Abstract
Background:
Inflammation-based prognostic scores including the Glasgow Prognostic Score (GPS), neutrophil to lymphocyte ratio (NLR), and Prognostic Nutritional Index (PNI) are associated with survival in patients with hepatocellular carcinoma (HCC). The aim of this study was to investigate the prognostic value of these inflammation-based prognostic scores in patients with HCC.
Methods:
In total, 150 patients with newly diagnosed HCC were prospectively evaluated. Patients were divided according to the GPS, modified GPS, NLR, platelet to lymphocyte ratio (PLR), Prognostic Index (PI), and PNI. The area under the receiver operating characteristics curve (AUC) was calculated to compare the predictive ability of each of the scoring systems. A univariate and multivariate analysis were performed to identify the clinicopathological variables associated with overall survival.
Results:
The GPS consistently had a higher AUC value at 6 months (0.768), 12 months (0.787), and 24 months (0.758) in comparison with other inflammation-based prognostic scores. A multivariate analysis showed that the GPS was independently associated with overall survival.
Conclusion:
This study demonstrates that the GPS, an inflammation-based prognostic score, is an independent marker of poor prognosis in patients with HCC and is superior to the other inflammation-based prognostic scores in terms of prognostic ability.
Similar content being viewed by others
Main
Hepatocellular carcinoma (HCC) is the fifth most frequently diagnosed cancer worldwide, and the third leading cause of cancer-related deaths. An estimated 748 300 new liver cancer cases and 695 900 liver cancer-related deaths occurred in 2008, reflecting the poor prognosis of this disease (Jemal et al, 2011). In contrast to other cancers, the prognosis and treatment options for patients with HCC depend not only on the tumour progression but also on the extent of liver dysfunction (Huitzil-Melendez et al, 2010).
A number of staging systems have been proposed for HCC from independent groups, including the Barcelona Clinic Liver Cancer (Llovet et al, 1999), Cancer Liver Italian Programme (CLIP; CLIP investigators, 1998), and Japanese Integrated Staging (JIS) Score systems (Kudo et al, 2004). However, there is no worldwide consensus on which is the best system in staging and predicting the prognosis of patients with HCC.
In contrast, there is increasing evidence that the presence of a systemic inflammation response as evidenced by an elevated C-reactive protein (CRP) concentration, is associated with poor survival in patients with various malignancy, including HCC (Hashimoto et al, 2005; Kinoshita et al, 2012). Moreover, several studies have shown that inflammation-based prognostic scores including a combination of serum CRP and albumin as the Glasgow Prognostic Score (GPS), a combination of neutrophil and lymphocyte counts as the neutrophil to lymphocyte ratio (NLR), and a combination of albumin and lymphocyte counts as the Prognostic Nutritional Index (PNI) are associated with survival in patients with HCC (Gomez et al, 2008; Ishizuka et al, 2012; Pinato et al, 2012). Moreover, Smith et al (2009) have demonstrated that the platelte to lymphocyte ratio (PLR) is a significant prognostic marker in patients with pancreatic cancer, and Kasymjanova et al (2010) have shown that the Prognostic Index (PI) as evidenced by a combination of serum the CRP and white cell count is a significant prognostic marker in patients with lung cancer. Recently, in a Glasgow Inflammation Outcome Study, Proctor et al (2011a) compared the prognostic value of these inflammation-based prognostic scores (the modified GPS, NLR, PLR, PI, and PNI) in patients with a variety of cancers including ‘hepatopancreaticobiliary cancer’ and shown that modified GPS and PI have prognostic value in cancer independent of the tumour site. However, hepatopancreaticobiliary cancer includes pancreatic cancer and biliary tract cancer besides HCC in their study. Consequently, which inflammation-based prognostic scores is more suitable for predicting outcome in patients with HCC has not been fully elucidated.
Therefore, this study compared the prognostic value of these inflammation-based prognostic scores (the GPS, mGPS, NLR, PLR, PI, and PNI) in patients with HCC in various stages of disease and different liver functional status.
