Abstract
Aim:
Tissue transglutaminase (tTG) catalyzes proteins, including β-amyloid (Aβ), to cross-link as a γ-glutamyl-ε-lysine structure isopeptide, which is highly resistant to proteolysis. Thus, tTG plays an important role in protein accumulation in Alzheimer's disease (AD). In the present study, we examined the effect of an irreversible tTG inhibitor, NTU283, on Aβ mimic-induced AD pathogenesis in SH-SY5Y cells.
Methods:
Western blot and in-cell Western analyses were used to detect tTG and isopeptide (representing the enzyme activity of tTG) protein levels. Moreover, Hoechst and PI co-staining was performed, and caspase-3 and caspase-7 activities and the Bax/Bcl-2 ratio were determined to evaluate the effects of NTU283 on apoptosis.
Results:
The results confirmed that tTG activity was inhibited by NTU283 20–500 μmol/L in a concentration-dependent manner in SH-SY5Y cells. Contrary to our expectations, however, the isopeptide bonds were increased when cells were co-treated with Aβ and NTU283. In addition, NTU283 alone did not induce apoptosis in SH-SY5Y cells. However, when co-applied with Aβ, NTU283 promoted rather than inhibited Aβ-induced apoptosis. Consistent with the apoptotic rate, pretreating cells with different concentrations of NTU283 and Aβ significantly increased the activities of caspase-3 and caspase-7 as well as the ratio of Bax/Bcl-2.
Conclusion:
Irreversible inhibition of tTG activity did not block but rather promoted Aβ-induced apoptosis, which indicated that tTG has complex functions in AD pathogenesis.
Similar content being viewed by others
Introduction
Tissue transglutaminase (tTG) or transglutaminase-2 (TG2) belongs to the transglutaminase family (EC 2.3.2.13) and has a molecular mass of 77–85 kDa. The transglutaminase family consists of nine members: factor XIII-A subunit, TG1, tTG (TG2), TG3, TG4 through TG7 and Band 4.21. The first five members have been found in active forms in humans, but only TG1, tTG, TG3, TG6, and TG7 were detected in the brain, especially in the frontal cortex2,3, and tTG showed the highest expression. tTG has been reported to be expressed in both neurons and glial cells2 and was localized in the cytosol, membranes, extracellular fractions, and even in the nuclei of neuroblastoma cells4.
Tissue transglutaminase is a calcium-dependent enzyme that catalyzes an acyl transfer reaction between the γ-carboxamide group of a polypeptide-bound glutamine residue and the ε-amino group of a polypeptide-bound lysine residue to form an γ-glutamyl-ε-lysine covalent isopeptide bond (called isopeptide below)5,6. This covalent bond was termed a cross-link. The catalyzed cross-links can be either intramolecular or intermolecular. Intramolecular cross-links change protein conformation, while intermolecular ones lead to the formation of rigid, stable and highly insoluble protein complexes7,8.
Alzheimer's disease (AD) is a devastating neurodegenerative disease characterized by progressive cognitive impairment culminating in dementia. The main pathological mechanism is extracellular aggregations of β-amyloid (Aβ) as senile plaques (SPs)9,10 and intracellular accumulation of hyperphosphorylated Tau protein as neurofibrillary tangles (NFTs)11. Given that tTG-catalyzed cross-linking can cause Aβ or Tau accumulation and aggregation in AD-related brain regions, it is possible that tTG is involved in forming isopeptide bonds. This suggests that tTG-catalyzed cross-linking of Aβ and Tau may contribute to the pathogenesis of AD12.
Neuronal loss from cell death is a typical feature of AD progression13,14. Apoptosis is a pattern of regulated cell death, which could be identified by a variety of morphological and biochemical changes to the cell architecture that alters the cells for removal by phagocytic cells15. The major biochemical and morphological changes that typify apoptosis are the activation of a subset of caspase family proteases, including caspase-3, 6, 7, 8, and 915,16. Protein expression changes, such as alterations in B-cell lymphoma 2 (Bcl-2) and Bcl-2-associated X protein (Bax), have been reported in apoptosis as critical components of the neuronal response to injury17,18. The balance between the pro-apoptotic factor Bax and the anti-apoptotic factor Bcl-2 is important in apoptosis. Once activated, Bax can relocate from the cytoplasm to the mitochondria and cause the mitochondrial permeability transition and trigger an apoptotic cascade. Bax forms a heterodimer with Bcl-2 to block anti-apoptotic processes19,20.
