Abstract
The aim of this cohort study was to compare a condensed schedule of consolidation therapy with high-dose cytarabine on days 1, 2 and 3 (HDAC-123) with the HDAC schedule given on days 1, 3 and 5 (HDAC-135) as well as to evaluate the prophylactic use of pegfilgrastim after chemotherapy in younger patients with acute myeloid leukemia in first complete remission. One hundred and seventy-six patients were treated with HDAC-135 and 392 patients with HDAC-123 with prophylactic pegfilgrastim at days 10 and 8, respectively, in the AMLSG 07-04 and the German AML Intergroup protocol. Time from start to chemotherapy until hematologic recovery with white blood cells >1.0 G/l and neutrophils >0.5 G/l was in median 4 days shorter in patients receiving HDAC-123 compared with HDAC-135 (P<0.0001, each), and further reduced by 2 days (P<0.0001) by pegfilgrastim. Rates of infections were reduced by HDAC-123 (P<0.0001) and pegfilgrastim (P=0.002). Days in hospital and platelet transfusions were significantly reduced by HDAC-123 compared with HDAC-135. Survival was neither affected by HDAC-123 versus HDAC-135 nor by pegfilgrastim. In conclusion, consolidation therapy with HDAC-123 leads to faster hematologic recovery and less infections, platelet transfusions as well as days in hospital without affecting survival.
Similar content being viewed by others
Introduction
Repetitive cycles of higher doses of cytarabine 0.5–3 g/m2 administered over 3 to 6 days have been widely used for conventional intensive consolidation therapy of acute myeloid leukemia with remaining open questions, for example, most effective schedule, dosage and number of cycles.1, 2, 3 The landmark study conducted by the Cancer and Leukemia Group B (CALGB) established the current standard for patients aged 60 years and younger.4 In this prospective upfront randomized study, four cycles of high-dose cytarabine (HDAC) (3 g/m2, two times a day, days 1, 3 and 5) were superior to intermediate- (400 mg/m2 cont. days 1–5) or standard-dose cytarabine (100 mg/m2 cont. days 1–5) with respect to relapse-free survival (RFS) and overall survival (OS).4 Several alternative intensive combination chemotherapy regimens have been evaluated in randomized trials in the past years, but still single agent HDAC (3 g/m2, b.i.d., days 1, 3 and 5) remains the standard in younger adults with low- and intermediate-risk acute myeloid leukemia (AML), whereas combination postremission therapy may be considered in high-risk patients.5, 6
Chemotherapy-induced neutropenia represents a major risk factor for infection-related morbidity and mortality during AML treatment with an incidence of neutropenic fever after a consolidation therapy reported of 50 to 90%.7, 8, 9, 10, 11 The meta-analysis performed by Smith et al.10 revealed that the use of prophylactic granulocyte colony-stimulating factor (G-CSF) after induction and consolidation chemotherapy for patients in complete remission (CR) decreases the incidence of infections and diminishes the likelihood of hospitalizations during intensive consolidation therapy. Similar results were obtained in a meta-analysis published in 2007 by Sung et al.,12 in which 148 randomized clinical trials were evaluated including patients with solid and hematologic malignancies treated with chemotherapy or hematopoietic cell transplantation (HCT). In the subgroup analysis focused on AML, the risk of microbiologically documented infections (hazard ratio (HR): 0.86; confidence interval (CI): 0.77–0.96) and febrile neutropenia (HR: 0.71; CI: 0.63–0.80) were found to be lower in the group randomized to G-CSF.12 However, the infection-related mortality was not significantly different with and without G-CSF (P=0.44). Different results came from a systematic review published by Gurion et al.,13 in which 19 randomized clinical trials were examined. They found no survival benefit and no decrease in infection rates in the group of patients treated with G-CSF after chemotherapy. In consequence, the last actualization of the AML-NCCN guidelines recommended the use of G-CSF in AML only within clinical trials.8 Pegfilgrastim is the pegylated formulation of G-CSF that allows one-time administration compared with daily administration with filgrastim. In a randomized phase-2 trial Sierra et al.14 found no clinically meaningful difference between a single dose of pegfilgrastim and daily dosing of filgrastim in terms of duration of severe neutropenia after induction and consolidation therapy.