Materials and methods
Patients
In total, 208 patients with newly diagnosed HCC that had been treated at the Department of Gastroenterology and Hepatology, The Jikei University Daisan Hospital, between January 2005 and October 2011 were prospectively enroled. All medical records were reviewed retrospectively. Twenty-three patients were lost to follow-up. Thirty-five patients whose entire set of laboratory data were not available were excluded from this study. Patients who showed clinical evidence of infection or other inflammatory conditions were excluded. In total, 150 patients with HCC were finally included and evaluated. All patients were included in a previous study (Kinoshita et al, 2012).
The diagnosis of HCC was confirmed pathologically or based on imaging techniques obtained by 4-phase multidetector computed tomography (CT), or dynamic contrast-enhanced magnetic resonance imaging. Diagnosis should be based on the typical hallmark of HCC (hypervascular in the arterial phase with washout in the portal venous or delayed phases; European Association For The Study Of The Liver and European Organisation For Research And Treatment Of Cancer, 2012). Tumour-related variables such as maximal tumour diameter, tumour number, vascular invasion, and extra hepatic metastases were evaluated by these imaging techniques. The clinical stage (TNM classification) was determined according to the Liver Cancer Study Group of Japan (Minagawa et al, 2007).
This study complied with the standards of the Helsinki Declaration and current ethical guideline and was approved by the Institutional Ethical Board.
Inflammation-based prognostic scores and other variables
Blood samples were obtained before initial treatment for measurement of CRP, albumin, aspartate aminotransferase (AST), alanine aminotransferase (ALT), total bilirubin, white blood cell count, neutrophil, lymphocyte, platelet (Plt) count, prothrombin time, and α-fetoprotein level (AFP). Cancer Liver Italian Programme was calculated based on these variables and imaging techniques.
The GPS, mGPS, NLR, PLR, PI, and PNI were constructed as described in Table 1.
Treatment and patient’s follow-up
The indications for surgical resection were patients with solitary lesion, Child-Pugh grade A, no main portal vein trunk involvement, or distant metastasis. Radiofrequency ablation (RFA) or percutaneous ethanol injection was performed for patients with lesions <3 cm in size and <3 in number. Transcatehter arterial chemoembolisation (TACE) or lipiodol-transcatehter arterial infusion (TAI) was performed for patients with >4 multiple lesions or those >3 cm in size. Systemic chemotherapy or targeted therapy including sorafenib was performed for patients with distant metastasis and preserved liver function. Only the best supportive care (BSC) was given for patients with Child-Pugh grade C or distant metastasis.
Patients were followed carefully after the initial treatment. The serum AFP was measured once every month. US and dynamic CT were performed every 3 months. A selective hepatic arterial angiography or a percutaneous biopsy was performed in patients with suspected tumour recurrence. The start date of follow-up was the date of initial diagnosis of HCC. The end of follow-up was the time of last follow-up (October 2011) or death.
Statistical analysis
Continuous variables are presented as the median and range. Categorical variables are presented as the number and percentages. The overall survival rates were calculated using the Kaplan–Meier method, and differences in the survival rates between the groups were compared by the log-rank test. A receiver operating characteristics (ROC) curve was also generated and the area under the curve (AUC) was calculated to evaluate the discriminatory ability of each scoring systems. A univariate and multivariate analysis was performed for the prognostic factors using the Cox proportional hazard model. Variables that proved to be significant in the univariate analysis were tested subsequently with the multivariate Cox proportional hazard model. The forward selection method was used for multivariate Cox proportional analysis. A P-value <0.05 was considered to be significant. All statistical analysis was performed using the IBM SPSS Statistics software package v.19.0 (IBM SPSS Inc., Chicago, IL, USA).
Results
Patient characteristics
The baseline characteristics of the patients are shown in Table 2. The median age of the patients was 72 (range 43–91) years. One hundred and six (70.7%) patients were males and 44(29.3%) patients were females. Eighty-four (56%) patients were positive for antibodies to hepatitis C virus (anti-HCV), 20 (13.3%) patients were positive for hepatitis B surface antigen. One hundred and seven patients (71.3%) had preserved liver function (Child-Pugh A grade), and 78 patients (52%) were classified as stage I or II. Surgical resection was performed in 9 (6%) patients, TACE or RFA were administered in 134 (89.3%) patients. The remaining 7 (4.7%) patients received BSC.