The SH-SY5Y neuroblastoma cell line is a well-characterized neuro-like cell line, which has been extensively used to study neurodegenerative diseases21,22. When treated with all-trans-retinoic acid (RA), SH-SY5Y cells stop dividing and undergo neuronal differentiation23. In particular, these cells overexpress tTG protein9,24. These properties make the cell line suitable to assess the effects of enhanced tTG expression in neurodegenerative diseases.
NTU283 (1,3-dimethyl-2-[(2-oxopropyl) sulfanyl]-1H-imidazol-3-ium) is a 2-[(2-oxopropyl) thio]imidazolium derivative. It has been used to irreversibly inhibit tTG activity as reported previously25,26,27. NTU283 can block the enzyme activity by covalently modifying the catalytic site of the enzyme and then preventing substrate binding.
Although tTG inhibitors have been assessed in several diseases, few studies focused on their applications in AD and other neurodegenerative diseases. Our previous study12 reported that tTG expression and activity were increased in patients with AD and a transgenic mouse model of AD with age. In addition, the isopeptide immunoreactivity co-localized with the neurodegeneration-related proteins neurofilament, Tau, and Aβ in both AD patients and AD transgenic mice. These results suggest that tTG may be a key factor in the pathogenesis of abnormal protein aggregation in the AD process. Because they are highly resistant to proteolysis, the formation of isopeptide bonds can induce protein aggregation and deposition. Thus, we hypothesized that tTG may be used as a target in AD studies. To address this hypothesis, in our present study, the tTG inhibitor NTU283 was applied to RA-stimulated SH-SY5Y cells to determine whether tTG could alleviate the pathological features of AD.
Materials and methods
Reagents
NTU283 was provided by the Mayo Clinic, Jacksonville. All-trans-retinoic acid (RA) was purchased from Sigma-Aldrich (Louis, MO, USA). Synthetic human Aβ1-40, 42 was purchased from Bachem (Torrance, CA, USA). Anti-tTG (transglutaminase-2) and anti-isopeptide (153-81D4) antibodies were obtained from Abcam (Cambridge, MA, USA). Anti-Bax, anti-Bcl-2, anti-β-actin and horseradish peroxidase (HRP)-conjugated secondary antibodies were purchased from ProteinTech (Chicago, IL, USA). Near-infrared fluorescent secondary antibodies (IRDye 680RD, IRDye 800CW) were obtained from LI-COR (Lincoln, NE, USA).
Cell culture and treatments
SH-SY5Y neuroblastoma cells, obtained from the American Type Culture Collection (ATCC), were cultured in Dulbecco's modified Eagle's medium (DMEM)/Ham's F12 (1:1), supplemented with 10% (v/v) fetal bovine serum (FBS, Gibco, USA), 100 U/mL penicillin, and 100 μg/mL streptomycin and maintained in a humidified atmosphere of 95% air and 5% CO2 at 37 °C.
Cells were seeded in 6-well, 24-well or 96-well plates at a density of 1×105 cells/mL and cultured for 24 h. The medium was removed and replaced by cell culture medium with 3% (v/v) FBS and 20 μmol/L RA every 3 d. Six days after RA administration, the medium was removed and replaced by serum-free medium containing NTU283 at different concentrations (0, 20, 100, and 500 μmol/L). After a 2 h pretreatment, 1 μmol/L Aβ1-40,42 (called Aβ below) was added to the medium. An equivalent volume of medium was added to the culture as a control. Cells were harvested for further measurement and analysis after 24 h.
