9): 1148–1154.
Dy GK, JP Thomas, G Wilding, L Bruzek, S Mandrekar, C Erlichman, D Alberti, K Binger, HC Pitot, SR Alberts, LJ Hanson, R Marnocha, K Tutsch, SH Kaufmann, AA Adjei (2005) A phase I and pharmacologic trial of two schedules of the proteasome inhibitor, Ps-341 (Bortezomib, Velcade), in patients with advanced cancer. Clin Cancer Res 11(9): 3410–3416.
Eder JP, GI Shapiro, LJ Appleman, AX Zhu, D Miles, H Keer, B Cancilla, F Chu, S Hitchcock-Bryan, L Sherman, S McCallum, EI Heath, SA Boerner, PM LoRusso (2010) A phase I study of foretinib, a multi-targeted inhibitor of C-Met and vascular endothelial growth factor receptor 2. Clin Cancer Res 16(13): 3507–3516.
Fakih MG, L Pendyala, G Fetterly, K Toth, JA Zwiebel, I Espinoza-Delgado, A Litwin, YM Rustum, ME Ross, JL Holleran, MJ Egorin (2009) A phase I, pharmacokinetic and pharmacodynamic study on vorinostat in combination with 5-fluorouracil, leucovorin, and oxaliplatin in patients with refractory colorectal cancer. Clin Cancer Res 15(9): 3189–3195.
Fakih MG, A Rajput, GY Yang, L Pendyala, K Toth, JL Smith, DD Lawrence, YM Rustum (2006) A phase I study of weekly intravenous oxaliplatin in combination with oral daily capecitabine and radiation therapy in the neoadjuvant treatment of rectal adenocarcinoma. Int J Radiat Oncol Biol Phys 65(5): 1462–1470.
Felip E, F Rojo, M Reck, A Heller, B Klughammer, G Sala, S Cedres, S Peralta, H Maacke, D Foernzler, M Parera, J Mocks, C Saura, U Gatzemeier, J Baselga (2008) A phase Ii pharmacodynamic study of erlotinib in patients with advanced non-small cell lung cancer previously treated with platinum-based chemotherapy. Clin Cancer Res 14(12): 3867–3874.
Fracasso PM, H Burris 3rd, MA Arquette, R Govindan, F Gao, LP Wright, SA Goodner, FA Greco, SF Jones, N Willcut, C Chodkiewicz, A Pathak, GM Springett, GR Simon, DM Sullivan, R Marcelpoil, SD Mayfield, D Mauro, CR Garrett (2007) A phase 1 escalating single-dose and weekly fixed-dose study of cetuximab: pharmacokinetic and pharmacodynamic rationale for dosing. Clin Cancer Res 13(3): 986–993.
Goss G, H Hirte, WH Miller Jr, IA Lorimer, D Stewart, G Batist, DA Parolin, P Hanna, S Stafford, J Friedmann, W Walsh, S Mathews, L Douglas, LK Seymour (2005) A phase I study of oral Zd 1839 given daily in patients with solid tumors: Ind.122, a study of the investigational new drug program of the national cancer institute of canada clinical trials group.’ Invest New Drugs 23(2): 147–155.
Haddad RI, LJ Weinstein, TJ Wieczorek, N Bhattacharya, H Raftopoulos, MW Oster, X Zhang, VM Latham Jr, R Costello, J Faucher, C DeRosa, M Yule, LP Miller, M Loda, MR Posner, GI Shapiro (2004) A phase Ii clinical and pharmacodynamic study of E7070 in patients with metastatic, recurrent, or refractory squamous cell carcinoma of the head and neck: modulation of retinoblastoma protein phosphorylation by a novel chloroindolyl sulfonamide cell cycle inhibitor. Clin Cancer Res 10(14): 4680–4687.
Herbst RS, AM Davies, RB Natale, TP Dang, JH Schiller, LL Garland, VA Miller, D Mendelson, AD Van den Abbeele, Y Melenevsky, DJ De Vries, DA Eberhard, B Lyons, SG Lutzker, BE Johnson (2007) Efficacy and safety of single-agent pertuzumab, a human epidermal receptor dimerization inhibitor, in patients with non small cell lung cancer. Clin Cancer Res 13(20): 6175–6181.