Thirty-one (20.7%) patients had an elevated CRP level (>10 g l−1) and 58 (38.7%) patients had hypoalbuminemia (<35 g l−1). Twenty (13.3%) patients had both elevated CRP level and hypoalbuminemia. Eighty-one (54%) patients were allocated to GPS 0, 49 (32.7%) patients were allocated to GPS 1, and 20 (13.3%) patients were allocated to GPS 2, respectively. In contrast, 119 (79.3%) patients were allocated to mGPS 0, 11 (7.3%) patients were allocated to mGPS 1, and 20 (13.3%) patients were allocated to mGPS 2, respectively. Three (0.02%) patients had an elevated white cell count (>11 × 109 l−1), 5 (3.3%) patients an elevated neutrophil count (>7.5 × 109 l−1), 29 (19.3%) patients a lowered lymphocyte count (<1.0 × 109 l−1), and 2 (1.3%) patients an elevated Plt count (>400 × 103 l−1). Fifteen patients (10%) had NLR >5, 32 patients (21.3%) had PLR >150, and 78 patients (52%) had PNI <45.Thirty-three patients (22%) were allocated to PI1or 2.
Survival
The median duration of follow-up was 18 (range 1–80) months. Seventy-seven (51.3%) patients were alive at the end of the follow-up period, and 73 (48.7%) patients had died. The 1-year, 3-year, and 5-year overall survival rates were 74.1%, 53.3%, and 28.4%, respectively.
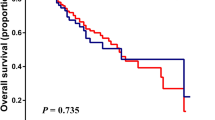
The relationship between the inflammation-based prognostic scores and overall survival is shown in Figures 1A–F. An elevated GPS, mGPS, NLR, PLR, PI, and PNI were associated with a reduced overall survival (all <0.05).
Receiver operating characteristic curves were constructed for survival status at 6-month, 12-month, and 24-month follow-up, and the area under the ROC curve (AUC) was compared (Table 3, Figures 2A–C) to assess the discrimination ability of each scoring system. The GPS consistently had a higher AUC value at 6 month (0.768), 12 month (0.787), and 24 month (0.758) in comparison with other inflammation-based prognostic scores.
Prognostic factors
The univariate analysis showed that AST (P=0.001), total serum bilirubin (P<0.0001), albumin (P<0.0001), pretreatment serum CRP level (P<0.0001), AFP (P<0.0001), Child-Pugh grade, CLIP (P<0.0001), TNM (P<0.0001), maximal tumour diameter (P<0.0001), multiple nodules (P<0.0001), vascular invasion (P<0.0001), extrahepatic metastasis (P=0.001), GPS (P<0.0001), mGPS (P<0.0001), NLR (P=0.01), PLR (P=0.001), PI (P<0.0001), and PNI (P<0.0001) were associated with overall survival (Table 4).
A multivariate analysis of these significant variables showed that only the GPS (HR 1.777, 95% CI 1.242–2.545, P=0.002) and CLIP (HR 2.246, 95% CI 1.786–2.824, P<0.0001) were independently associated with overall survival (Table 4).
Discussion
This study has demonstrated that the GPS, an inflammation-based prognostic score, is an independent marker of poor prognosis in patients with HCC and is superior to the mGPS, NLR, PLR, PI, and PNI in terms of prognostic ability.
The host inflammatory response has an important role in the development and progression of cancer (Mantovani et al, 2008). Inflammation promotes tumour angiogenesis, invasion, and metastasis through recruitment of regulatory T lymphocytes and chemokines, activation of interleukin-6 and tumour necrosis factor alpha, secretion of CRP, induction of neutrophilia, subversion of adaptive immune response, and aberration of response to hormones and chemotherapeutic agents (Heikkila et al, 2007; Mantovani et al, 2008; Wang et al, 2012).