The main objectives of our study were to analyze the effect of a condensed regimen of HDAC, 3 g/m2 administered every 12 h on days 1, 2 and 3 (HDAC-123), compared with the commonly used regimen of HDAC, 3 g/m2 every 12 h on days 1, 3 and 5 (HDAC-135), as well as to evaluate the effect of pegfilgrastim in the consolidation therapy of younger adult patients with AML with regard to hematologic reconstitution, infectious complications, supportive care and days in hospital.
Patients and methods
Patients
Patients aged between 18 and 60 years with newly diagnosed AML including de novo AML, secondary AML (sAML) with a preceding history of myelodysplastic or myeloproliferative disorder, and therapy-related AML (tAML) following treatment of a primary malignancy, as defined by the World Health Organization 2001 classification15 were eligible for the trials. Patients with acute promyelocytic leukemia as well as patients with concomitant renal (creatinine>1.5xupper normal serum level), liver (bilirubin, aspartate aminotransferase or alkaline phosphatase>2 x upper normal serum level) or cardiac dysfunction (New York Heart Association III/IV), uncontrolled infectious disease, primary coagulation disturbance, performance status (Eastern Cooperative Oncology Group) >2 or active concomitant malignant disease were excluded.
Study design
Between 2004 and 2009 patients were upfront randomized 1:10 between the standard German Intergroup-arm16 and the AMLSG 07-04 study.17 Patients randomized into the AMLSG 07-04 protocol were again randomized between 2004 and 2006 in a 2 × 2 factorial design to receive intensive induction chemotherapy with or without all-trans retinoic acid (ATRA) and with or without valproic acid (VPA).17 In 2006 the AMLSG 07-04 study was amended and randomization for VPA was terminated based on excessive hematologic toxicity of VPA in combination with chemotherapy similarly noted in older patients.18 Induction therapy consisted of ICE (idarubicin, 12 mg/m2 intravenously, days 1, 3 and 5; cytarabine, 100 mg/m2 cont. intravenously, days 1–7; etoposide 100 mg/m2 intravenously, days 1–3). Patients achieving a CR or partial remission after the first induction received a second cycle according to their initial randomization with a reduced dosage of idarubicin (12 mg/m2, days 1 and 3). Patients with high-risk AML defined either by high-risk cytogenetics or induction failure19 were assigned to receive allogeneic HCT from a matched-related or -unrelated donor. If a matched-related donor was available, allogeneic HCT was intended in first CR in all patients except those with core-binding factor AML. Starting in December 2006, AML patients exhibiting a FLT3 internal tandem duplication were also categorized as high risk. All other patients were assigned to three cycles of consolidation chemotherapy with HDAC. From August 2004 to November 2006 patients were treated with HDAC-135 and pegfilgrastim on day 10 (cohort 1). From November 2006 on, patients were treated with the condensed schedule HDAC-123 and pegfilgrastim on day 8 (cohort 2). Patients randomized to the German AML Intergroup-arm16 received HDAC-135 without prophylactic growth factor support.
Cyto- and molecular genetics
Chromosome banding analysis was performed centrally in the AMLSG Laboratory for Cytogenetic and Molecular Diagnosis. Karyotypes were designated according to the International System for Human Cytogenetic Nomenclature.20 Leukemia samples were analyzed for mutations in FLT3 (FLT3 internal tandem duplication, FLT3 tyrosine kinase domain mutations at codons D835/I836) CEBPA and NPM1 as described previously.21
Clinical end points
Hematologic recovery was determined in each patient in every cycle of consolidation therapy. Hematologic recovery was defined as absolute white blood cell (WBC) count ⩾1.0 × 109/l, absolute neutrophil count of ⩾0.5 × 109/l and a platelet count ⩾20 × 109/l. Time to WBC, neutrophil and platelet recovery was defined as the duration of days from the first day of chemotherapy of each cycle until the first day of achievement of the above defined cutoff hematologic recovery definitions. Platelet recovery times were not documented in the German AML Intergroup-arm.16 The data on the number of units of packed red blood cells and platelets were collected in each therapy cycle. One human leukocyte antigen class-I-compatible single donor platelet unit was considered equal to four platelet units from random donors. Infection was defined as microbiologically documented infection and/or febrile neutropenia. The duration of hospitalization was defined as the time from first day of therapy until discharge. Secondary outcomes were overall survival (OS), RFS and cumulative incidence of relapse and death. These were defined as recommended.1, 2
Statistical analysis
Type of cytarabine regimen (HDAC-123 versus HDAC-135) according to upfront randomization (German AML Intergroup) and cohort assignment (AMLSG 07-04 study; cohorts 1 and 2) as well as administration of pegfilgrastim within the 07-04 study (yes or no) were analyzed.