Western blot analysis
SH-SY5Y cell samples were harvested from 6-well plates and lysed in RIPA buffer containing protease inhibitor cocktail and phenylmethanesulfonyl fluoride (PMSF). Lysates were incubated for 30 min on ice and then centrifuged at 10 000×g for 10 min at 4 °C. The protein concentration in the supernatant was determined using BCA kits with bovine serum albumin (BSA) as a standard. Samples were denatured in protein sample buffer (100 mmol/L Tris–HCl, pH 6.8, 4% sodium dodecyl sulfate, 0.2% Bromophenol Blue, 20% glycerol, 20% H2O, and 200 mmol/L dithiothreitol) at 100°C for 5 min. Equal amounts of protein (30 μg per lane) were separated with 10% SDS-PAGE and then transferred to PVDF membranes (Millipore, Billerica, MA, USA). The membrane was blocked in TBST (Tris-buffered saline with 0.05% Tween) containing 5% non-fat milk for 1 h at room temperature (RT) and then incubated with primary antibodies (tTG, 1:2000; isopeptide, 1:500; Bax, 1:1000; Bcl-2, 1:500; β-actin, 1:10 000) overnight at 4 °C. After 3×10 min washes in TBST, immunodetection was performed using HRP-conjugated secondary antibodies. The membrane was visualized with an electrochemiluminescence (ECL) system and then detected on a Bio-Imaging System (DNR Lumi BIS, Jerusalem, Israel). The densities of the target bands were measured by Image-Pro Plus 6.0 and expressed as relative levels with respect to the β-actin loading control.
In-cell Western
In-cell Western (ICW) is an assay that can detect protein expression in situ28. It was performed using the Odyssey Application Protocol Manual (LI-COR). The ICW assay is a quantitative immunofluorescence-based technique, which directly detects the antigen in the cells. It can reduce sample loss compared to regular Western blots with a more complicated procedure. In addition, this assay has high sensitivity and can decrease background with near-infrared fluorescence detection. Additionally, it can be used for high through-put detection with a double-labeled method in 96-well plates.
After treatment with NTU283 or Aβ, the medium of SH-SY5Y cells, which were seeded in 96-well black plates with a clear bottom (REF655090, Greiner Bio-One, Germany), was removed. Cells were fixed by 4% polyformaldehyde in PBS for 20 min at RT, and then permeabilized by PBST-X (0.1% Triton X-100 in PBS) for 4×5 min. After 2 h blocking with 10% goat serum, primary antibodies (tTG+β-actin or isopeptide+β-actin) were incubated overnight at 4 °C. The primary antibodies were chosen in different hosts [tTG+β-actin: rabbit (IgG)+mouse (IgG); isopeptide+β-actin: mouse (IgM)+rabbit (IgG)]. The plates were washed with PBST (PBS with 0.1% Tween-20), and then, near-infrared fluorescent secondary antibodies (LI-COR) were added and incubated for 1 h at RT. Double-labeled secondary antibodies (IRDye 680RD, IRDye 800CW) for target proteins and β-actin were chosen depending on the hosts of the primary antibodies. Quantitative fluorescence signals were detected with an Odyssey SA Imaging System (LI-COR) and analyzed with Image Studio Ver 3.1. tTG and isopeptide protein levels were expressed as relative values to the β-actin levels.
Cell viability assay
To evaluate cell viability, we detected total mitochondrial activity with a Cell Counting Kit-8 (CCK-8) (Beyotime, Haimen, China). After the cells were treated with various concentrations of NTU283 (1, 4, 20, 100, and 500 μmol/L) for 24 h, the cells were lysed with one-tenth volume of CCK-8 reagent and incubated for 4 h at 37 °C. The absorbance at 450 nm and 630 nm of all samples was measured using a microplate reader (PerkinElmer, USA). The OD value was calculated to determine the viability of SH-SY5Y cells.
Hoechst 33342 and PI co-staining
Apoptotic or necrotic cell death was characterized using Hoechst 33342 and propidium iodide (PI) double staining29,30. SH-SY5Y cells with different treatments were stained with Hoechst 33342 and PI for 30 min on ice. After washing with D-Hanks' solution, the cells were observed and imaged with an imaging system attached to a fluorescence microscope (Nikon, ELWD, 0.3, JP) at 350 nm and 550 nm excitation wavelength. Five random photographs were captured in each sample in different sections, and Image-Pro Plus 6.0 was used to analyze the apoptotic cell count.