Furthermore, the presence of an inflammatory response is proposed to be pathogenic in the development of cancer-associated malnutrition, resulting in poor performance status and increased mortality in patients with cancer (Argiles et al, 2003). This is of particular concern in patients with HCC, given the concomitant underlying illness and possible impaired nutritional status secondary to cirrhosis (Meng et al, 2010; Pinato et al, 2012).
These theoretical backgrounds have led to the proposal of several inflammation-based prognostic scores in patients with cancer over the last 10 years.
Several studies have shown that an elevated NLR is associated with poor prognosis in patients with HCC undergoing surgical resection (Gomez et al, 2008), transplantation (Halazun et al, 2009), transarterial chemoembolisation (Huang et al, 2011), and RFA (Chen et al, 2012). However, the cutoff points of NLR in these studies were different (2.4, 3.3, and 5) and non-optimal cutoff point has been determined. This study evaluated cutoff levels of NLR at 2.4, 3.3, and 5 and revealed that the NLR was not independently associated with survival at any of the cutoff levels (data not shown). Moreover, these studies did not compare the NLR to the GPS, mGPS, PNI, PLR, and PI.
Pinato et al (2012) demonstrated that the PNI is an independent predictor of poor overall survival in patients with HCC in various stages of the diseases and different liver functional status. However, their study did not compare the PNI with the GPS, mGPS, NLR, PLR, and PI.
The univariate analysis in this study demonstrated that the GPS, mGPS, NLR, PLR, PNI, and the PI were significantly associated with overall survival. However, the multivariate analysis showed that only the GPS was independently associated with overall survival. Moreover, the AUC analysis has shown that the GPS was superior to other inflammation-based prognostic scores in terms of predictive accuracy. These results confirm Ishizuka’s study demonstrating the predictive usefulness of the GPS on survival in patients with HCC after surgical resection (Ishizuka et al, 2012). In addition to their study, this study showed the superior prognostic ability of the GPS over the mGPS, NLR, PLR, PI, and PNI. This study is the first to show the GPS to be superior to other inflammation-based prognostic scores for the prediction of prognosis in patients with HCC.
A Glasgow Inflammation Outcome Study conducted by Proctor et al (2011a) showed that mGPS has prognostic value in cancer independent of the tumour site and was superior to other inflammation-based prognostic scores in terms of differentiating good from poor prognostic groups. Proctor et al (2011b) also indicated that mGPS is superior to the original GPS and has greater consistency and is of more use. Their observations were based on the results that a low albumin concentration alone was uncommon (<10% of all patients) and was not significantly associated with cancer-specific survival in many cancers including hepatopancreaticobiliary cancer (P=0.209). In contrast, this study included 38 (25.3%) patients with low albumin concentration alone and the serum albumin level is one of the components of the Child-Pugh classification. In fact, hypoalbuminemia is reported to as an independent poor prognostic factor in patients with HCC (Cho et al, 2008). Moreover, ‘hepatopancreaticobiliary cancer’ includes pancreatic cancer and biliary tract cancer besides HCC in a Glasgow Inflammation Outcome Study. Therefore, the GPS may be more suitable than mGPS for patients with HCC.
Impaired nutritional status and elevated levels of acute-phase plasma proteins have been associated with increased toxicity from chemotherapy. There is evidence from preclinical and clinical studies in cancer and other inflammatory diseases that disease-associated cytokines responsible for the hepatic acute-phase response may also reduce the expression and protein levels of a number of drug-metabolising enzymes and transporters, especially cytochrome P450 3A4. This results in increased toxicity during chemotherapy (Kasymjanova et al, 2010; Clarke et al, 2011). Accordingly, the GPS reflecting both the presence of the systemic inflammatory response and the progressive nutritional decline might provide substantial opportunities for clinicians to predict and reduce toxicities in HCC patients undergoing transarterial chemoembolisation or sorafenib treatment (Clarke et al, 2011). Further evaluation is required to confirm this hypothesis.