Comparisons between patient subgroups were performed by the Mann–Whitney or Kruskal–Wallis test for continuous variables and by Fisher’s exact test for categorical variables. Differences between cumulative incidences of hematological recovery times (WBC, neutrophils) were calculated using the method described by Gray.22 Survival curves were compared using the log-rank test. Multivariable analysis for the endpoint WBC recovery was performed using an extended Cox regression model by application of the method of Wei, Lin and Weissfeld method with respect to repeated WBC recovery within all applied consolidation cycles.23 Similarly, a conditional logistic regression model was used to analyze infectious complications during consolidation therapy with stratification for the cycles in patients receiving three consolidation cycles.24 All statistical analyses were performed with the statistical software environment R, version 3.0.1, using the R packages rms, version 3.3-1, and cmprsk, version 2.2-2.25
Results
Patients and baseline characteristics
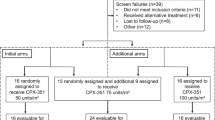
According to upfront randomization into the German AML Intergroup protocol, 41 patients were assigned to at least one standard consolidation therapy with HDAC-135 and 527 patients received at least one consolidation therapy within the AMLSG 07-04 protocol (Figure 1), with 135 patients (cohort 1) assigned to HDAC-135 (between August 2004 and December 2006) and 392 patients (cohort 2) to HDAC-123 (from January 2006 to August 2009). There were no significant differences in patient characteristics at diagnosis between the three groups (Table 1).
Influence of HDAC schedules on time to WBC and neutrophil recovery
Within the AMLSG 07-04 protocol initial upfront randomization into four treatment arms (standard, ATRA, VPA, VPA-ATRA) was stopped for VPA in July 2006 due to excessive hematotoxicity.18 In an as-treated analysis we found no influence of VPA or ATRA in the hematologic recovery end points of cohort 1 treated with HDAC-135 (neutrophils, P=0.78; WBC, P=0.49; platelets, P=0.67) and of ATRA in cohort 2 treated with HDAC-123 (neutrophils, P=0.65; WBC, P=0.40, platelets, P=0.30). Therefore, patients were grouped for further analysis into cohorts 1 and 2 for as described above.
Consistently for all three consolidation cycles, shorter median recovery times were identified for WBC in HDAC-123 by an average 4 days compared with HDAC-135 and the German AML Intergroup (P=0.0008, P=0.0003, P=0.001, respectively; Table 2). Time to neutrophil recovery was also shorter in all three consolidation cycles in HDAC-123 compared with HDAC-135 and the German AML Intergroup by ~4 days, with a significant difference in the first and second consolidation cycle (Table 2). Exemplarily, WBC and neutrophil recovery is illustrated after the first consolidation cycle according to the assigned treatment group in Figure 2. In a subgroup analysis, we compared the upfront randomized patients between the German AML Intergroup and the AMLSG 07-04 study both assigned to receive HDAC-135 with the intended prophylactic use of pegfilgrastim at day 10 in the AMLSG 07-04 study compared with no prophylactic growth factor support in the German AML Intergroup study. This analysis revealed a reduction of WBCs and neutrophil median recovery times by one to two days in all three consolidation cycles without reaching statistical significance (Table 2). Three hundred and eighty-one patients received all three consolidation cycles; median time intervals between cycles 1 and 2, as well as cycles 2 and 3 were 40 days (range, 28–140 days) and 41 days (range, 26–129 days). Interestingly, we observed no cumulative hematologic toxicity with increasing number of applied consolidation cycles, when the median time to hematologic recovery was compared in patients receiving all three cycles in terms of WBC and neutrophil recovery for HDAC-135 (P=0.26, P=0.90, respectively; n=97), HDAC-123 (P=0.17, P=0.61, respectively; n=258) and the German AML Intergroup-arm (P=0.78, P=0.74, respectively; n=26).