Caspase-3 and caspase-7 activity assay
The caspase-3 and caspase-7 activity kits were purchased from Promega (Madison, WI, Apo-ONE®). The substrate was diluted 1:100 with the buffer to obtain the working reagent at the desired volume. SH-SY5Y cells were treated in 96-well black plates (REF655090, Greiner Bio-One) and divided into following groups: blank (reagent+cell culture medium), negative control (reagent+vehicle-treated SH-SY5Y cells), and assay (reagent+treated SH-SY5Y cells). Reagent was added to each well at a volume of 100 μL (total volume 200 μL) and mixed thoroughly, followed by incubation for 4 h in the dark at RT. The fluorescence signal was detected on a microplate reader (PerkinElmer, USA) with a 499 nm excitation wavelength and a 521 nm emission wavelength.
Statistical analysis
All data are expressed as the mean±standard error of the mean (SEM). Statistical analyses were performed with one-way ANOVA followed by post hoc comparison and linear regression analysis with a threshold of P<0.05 using SPSS 17.0 software. The results from individual experiments were averaged within each experimental group.
Results
NTU283 did not affect SH-SY5Y cell viability
We first examined the toxicity of NTU283 on SH-SY5Y cells by comparing the cells treated with 5 concentrations of NTU283 to the untreated control. No significant decrease in cell viability was observed following incubation of the cells with up to 500 μmol/L of NTU283 (data not shown).
Isopeptide level increased after co-treatment with NTU283 and Aβ
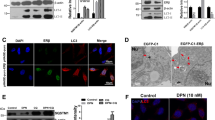
To test the inhibitory effects of NTU283 on tTG, we examined tTG and isopeptide (representing the enzyme activity of tTG) protein levels by Western blot analyses. Although NTU283 did not decrease the tTG protein level (Figure 1A), NTU283 reduced the isopeptide level (Figure 1B), suggesting this chemical may inhibit tTG enzyme activity to decrease isopeptide cross-linking.
Western blotting analysis of tTG and isopeptide in NTU283- and Aβ-treated SH-SY5Y cells. Cells were seeded in 6-well plates. After 6 d of administration of RA, serum-free medium containing different concentrations of NTU283 (0, 20, 100, and 500 μmol/L) with or without 1 μmol/L Aβ was added to each well. Cells were harvested after a 24-h treatment. Western blot analysis was applied to detect tTG and isopeptide protein levels. Western blotting images were collected using a Bio-Imaging System (DNR Lumi BIS) with an ECL system and analyzed by Image-Pro Plus 6.0. (A) Representative Western blots for tTG protein levels (top) and quantitative analysis (bottom) are shown. (B) Representative Western blots for isopeptide protein levels (top) and quantitative analysis (bottom) are shown. All bands were quantified and normalized by β-actin. Statistical data are expressed as the mean±SEM from six independent experiment. *P<0.05, **P<0.01 vs each control (0 μmol/L NTU283) with or without Aβ; #P<0.05, ##P<0.01 Aβ(-) group vs Aβ(+) group.
Neither NTU283 nor Aβ alone or combined altered the tTG protein levels, while the levels of isopeptide were significantly reduced by NTU283 alone (P<0.05, Lane 4) in a concentration-dependent manner (Figure 1B), indicating the inhibition of tTG activity. As expected, Aβ alone increased the isopeptide level (Lane 5 compared to Lane 1). However, when cells were pretreated with NTU283, addition of Aβ substantially increased the level of isopeptide at the same concentrations (Figure 1B). The highest level was observed at 100 μmol/L NTU283 (P<0.01, Lane 7).
To confirm the results of the Western blot analyses, we further examined tTG and isopeptide protein levels using in-cell Western assays. Consistent with the results above, no significant changes in tTG level were observed following NTU283 or Aβ treatment or a combination (Figure 2A). However, isopeptide levels were significantly enhanced in the cells co-treated with NTU283 and Aβ (P<0.01), although NTU283 alone inhibited the formation of isopeptide cross-linking (Figure 2B).