A potential limitation of this study is that is a retrospective, single-centre study. Therefore, a large-scale prospective validation study is needed to confirm the results.
In conclusion, our study has demonstrated that the GPS, an inflammation-based prognostic score, is an independent marker of poor prognosis in patients with HCC and is superior to the other inflammation-based prognostic scores in terms of prognostic ability.
Change history
18 August 2012
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Argiles JM, Busquets S, Lopez-Soriano FJ (2003) Cytokines in the pathogenesis of cancer cachexia. Curr Opin Clin Nutr Metab Care 6: 401–406
Chen TM, Lin CC, Huang PT, Wen CF (2012) Neutrophil-to-lymphocyte ratio associated with mortality in early hepatocellular carcinoma patients after radiofrequency ablation. J Gastroenterol Hepatol 27: 553–561
Cho YK, Chung JW, Kim JK, Ahn YS, Kim MY, Park YO, Kim WT, Byun JH (2008) Comparison of 7 staging systems for patients with hepatocellular carcinoma undergoing transarterial chemoembolization. Cancer 112: 352–361
Clarke SJ, Chua M, Moore M, Kao S, Phan V, Tan C, Charles K, McMillan DC (2011) Use of inflammatory markers to guide cancer treatment. Clin Pharmacol Ther 90: 475–478
European Association for the Study of The Liver and European Organisation for Research and Treatment of Cancer (2012) EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol 56: 908–943
Gomez D, Farid S, Malik HZ, Young AL, Toogood GJ, Lodge JP, Prasad KR (2008) Preoperative neutrophil-to-lymphocyte ratio as a prognostic predictor after curative resection for hepatocellular carcinoma. World J Surg 32: 1757–1762
Halazun KJ, Hardy MA, Rana AA, Woodland DC, Luyten EJ, Mahadev S, Witkowski P, Siegel AB, Brown RS, Emond JC (2009) Negative impact of neutrophil-lymphocyte ratio on outcome after liver transplantation for hepatocellular carcinoma. Ann Surg 250: 141–151
Hashimoto K, Ikeda Y, Korenaga D, Tanoue K, Hamatake M, Kawasaki K, Yamaoka T, Iwatani Y, Akazawa K, Takenaka K (2005) The impact of preoperative serum C-reactive protein on the prognosis of patients with hepatocellular carcinoma. Cancer 103: 1856–1864
Heikkila K, Ebrahim S, Lawlor DA (2007) A systemic review of the association between circulating concentrations of C reactive protein and cancer. J Epidemiol Community Health 61: 824–833
Huang ZL, Luo J, Chen MS, Li JQ, Shi M (2011) Blood neutrophil-to-lymphocyte ratio predicts survival in patients with unresectable hepatocellular carcinoma undergoing transarterial chemoembolization. J Vasc Interv Radiol 22: 702–709
Huitzil-Melendez FD, Capanu M, O’Reilly EM, Duffy A, Gansukh B, Saltz LL, Abou-Alfa GK (2010) Advanced hepatocellular carcinoma: which staging systems best predict prognosis? J Clin Oncol 28: 2889–2895
Ishizuka M, Kubota K, Kita J, Shimoda M, Kato M, Sawada T (2012) Impact of an inflammation-based prognostic system on patients undergoing surgery for hepatocellular carcinoma: a retrospective study of 398 Japanese patients. Am J Surg 203: 101–106
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61: 69–90
Kasymjanova G, MacDonald N, Agulnik JS, Cohen V, Pepe C, Kreisman H, Sharma R, Small D (2010) The predictive value of pre-treatment inflammatory markers in advanced non-small-cell lung cancer. Curr Oncol 17: 52–58
Kinoshita A, Onoda H, Takano K, Imai N, Saeki C, Fushiya N, Miyakawa Y, Nishino H, Tajiri H (2012) Pretreatment serum C-reactive protein level predicts poor prognosis in patients with hepatocellular carcinoma. Med Oncol; doi:10.1007/s12032-012-0220-1(in press)