Influence of pegfilgrastim on time to WBC, neutrophil and platelet recovery
Within the AMLSG 07-04 study pegfilgrastim 6 mg subcutaneously was intended to be applied at day 10 in the HDAC-135 and at day 8 in the HDAC-123 schedule. In an as-treated analysis, patients receiving pegfilgrastim were compared with those not receiving pegfilgrastim (Table 3). The as-treated multivariable analysis including all consolidation cycles administered within the 07-04 study stratified by consolidation cycle showed an overall reduction of duration of leukopenia by 3 days in patients treated with pegfilgrastim in both chemotherapy schedules (P<0.0001, each). We also found a reduction in the duration of neutropenia by 5 and 3 days for HDAC-135 and HDAC-123 (P=0.03 and P=0.003, respectively) in patients treated with pegfilgrastim without impact on duration of thrombocytopenia (P=0.77 and P=0.70, respectively).
Multivariable Wei–Lin–Weissfeld model on WBC recovery
Multivariable analysis based on the Wei–Lin–Weissfeld model for recurring events integrating all applied consolidation cycles including all patients receiving three consolidation cycles within the AMLSG 07-04 study stratified by consolidation cycle revealed that HDAC-123 (HR, 1.94; P<0.0001) and treatment with pegfilgrastim (HR, 1.58; P<0.0001) were significantly associated with shorter WBC recovery, whereas older age was associated with longer WBC recovery (HR of a 10-year age difference, 0.89; P=0.001). Factors without significant impact were gender, risk group according to 2010-European LeukemiaNet recommendations and type of AML (de novo versus secondary/treatment related AML) (Table 4).
Infection rates, days of hospitalization and number of platelet transfusions with HDAC-135 and -123
The overall infectious complications including infection with clinical focus (mostly pneumonia) and febrile neutropenia were 37.3%, 40.0% and 41.3% in the three consecutive consolidation cycles, respectively. Overall, the infectious complication rates were highest in the Intergroup-arm after HDAC-135 without prophylactic growth factor support ranging from 74% to 83% and lowest in the HDAC-123 schedule of the AMLSG 07-04 protocol with administration of prophylactic pegfilgrastim ranging from 30 to 36% (Table 5). We found that 59% of patients, who had an infection during the first consolidation cycle, developed an infection in the second consolidation. In contrast, in patients who did not experience an infection in the first consolidation cycle, only 28% had an infection in the second consolidation (odds ratio (OR), 3.80; 95% CI: 2.40–6.06; P<0.0001). Nearly the same was true if infections in consolidation cycles 2 and 3 were analyzed (OR, 4.48; 95% CI: 2.83–7.18; P<0.0001). A conditional logistic regression model based on all consolidation cycles and stratified for them revealed that HDAC-123 (OR, 0.58; P<0.0001) and the administration of pegfilgrastim (OR, 0.68; P=0.002) were associated with a reduction in infection rates, whereas patients with a secondary AML had a higher risk of infections (OR, 1.62; P=0.05). Age, gender and 2010-European LeukemiaNet risk category had no significant impact (Table 6). The lower rate of infections and shorter hematologic recovery times probably led to significantly shortened time in hospital with the HDAC-123 schedule of the AMLSG 07-04 protocol with the administration of pegfilgrastim (Table 5). This was mainly due to a substantial proportion of patients being discharged within 10 days of 28%, 39% and 40% after consolidation cycles 1, 2 and 3, respectively. About one-third of these early discharged patients were readmitted because of infections necessitating intravenous antibiotic treatment. None of the early discharged patients died owing to infections during this phase. The need of platelet transfusions was markedly reduced in the HDAC-123 schedule from median 8 units in the HDAC-135 schedule to median 4 units in the HDAC-123 schedule (P<0.0001, Table 5). As expected, the platelet transfusion needs were not affected by the administration of pegfilgrastim (Table 5).