In-cell Western analysis of tTG and isopeptide in NTU283- and Aβ-treated SH-SY5Y cells. SH-SY5Y cells were plated in 96-well black plates with clear bottoms. After 6 d of administration of RA, serum-free medium containing different concentration of NTU283 (0, 20, 100, and 500 μmol/L) with or without 1 μmol/L Aβ was added to each well. After a 24 h treatment, cells were fixed and permeabilized. Samples were co-incubated with anti-tTG or anti-isopeptide and anti-β-actin antibodies overnight at 4 °C. Then, near-infrared fluorescent second antibodies (LI-COR) were incubated for 1 h at RT. The target proteins and β-actin were double-labeled with different second antibodies (IRDye 680RD, IRDye 800CW) matching their primary antibodies host. Fluorescence signals were detected by Odyssey SA (LI-COR) and analyzed with Image Studio Ver 3.1. All fluorescence densities were quantified and normalized by β-actin. The representative images of tTG (A), isopeptide (B) and relative quantification levels are shown. Statistical data are expressed as the mean±SEM from six independent experiment groups (n=6). *P<0.05 vs each control (0 μmol/L NTU283) with or without Aβ. #P<0.05, ##P<0.01 Aβ(-) group vs Aβ(+) group.
NTU283 promotes Aβ-induced apoptosis
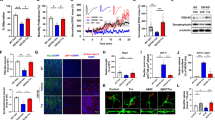
To explain the unexpected findings above, we determined whether apoptosis had occurred. Double staining of Hoechst 33342 and PI was used to detect apoptosis. There was almost no neuronal apoptosis detected following treatment with NTU283 alone at different concentrations for 24 h. Aβ at 1 μmol/L could induce low levels of apoptosis in SH-SY5Y cells, as shown by the Hoechst-positive cells with condensed, fragmented or degraded nuclei, as well as the sparse numbers of PI-labeled cells compared to vehicle-control cells (Aβ-free) (P<0.01) (Figure 3A). However, when the cells were pretreated with NTU283 and then co-incubated with Aβ, a dramatic increase in apoptotic cells was observed in a concentration-dependent manner. Only a few necrotic cells were observed. The highest apoptotic rate (approximately 50%) was observed following co-treatment with 100 μmol/L NTU283 and Aβ (P<0.01) (Figure 3B).
Apoptotic detection of SH-SY5Y by Hoechst 33342/PI co-staining and caspase assays. (A) SH-SY5Y cells were pretreated as indicated in 24-well plates. Hoechst 33342 and PI co-staining were applied and then detected with fluorescence microscopy at 350 nm excitation and 550 nm excitation wavelength. Then, 150–250 cells were observed per vision field. Representative images of Hoechst (blue), PI (red), and Merged (purple) are shown on the left. Aβ (–) groups included different NTU283 concentrations: 0 μmol/L (a–c), 20 μmol/L (d–f), 100 μmol/L (g–i), and 500 μmol/L (j–l), as well as Aβ (+) groups containing 0 μmol/L (m–o), 20 μmol/L (p–r), 100 μmol/L (s–u), and 500 μmol/L (v–x) NTU283. (B) Cell apoptosis determined by Hoechst 33342/PI co-staining was analyzed by Image-Pro Plus 6.0. Bright blue nuclear staining without red staining indicated apoptotic cells. The rate was calculated by the number of apoptotic cells/the number of total cells. Five independent captured images from each sample of three experimental groups (n=3) were analyzed. (C) Caspase-3 and caspase-7 enzyme activities were detected in SH-SY5Y cells that were pretreated as indicated and plated in 96-well black plates. Caspase kit reagent was added to each well at a volume of 100 μL (total volume 200 μL) and mixed thoroughly, and the samples were incubated for 4 h in the dark at RT. Fluorescence intensity was detected by a microplate reader. Statistical data were calculated from three replicate samples in each independent group from three experimental groups and are expressed as the mean±SEM. *P<0.05, **P<0.01 vs each control (0 μmol/L NTU283) with or without Aβ. ##P<0.01 Aβ(-) group vs Aβ(+) group.