Jansen B, V Wacheck, E Heere-Ress, H Schlagbauer-Wadl, C Hoeller, T Lucas, M Hoermann, U Hollenstein, K Wolff, H Pehamberger (2000) Chemosensitisation of malignant melanoma by Bcl2 antisense therapy. Lancet 356(9243): 1728–1733.
Jatoi A, VJ Suman, P Schaefer, M Block, C Loprinzi, P Roche, S Garneau, R Morton, PJ Stella, SR Alberts, M Pittelkow, J Sloan, R Pagano (2003) A phase Ii study of topical ceramides for cutaneous breast cancer. Breast Cancer Res Treat 80(1): 99–104.
Joensuu G, T Joensuu, P Nokisalmi, C Reddy, J Isola, M Ruutu, M Kouri, PA Kupelian, J Collan, S Pesonen, A Hemminki (2010) A phase I/Ii trial of gefitinib given concurrently with radiotherapy in patients with nonmetastatic prostate cancer. Int J Radiat Oncol Biol Phys 78(1): 42–49.
Koon HB, GJ Bubley, L Pantanowitz, D Masiello, B Smith, K Crosby, J Proper, W Weeden, TE Miller, P Chatis, MJ Egorin, SR Tahan, BJ Dezube (2005) Imatinib-induced regression of AIDS-related kaposi’s sarcoma. J Clin Oncol 23(5): 982–989.
Kummar S, R Kinders, ME Gutierrez, L Rubinstein, RE Parchment, LR Phillips, J Ji, A Monks, JA Low, A Chen, AJ Murgo, J Collins, SM Steinberg, H Eliopoulos, VL Giranda, G Gordon, L Helman, R Wiltrout, JE Tomaszewski, JH Doroshow (2009) Phase 0 clinical trial of the poly (Adp-ribose) polymerase inhibitor Abt-888 in patients with advanced malignancies. J Clin Oncol 27(16): 2705–2711.
Lin J, J Gilbert, MA Rudek, JA Zwiebel, S Gore, A Jiemjit, M Zhao, SD Baker, RF Ambinder, JG Herman, RC Donehower, MA Carducci (2009) A phase I dose-finding study of 5-azacytidine in combination with sodium phenylbutyrate in patients with refractory solid tumors. Clin Cancer Res 15(19): 6241–6249.
LoRusso PM, SS Krishnamurthi, JJ Rinehart, LM Nabell, L Malburg, PB Chapman, SE DePrimo, S Bentivegna, KD Wilner, W Tan, AD Ricart (2010) Phase I pharmacokinetic and pharmacodynamic study of the oral Mapk/Erk kinase inhibitor Pd-0325901 in patients with advanced cancers. Clin Cancer Res 16(6): 1924–1937.
Mackay H, D Hedley, P Major, C Townsley, M Mackenzie, M Vincent, P Degendorfer, MS Tsao, T Nicklee, D Birle, J Wright, L Siu, M Moore, A Oza (2005) A phase Ii trial with pharmacodynamic endpoints of the proteasome inhibitor bortezomib in patients with metastatic colorectal cancer. Clin Cancer Res 11(15): 5526–5533.
Makower D, A Rozenblit, H Kaufman, M Edelman, ME Lane, J Zwiebel, H Haynes, S Wadler (2003) ‘Phase Ii clinical trial of intralesional administration of the oncolytic adenovirus onyx-015 in patients with hepatobiliary tumors with correlative P53 studies. Clin Cancer Res 9(2): 693–702.
Mathew P, PF Thall, D Jones, C Perez, C Bucana, P Troncoso, SJ Kim, IJ Fidler, C Logothetis (2004) Platelet-derived growth factor receptor inhibitor imatinib mesylate and docetaxel: a modular phase I trial in androgen-independent prostate cancer. J Clin Oncol 22(16): 3323–3329.
McDaid HM, S Mani, HJ Shen, F Muggia, D Sonnichsen, SB Horwitz (2002) Validation of the pharmacodynamics of Bms-247550, an analogue of epothilone B, during a phase I clinical study. Clin Cancer Res 8(7): 2035–2043.
Millward MJ, A Joshua, R Kefford, S Aamdal, D Thomson, P Hersey, G Toner, K Lynch (2005) Multi-centre phase Ii trial of the polyamine synthesis inhibitor Sam486a (Cgp48664) in patients with metastatic melanoma. Invest New Drugs 23(3): 253–256.