Survival analyses
For survival analyses only patients treated in the AMLSG 07-04 study were included. There was no difference (P=0.90) between HDAC-135 (n=135) and HDAC-123 (n=392) in terms of OS. The same was true for RFS (P=0.48) and RFS censored at the date of transplant for patients receiving an allogeneic HCT in first CR (P=0.78). In addition, there was no difference in cumulative incidence of relapse and cumulative incidence of death again censoring of patients receiving an allo-HCT in first CR at the date of transplant between the two HDAC schedules (P=0.75 and P=0.10, respectively). In the subgroup receiving all three consolidation cycles, again no difference between the two schedules was evident.
Discussion
In our prospective study we compared two different schedules of HDAC; the commonly used regimen with cytarabine administered on days 1, 3 and 5 as well as a condensed regimen with cytarabine administered on days 1 to 3. We were able to show a significantly and clinically meaningful reduction by 4 days of time to WBC and neutrophil recovery with the HDAC-123 compared with the HDAC-135 regimen. In addition, this significant reduction in WBC and neutrophil recovery with HDAC-123 was associated with a significantly lower rate of infections, fewer platelet transfusions and fewer days in hospital. In an upfront randomized comparison, we were not able to show a difference between the German AML Intergroup protocol with HDAC-135 without use of pegfilgrastim compared with HDAC-135 within the AMLSG 07-04 protocol with the intended use of pegfilgrastim in terms of WBC and neutrophil recovery, which may be related to the limited protocol adherence with only 68% of the patients receiving the intended pegfilgrastim. In contrast, a per-protocol analysis of the whole cohort revealed that pegfilgrastim was effective in reducing the time to WBC and neutrophil recovery by 3 days in the HDAC-135 and by 2 days in the HDAC-123 schedule, which is in accordance with previous randomized studies demonstrating a significant reduction in the duration of severe neutropenia.11, 12, 13 These observations were supported by multivariable analysis in which HDAC-123 as well as the administration of pegfilgrastim were significantly associated with shorter WBC recovery times. This finding was paralleled by the results of the univariable and multivariable analysis with the endpoint infections including fever in neutropenia and infections with clinical focus. Consistently, we observed that both HDAC-123 and the administration of pegfilgrastim were associated with a significant reduction in the rate of infectious complications. These results are concordant with the findings of a systematic meta-analysis12 showing that the use of G-CSF during consolidation therapy leads to a reduction in the duration of neutropenia and decreases the rate of infections as well as febrile neutropenia.
Our study also shows that there was no cumulative hematologic toxicity with the repetitive use of HDAC-123 and HDAC-135, with similar median WBC and neutrophil recovery times following the three cycles of consolidation therapy. This argues against a hematopoietic stem cell damage induced by repetitive cycles of HDAC.26 However, our multivariable analyses revealed that WBC recovery times tended to be longer with increasing age. This correlation may reflect hematopoietic stem cell aging described in in vivo and in vitro studies, where older stem cells have been found to be less effective in contributing to hematopoiesis.27
Importantly, no difference in any survival endpoint analyzed was found between HDAC-123 and HDAC-135 in the AMLSG 07-04 study, providing evidence for at least equivalent efficacy of both schedules. One important limitation of our study is the sequential cohort design rather than an upfront randomized study. Therefore, our results have to be interpreted with caution. However, the overall favorable results of the AMLSG 07-04 study with an OS after 4 years of 54% (95% CI: 50–58) also would argue against inferiority of the HDAC-123 schedule.17 Thus, our results favor the use of the condensed HDAC-123 schedule combined with prophylactic pegfilgrastim at day 8 in the consolidation therapy of younger adult patients with AML.
In conclusion, data from our study suggest that a condensed schedule of HDAC on days 1, 2 and 3 for consolidation therapy in younger adult patients with AML appears to be the preferred treatment schedule, resulting in faster hematologic recovery, lower infection rate as well as fewer platelet transfusions and days in hospital without affecting the RFS and OS rates. The administration of pegfilgrastim may further reduce rate of infections and duration of hospitalization.