Kudo M, Chung H, Haji S, Osaki Y, Oka H, Seki T, Kasugai H, Sasaki Y, Matsunaga T (2004) Validation of a new prognostic staging system for hepatocellular carcinoma: the JIS score compared with the CLIP score. Hepatology 40: 1396–1405
Llovet JM, Bru C, Bruix J (1999) Prognosis of hepatocellular carcinoma: the BCLC staging classification. Semin Liver Dis 19: 329–338
Mantovani A, Allavena P, Sica A, Balkwill F (2008) Cancer-related inflammation. Nature 454: 436–444
Meng QH, Yu HW, Li J, Wang JH, Ni MM, Feng YM, Jin Y, Hou W, Yu SQ, Wang X, Liu Y (2010) Inadequate nutritional intake and protein-energy malnutrition involved in acute and chronic viral hepatitis Chinese patients especially in cirrhosis patients. Hepatogastroenterology 57: 845–851
Minagawa M, Ikai I, Matsuyama Y, Yamaoka Y, Makuuchi M (2007) Staging of hepatocellular carcinoma: assessment of the Japanese TNM and AJCC/UICC TNM systems in a cohort of 13,772 patients in Japan. Ann Surg 245: 909–922
Pinato DJ, North BV, Sharma R (2012) A novel, externally validated inflammation-based prognostic algorithm in hepatocellular carcinoma: the prognostic nutritional index (PNI). Br J Cancer 106: 1439–1445
Proctor MJ, Morrison DS, Talwar D, Balmer SM, Fletcher CD, O’Reilly DS, Foulis AK, Horgan PG, McMillan DC (2011a) A comparison of inflammation-based prognostic scores in patients with cancer. A Glasgow Inflammation Outcome Study. Eur J Cancer 47: 2633–2641
Proctor MJ, Morrison DS, Talwar D, Balmer SM, O’Reilly DS, Foulis AK, Horgan PG, McMillan DC (2011b) An inflammation-based prognostic score (mGPS) predicts cancer survival independent of tumour site: a Glasgow Inflammation Outcome Study. Br J Cancer 104: 726–734
Smith RA, Bosonnet L, Raraty M, Sutton R, Neoptolemos JP, Campbell F, Ghaneh P (2009) Preoperative platelet-lymphocyte ratio is an independent significant prognostic marker in resected pancreatic ductal adenocarcinoma. Am J Surg 197: 466–472
The Cancer of the Liver Italian Program (CLIP) Investigators (1998) A new prognostic system for hepatocellular carcinoma: a retrospective study of 435 patients. Hepatology 28: 751–755
Wang DS, Luo HY, Qiu MZ, Wang ZQ, Zhang DS, Wang FH, Li YH, Xu RH (2012) Comparison of the prognostic values of various inflammation based factors in patients with pancreatic cancer. Med Oncol doi:10.1007/s12032-012-0226-8(in press)
Author information
Authors and Affiliations
Corresponding author
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Kinoshita, A., Onoda, H., Imai, N. et al. Comparison of the prognostic value of inflammation-based prognostic scores in patients with hepatocellular carcinoma. Br J Cancer 107, 988–993 (2012). https://doi.org/10.1038/bjc.2012.354
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2012.354
Keywords
This article is cited by
-
Presepsin is a more useful predictor of septic AKI and ARDS for very-old sepsis patients than for young sepsis patients in ICUs: a pilot study
BMC Research Notes (2024)
-
RNF31 promotes proliferation and invasion of hepatocellular carcinoma via nuclear factor kappaB activation
Scientific Reports (2024)
-
The impact of sarcopenia on esophagectomy for cancer: a systematic review and meta-analysis
BMC Surgery (2023)
-
C-reactive protein/albumin ratio is the most significant inflammatory marker in unresectable pancreatic cancer treated with FOLFIRINOX or gemcitabine plus nab-paclitaxel
Scientific Reports (2023)
-
Phytochemical profiling and cytotoxic potential of Arnebia nobilis root extracts against hepatocellular carcinoma using in-vitro and in-silico approaches
Scientific Reports (2023)