Moschos SJ, CA Sander, W Wang, SL Reppert, LM Drogowski, DM Jukic, UN Rao, C Athanassiou, M Buzoianu, M Mandic, L Richman, L McKinney, J Leininger, DA Tice, L Hammershaimb, JM Kirkwood (2010) Pharmacodynamic (phase 0) study using etaracizumab in advanced melanoma.’ J Immunother 33(3): 316–325.
Moulder SL, WF Symmans, DJ Booser, TL Madden, C Lipsanen, L Yuan, AM Brewster, M Cristofanilli, KK Hunt, TA Buchholz, J Zwiebel, V Valero, GN Hortobagyi, FJ Esteva (2008) Phase I/Ii study of G3139 (Bcl-2 antisense oligonucleotide) in combination with doxorubicin and docetaxel in breast cancer. Clin Cancer Res 14(23): 7909–7916.
Mullamitha SA, NC Ton, GJ Parker, A Jackson, PJ Julyan, C Roberts, GA Buonaccorsi, Y Watson, K Davies, S Cheung, L Hope, JW Valle, JA Radford, J Lawrance, MP Saunders, MC Munteanu, MT Nakada, JA Nemeth, HM Davis, Q Jiao, U Prabhakar, Z Lang, RE Corringham, RA Beckman, GC Jayson (2007) ‘Phase I evaluation of a fully human anti-alphav integrin monoclonal antibody (Cnto 95) in patients with advanced solid tumors. Clin Cancer Res 13(7): 2128–2135.
O′Neil BH, L Raftery, BF Calvo, AB Chakravarthy, A Ivanova, MO Myers, HJ Kim, E Chan, PE Wise, LS Caskey, SA Bernard, HK Sanoff, RM Goldberg, JE Tepper (2010) A phase I study of bortezomib in combination with standard 5-fluorouracil and external-beam radiation therapy for the treatment of locally advanced or metastatic rectal cancer. Clin Colorectal Cancer 9(2): 119–125.
Ord JJ, E Streeter, A Jones, K Le Monnier, D Cranston, J Crew, SP Joel, MA Rogers, RE Banks, IS Roberts, AL Harris (2005) Phase I trial of intravesical suramin in recurrent superficial transitional cell bladder carcinoma. Br J Cancer 92(12): 2140–2147.
Overmoyer B, P Fu, C Hoppel, T Radivoyevitch, R Shenk, M Persons, P Silverman, K Robertson, NP Ziats, JK Wasman, FW Abdul-Karim, JA Jesberger, J Duerk, P Hartman, S Hanks, J Lewin, A Dowlati, K McCrae, P Ivy, SC Remick (2007) Inflammatory breast cancer as a model disease to study tumor angiogenesis: results of a phase Ib trial of combination Su5416 and doxorubicin. Clin Cancer Res 13(19): 5862–5868.
Perez RP, LD Lewis, AP Beelen, AJ Olszanski, N Johnston, CH Rhodes, B Beaulieu, MS Ernstoff, A Eastman (2006) Modulation of cell cycle progression in human tumors: a pharmacokinetic and tumor molecular pharmacodynamic study of cisplatin plus the Chk1 inhibitor Ucn-01 (Nsc 638850). Clin Cancer Res 12(23): 7079–7085.
Perotti A, A Locatelli, C Sessa, D Hess, L Vigano, G Capri, M Maur, T Cerny, S Cresta, F Rojo, J Albanell, S Marsoni, I Corradino, L Berk, VM Rivera, F Haluska, L Gianni (2010) Phase Ib study of the Mtor inhibitor ridaforolimus with capecitabine. J Clin Oncol 28(30): 4554–4561.
Posadas EM, MS Liel, V Kwitkowski, L Minasian, AK Godwin, MM Hussain, V Espina, BJ Wood, SM Steinberg, EC Kohn (2007) A phase Ii and pharmacodynamic study of gefitinib in patients with refractory or recurrent epithelial ovarian cancer. Cancer 109(7): 1323–1330.
Puglisi F, GG Cardellino, D Crivellari, C Di Loreto, MD Magri, AM Minisini, M Mansutti, C Andreetta, S Russo, D Lombardi, T Perin, G Damante, A Veronesi (2008) Thymidine phosphorylase expression is associated with time to progression in patients receiving low-dose, docetaxel-modulated capecitabine for metastatic breast cancer. Ann Oncol 19(9): 1541–1546.