References
Döhner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Buchner T et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 2016; 129: 424–447.
Döhner H, Estey EH, Amadori S, Appelbaum FR, Buchner T, Burnett AK et al. Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European Leukemia. Net Blood 2010; 115: 453–474.
Schlenk RF . Is there justification for 4 cycles of consolidation therapy in AML? Best Pract Res Clin Haematol 2016; 29: 341–344.
Mayer RJ, Davis RB, Schiffer CA, Berg DT, Powell BL, Schulman P et al. Intensive postremission chemotherapy in adults with acute myeloid leukemia. Cancer and Leukemia Group B. N Engl J Med 1994; 331: 896–903.
Dombret H, Gardin C . An update of current treatments for adult acute myeloid leukemia. Blood 2016; 127: 53–61.
Schlenk RF . Post-remission therapy for acute myeloid leukemia. Haematologica 2014; 99: 1663–1670.
Heil G, Hoelzer D, Sanz MA, Lechner K, Liu Yin JA, Papa G et al. A randomized, double-blind, placebo-controlled, phase III study of filgrastim in remission induction and consolidation therapy for adults with de novo acute myeloid leukemia. The International Acute Myeloid Leukemia Study Group. Blood 1997; 90: 4710–4718.
O'Donnell MR, Abboud CN, Altman J, Appelbaum FR, Arber DA, Attar E et al. Acute myeloid leukemia. J Natl Compr Canc Netw 2012; 10: 984–1021.
Ottmann OG, Bug G, Krauter J . Current status of growth factors in the treatment of acute myeloid and lymphoblastic leukemia. Semin Hematol 2007; 44: 183–192.
Smith TJ, Khatcheressian J, Lyman GH, Ozer H, Armitage JO, Balducci L et al. 2006 update of recommendations for the use of white blood cell growth factors: an evidence-based clinical practice guideline. J Clin Oncol 2006; 24: 3187–3205.
Archimbaud E, Ottmann OG, Yin JA, Lechner K, Dombret H, Sanz MA et al. A randomized, double-blind, placebo-controlled study with pegylated recombinant human megakaryocyte growth and development factor (PEG-rHuMGDF) as an adjunct to chemotherapy for adults with de novo acute myeloid leukemia. Blood 1999; 94: 3694–3701.
Sung L, Nathan PC, Alibhai SM, Tomlinson GA, Beyene J . Meta-analysis: effect of prophylactic hematopoietic colony-stimulating factors on mortality and outcomes of infection. Ann Intern Med 2007; 147: 400–411.
Gurion R, Belnik-Plitman Y, Gafter-Gvili A, Paul M, Vidal L, Ben-Bassat I et al. Colony-stimulating factors for prevention and treatment of infectious complications in patients with acute myelogenous leukemia. Cochrane Database Syst Rev 2012 (6): CD008238.
Sierra J, Szer J, Kassis J, Herrmann R, Lazzarino M, Thomas X et al. A single dose of pegfilgrastim compared with daily filgrastim for supporting neutrophil recovery in patients treated for low-to-intermediate risk acute myeloid leukemia: results from a randomized, double-blind, phase 2 trial. BMC Cancer 2008; 8: 195.
Jaffe ES, Harris NL, Stein H, Vardiman JW Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues, 3rd edn. IARC Press: Lyon, France, 2001.
Büchner T, Schlenk RF, Schaich M, Dohner K, Krahl R, Krauter J et al. Acute myeloid leukemia (AML): different treatment strategies versus a common standard arm-combined prospective analysis by the German AML Intergroup. J Clin Oncol 2012; 30: 3604–3610.
Schlenk RF, Lubbert M, Benner A, Lamparter A, Krauter J, Herr W et al. All-trans retinoic acid as adjunct to intensive treatment in younger adult patients with acute myeloid leukemia: results of the randomized AMLSG 07-04 study. Ann Hematol 2016; 95: 1931–1942.