Caspase-3 and caspase-7 enzyme activities were further detected to confirm the above findings. The results demonstrated (Figure 3C) that caspase activities were higher in Aβ-treated cells compared to those of the vehicle control (Aβ-free) (P<0.01). Consistent with the apoptotic rate, after pretreating the cells with different concentrations of NTU283, the activities of caspase-3 and caspase-7 were significantly enhanced by Aβ stimulation (P<0.01).
Bax and Bcl-2 were detected by Western blot analysis as well. Bax showed higher expression in Aβ-treated cells compared to that of the vehicle control (Aβ-free) (Figure 4A). Significant differences were observed at a concentration of 100 μmol/L of NTU283 (P<0.05, Lane 3 vs Lane 7). Treatment with NTU283 alone did not significantly increase Bax protein levels. However, co-treatment of cells with NTU283 and Aβ substantially increased the Bax level, with a peak at 100 μmol/L (P<0.01, Lane 7). Meanwhile, lower levels of Bcl-2 were observed in the groups treated with Aβ alone and pretreated with NTU283 (Figure 4B). A high concentration of NTU283 (500 μmol/L) also decreased Bcl-2 expression (P<0.05, Lane 4). Regardless of Aβ co-treatment, the Bax/Bcl-2 ratio increased with NTU283 concentration (Figure 4C). Consistent with the above results, co-treatment of cells with NTU283 and Aβ enhanced the ratio of Bax/Bcl-2 significantly, although NTU283 alone also increased the ratio to some extent. The peak was observed at 100 μmol/L of NTU283 with Aβ (P<0.01).
Alterations in the apoptotic signal proteins Bax and Bcl-2. SH-SY5Y cells were pretreated as indicated in 6-well plates. Bax (A) and Bcl-2 (B) protein levels were detected by Western blot analysis. Western blotting images were captured on a Bio-Imaging System (DNR Lumi BIS) using an ECL system and were analyzed by Image-Pro Plus 6.0. Representative bands of Bax, Bcl-2, and β-actin are shown. All bands were quantified and normalized by β-actin. (C) The Bax/Bcl-2 ratio is shown on the graph to demonstrate apoptosis more objectively. Statistical data are expressed as the mean±SEM from six independent experiment groups. *P<0.05, **P< 0.01 vs each control (0 μmol/L NTU283) with or without Aβ. #P<0.05, ##P<0.01 Aβ(-) group vs Aβ(+) group.
In summary, NTU283 can promote Aβ-induced apoptosis, although NTU283 itself cannot induce apoptosis in SH-SY5Y cells alone.
Discussion
tTG expression and activity have been reported to be increased in Alzheimer's disease. tTG can catalyze Aβ cross-linking to form isopeptide polymers3,12. Being highly resistant to proteolysis, these bonds can induce protein aggregation and deposition. It has been ever hypothesized that multiple factors, such as trauma31,32, inflammation33,34 or ischemic damage35,36, in sporadic AD or overproduction of Aβ in familial AD may lead to cross-linking of AD-related proteins. The overexpression of tTG in turn catalyzed protein cross-linking, and aggravated the pathogenesis of AD16. Thus, tTG may be an AD therapeutic target.
We tested the effect of a tTG inhibitor on SH-SY5Y cells, which were stimulated to overexpress tTG by RA. Moreover, Aβ was added to mimic an AD pathological condition because it is involved in protein aggregation and neural toxicity in AD pathogenesis37,38,39. tTG and its product isopeptide were detected by Western blot analysis at different molecular weights or by in-cell Western assays. In the present study, NTU283 was applied as an irreversible inhibitor of tTG25,27,40 to block the formation of covalent bonding. As expected, tTG enzyme activity (isopeptide level) was effectively inhibited in a concentration-dependent manner with NTU283 alone. Thus, we speculated that NTU283 would inhibit isopeptide formation in the neuronal cells with Aβ stimulation. However, in contrast to our hypothesis, the isopeptide level substantially increased rather than decreased with co-treatment of NTU283 and Aβ.