Ramanathan RK, MJ Egorin, C Erlichman, SC Remick, SS Ramalingam, C Naret, JL Holleran, CJ TenEyck, SP Ivy, CP Belani (2010) Phase I pharmacokinetic and pharmacodynamic study of 17-dimethylaminoethylamino-17-demethoxygeldanamycin, an inhibitor of heat-shock protein 90, in patients with advanced solid tumors. J Clin Oncol 28(9): 1520–1526.
Ranson M, P Hersey, D Thompson, J Beith, GA McArthur, A Haydon, ID Davis, RF Kefford, P Mortimer, PA Harris, S Baka, A Seebaran, A Sabharwal, AJ Watson, GP Margison, MR Middleton (2007) Randomized trial of the combination of lomeguatrib and temozolomide compared with temozolomide alone in chemotherapy naive patients with metastatic cutaneous melanoma. J Clin Oncol 25(18): 2540–2545.
Rao S, N Starling, D Cunningham, M Benson, A Wotherspoon, C Lupfert, R Kurek, J Oates, J Baselga, A Hill (2008) Phase I study of epirubicin, cisplatin and capecitabine plus matuzumab in previously untreated patients with advanced oesophagogastric cancer. Br J Cancer 99(6): 868–874.
Rojo F, E Gracias, N Villena, T Cruz, JM Corominas, I Corradino, M Cedeno, C Campas, M Osorio, N Iznaga, B Bellosillo, A Rovira, S Marsoni, P Gascon, S Serrano, C Sessa, T Crombet, J Albanell (2010) Pharmacodynamic trial of nimotuzumab in unresectable squamous cell carcinoma of the head and neck: a sendo foundation study. Clin Cancer Res 16(8): 2474–2482.
Rojo F, J Tabernero, J Albanell, E Van Cutsem, A Ohtsu, T Doi, W Koizumi, K Shirao, H Takiuchi, S Ramon y Cajal, J Baselga (2006) Pharmacodynamic studies of gefitinib in tumor biopsy specimens from patients with advanced gastric carcinoma. J Clin Oncol 24(26): 4309–4316.
Rothenberg ML, B LaFleur, DE Levy, MK Washington, SL Morgan-Meadows, RK Ramanathan, JD Berlin, AB Benson 3rd, RJ Coffey (2005) Randomized phase Ii trial of the clinical and biological effects of two dose levels of gefitinib in patients with recurrent colorectal adenocarcinoma. J Clin Oncol 23(36): 9265–9274.
Siu LL, EK Rowinsky, LA Hammond, GR Weiss, M Hidalgo, GM Clark, J Moczygemba, L Choi, R Linnartz, NC Barbet, IT Sklenar, R Capdeville, G Gan, CW Porter, DD Von Hoff, SG Eckhardt (2002) A phase I and pharmacokinetic study of Sam486a, a novel polyamine biosynthesis inhibitor, administered on a daily-times-five every-three-week schedule in patients with advanced solid malignancies. Clin Cancer Res 8(7): 2157–2166.
Stearns V, A Gallagher, CG Kleer, B Singh, M Freedman, BR Haddad, C Isaacs, R Warren, M Brown, J Cullen, B Trock, DF Hayes (2004) A pilot study to establish a clinical model to perform phase Ii studies of breast cancer chemopreventive agents in women at high risk with biomarkers as surrogate endpoints for activity. Clin Cancer Res 10(24): 8332–8340.
Tabernero J, F Rojo, E Calvo, H Burris, I Judson, K Hazell, E Martinelli, S Ramon y Cajal, S Jones, L Vidal, N Shand, T Macarulla, FJ Ramos, S Dimitrijevic, U Zoellner, P Tang, M Stumm, HA Lane, D Lebwohl, J Baselga (2008) Dose- and schedule-dependent inhibition of the mammalian target of rapamycin pathway with everolimus: a phase I tumor pharmacodynamic study in patients with advanced solid tumors. J Clin Oncol 26(10): 1603–1610.
Thomas F, P Rochaix, A Benlyazid, J Sarini, M Rives, JL Lefebvre, BC Allal, F Courbon, E Chatelut, JP Delord (2007) Pilot study of neoadjuvant treatment with erlotinib in nonmetastatic head and neck squamous cell carcinoma. Clin Cancer Res 13(23): 7086–7092.