Tassara M, Dohner K, Brossart P, Held G, Gotze K, Horst HA et al. Valproic acid in combination with all-trans retinoic acid and intensive therapy for acute myeloid leukemia in older patients. Blood 2014; 123: 4027–4036.
Schlenk RF, Dohner K, Mack S, Stoppel M, Kiraly F, Gotze K et al. Prospective evaluation of allogeneic hematopoietic stem-cell transplantation from matched related and matched unrelated donors in younger adults with high-risk acute myeloid leukemia: German–Austrian trial AMLHD98A. J Clin Oncol 2010; 28: 4642–4648.
Mitelman F . An International System for Human Cytogenetic Nomenclature, ISCN edn. Karger: Basel, Switzerland, 1995.
Schlenk RF, Dohner K, Krauter J, Frohling S, Corbacioglu A, Bullinger L et al. Mutations and treatment outcome in cytogenetically normal acute myeloid leukemia. N Engl J Med 2008; 358: 1909–191.
Gray RJ . A class of k-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat 1988; 16: 1141–1154.
Harrell FE . Regression Modeling Strategies: With Applications to Linear Models, Logistic Regression, and Survival Analysis. Springer: New York, NY, 2001.
Gail MH, Lubin JH, Rubinstein LV . Likelihood calculations for matched case-control studies and survival studies with tied death times. Biometrika 1980; 68: 703–707.
R Development Core Team R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, 2009.
Jung J, Buisman S, de Haan G . Hematopoiesis during development, aging, and disease. Exp Hematol 2016; 44: 689–695.
Morrison SJ, Wandycz AM, Akashi K, Globerson A, Weissman IL . The aging of hematopoietic stem cells. Nat Med 1996; 2: 1011–1016.
Acknowledgements
This work was supported by Grants 01GI9981 (Network of Competence Acute and Chronic Leukemias) and 01KG0605 (IPD-Meta-Analysis: A model-based hierarchical prognostic system for adult patients with acute myeloid leukemia (AML)) from the German Bundesministerium für Bildung und Forschung (BMBF), the Deutsche José Carreras Leukämie-Stiftung (DJCLS H 05/02) and an unrestricted grant from Pfizer and Amgen. We are also grateful to all members of the German-Austrian AML Study Group (AMLSG) for providing leukemia specimens and clinical data; a list of AMLSG institutions and investigators participating in this study appears in the Supplementary Appendix.
Author contributions
Conception and design: RFS, SJ, HD. Provision of study materials or patients: JK, HM, TK, MB, HRS, GH, C-HK, KG, ML, AK, PB, MW, HS, BH, DN, GW, H-AH, HK, WF, AR, GR, SK, EK, VR, GH, AG, HD, RFS. Collection and assembly of data: SJ, DW, RFS. Data analysis and interpretation: SJ and RFS. Manuscript writing: SJ, RFS, HD. Final approval of manuscript: SJ, AB, JK, HM, TK, MB, HRS, GH, C-HK, KG, ML, AK, PB, MW, HS, BH, DN, GW, H-AH, HK, WF, AR, GR, SK, EK, VR, GH, DW, BS, KD, AG, HD, RFS.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Blood Cancer Journal website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Jaramillo, S., Benner, A., Krauter, J. et al. Condensed versus standard schedule of high-dose cytarabine consolidation therapy with pegfilgrastim growth factor support in acute myeloid leukemia. Blood Cancer J. 7, e564 (2017). https://doi.org/10.1038/bcj.2017.45
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bcj.2017.45
This article is cited by
-
Efficacy of delayed pegfilgrastim administration following consolidation therapy with high-dose cytarabine (HiDAC) in acute myeloid leukemia (AML) patients
Supportive Care in Cancer (2024)
-
Cost comparison of post-remission strategies in younger and older AML patients in France
Blood Cancer Journal (2023)
-
Digital twins in oncology
Journal of Cancer Research and Clinical Oncology (2023)
-
AML consolidation therapy: timing matters
Journal of Cancer Research and Clinical Oncology (2023)
-
Outcomes of HIDAC 18 g Versus IDAC 9 g in Consolidation Therapy of Acute Myeloid Leukemia: A Retrospective Study
Indian Journal of Hematology and Blood Transfusion (2022)