Ton NC, GJ Parker, A Jackson, S Mullamitha, GA Buonaccorsi, C Roberts, Y Watson, K Davies, S Cheung, L Hope, F Power, J Lawrance, J Valle, M Saunders, R Felix, JA Soranson, L Rolfe, K Zinkewich-Peotti, GC Jayson (2007) Phase I evaluation of Cdp791, a pegylated di-fab' conjugate that binds vascular endothelial growth factor receptor 2. Clin Cancer Res 13(23): 7113–7118.
Townsley CA, P Major, LL Siu, J Dancey, E Chen, GR Pond, T Nicklee, J Ho, D Hedley, M Tsao, MJ Moore, AM Oza (2006) Phase Ii study of erlotinib (Osi-774) in patients with metastatic colorectal cancer. Br J Cancer 94(8): 1136–1143.
Tse AN, DS Klimstra, M Gonen, M Shah, T Sheikh, R Sikorski, R Carvajal, J Mui, C Tipian, E O'Reilly, K Chung, R Maki, R Lefkowitz, K Brown, K Manova-Todorova, N Wu, MJ Egorin, D Kelsen, GK Schwartz (2008) A phase 1 dose-escalation study of irinotecan in combination with 17-allylamino-17-demethoxygeldanamycin in patients with solid tumors. Clin Cancer Res 14(20): 6704–6711.
Van Waes C, CT Allen, D Citrin, D Gius, AD Colevas, NA Harold, S Rudy, L Nottingham, C Muir, Z Chen, AK Singh, J Dancey, JC Morris (2010) Molecular and clinical responses in a pilot study of gefitinib with paclitaxel and radiation in locally advanced head-and-neck cancer. Int J Radiat Oncol Biol Phys 77(2): 447–454.
Watson AJ, MR Middleton, G McGown, M Thorncroft, M Ranson, P Hersey, G McArthur, ID Davis, D Thomson, J Beith, A Haydon, R Kefford, P Lorigan, P Mortimer, A Sabharwal, O Hayward, GP Margison (2009) O(6)-Methylguanine-DNA methyltransferase depletion and DNA damage in patients with melanoma treated with temozolomide alone or with lomeguatrib. Br J Cancer 100(8): 1250–1256.
Welch S, HW Hirte, MS Carey, SJ Hotte, MS Tsao, S Brown, GR Pond, JE Dancey, AM Oza (2007) Ucn-01 in combination with topotecan in patients with advanced recurrent ovarian cancer: a study of the princess margaret hospital phase Ii consortium. Gynecol Oncol 106(2): 305–310.
Weng DE, PA Masci, SF Radka, TE Jackson, PA Weiss, R Ganapathi, PJ Elson, WB Capra, VP Parker, JA Lockridge, JW Cowens, N Usman, EC Borden (2005) A phase I clinical trial of a ribozyme-based angiogenesis inhibitor targeting vascular endothelial growth factor receptor-1 for patients with refractory solid tumors. Mol Cancer Ther 4(6): 948–955.
Xiong HQ, R Herbst, SC Faria, C Scholz, D Davis, EF Jackson, T Madden, D McConkey, M Hicks, K Hess, CA Charnsangavej, JL Abbruzzese (2004) A phase I surrogate endpoint study of Su6668 in patients with solid tumors. Invest New Drugs 22(4): 459–466.
Zhang D, T Pier, DG McNeel, G Wilding, A Friedl (2007) Effects of a monoclonal anti-alphavbeta3 integrin antibody on blood vessels: a pharmacodynamic study. Invest New Drugs 25(1): 49–55.
Zhou EH, RJ Ellis, E Cherullo, V Colussi, F Xu, WD Chen, S Gupta, CC Whalen, D Bodner, MI Resnick, AA Rimm, SM Koroukian (2009) Radiotherapy and survival in prostate cancer patients: a population-based study. Int J Radiat Oncol Biol Phys 73(1): 15–23.
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Freeman, G., Kimmelman, J., Dancey, J. et al. Reporting practices of pharmacodynamic studies involving invasive research procedures in cancer trials. Br J Cancer 109, 897–908 (2013). https://doi.org/10.1038/bjc.2013.417
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2013.417
Keywords
This article is cited by
-
Variability in biopsy quality informs translational research applications in hepatocellular carcinoma
Scientific Reports (2021)
-
Safety and utility of image-guided research biopsies in relapsed high-grade serous ovarian carcinoma—experience of the BriTROC consortium
British Journal of Cancer (2017)