The loss of neurons is a typical feature of AD14,41. It has been reported that neuronal apoptosis occurs in AD pathogenesis42,43. The Aβ oligomers were toxic to neurons and may trigger oxidative stress and apoptosis44. tTG-mediated cross-linking may also play a role in Aβ aggregation and accumulation in cells. In our study, apoptosis was induced by Aβ stimulation in SH-SY5Y cells as verified by morphological staining, caspase activities and Bax/Bcl-2 protein immunoblotting. However, instead of apoptosis inhibition, increased apoptosis was observed after NTU283 and Aβ co-treatment, which was contrary to our expectations. These results suggested that tTG enzyme activity is involved in the regulation of apoptosis.
Previous studies have indicated that transglutaminases are involved in the regulation of apoptosis45,46. tTG has crucial roles in many cell biological processes, including differentiation, adhesion, extracellular matrix formation, and cell apoptosis, by modifying its substrate proteins47. It was suggested that tTG not only regulated apoptosis but also interacted with apoptosis-related factors48. Recent studies have indicated that the complexes formed by tTG and other proteins could inhibit apoptotic cascades47,49. Additionally, tTG can interact with the pro-apoptotic protein Bax to inhibit apoptosis49,50.
Moreover, because of the calcium-binding activity of tTG, increased free calcium may be released to the cytoplasm and extracellular matrix after irreversible inhibition of the enzyme. Under these conditions, high concentrations of free calcium would cause cell stress and finally lead to cell death51.
From our data and previous reports on the roles of tTG in apoptosis, we hypothesized that the effects of the tTG inhibitor NTU283 on Aβ-induced apoptosis may be related to inhibition of tTG interacting with the Bax protein, resulting in the release of free Bax and activation of apoptotic cascades. With increased tTG inhibition, more active Bax would aggravate cell apoptosis. Meanwhile, a high concentration of calcium was released to the cellular environment. An increased risk of apoptosis and calcium-induced cell stress may directly lead to neural cell death. To alleviate cell death and lysis, tTG may catalyze more proteins, including neurodegenerative-related proteins, fibronectin52, and fibrinogen53 to form isopeptide cross-linking bonds on the cell surface for packing cell as a cocoon, which is involved in wound healing and adhesion functions of tTG54,55,56. Highly resistant to proteolysis, these proteins would protect cells from apoptotic death or other external risks. This may explain why the isopeptide level was increased with NTU283 and Aβ co-treatment. This hypothesis should be assessed in further studies.
In conclusion, tissue transglutaminase can catalyze Aβ cross-linking as γ-glutamyl-ε-lysine isopeptide bonds in AD, resulting in protein aggregation and deposition. Thus, tTG may be a target for alleviating AD. Inhibition of tTG activity with NTU283 alone can reduce isopeptide protein aggregation in SH-SY5Y cells. However, in contrast to our expectations, the isopeptide levels were increased following Aβ and NTU283 co-treatment. Moreover, it should be emphasized that irreversible inhibitors such as NTU283 could promote Aβ-induced neuron apoptosis, although NTU283 itself cannot induce apoptosis. This type of irreversible inhibitor treatment must be evaluated in future studies of AD because its irreversible inhibitory effects may affect the normal functions of tTG.
Author contribution
Rui WANG designed and guided the research, analyzed the data and polished the paper; Ji ZHANG designed and performed the research, analyzed the data and wrote the paper; Yi-rong DING assisted in performing the research.
References
Wilhelmus MM, van Dam AM, Drukarch B . Tissue transglutaminase: a novel pharmacological target in preventing toxic protein aggregation in neurodegenerative diseases. Eur J Pharmacol 2008; 585: 464–72.
Kim SY, Grant P, Lee JH, Pant HC, Steinert PM . Differential expression of multiple transglutaminases in human brain increased expression and cross-linking by transglutaminases 1 and 2 in Alzheimer's disease. J Biol Chem 1999; 274: 30715–21.
Wang DS, Uchikado H, Bennett DA, Schneider JA, Mufson EJ, Wu J, et al. Cognitive performance correlates with cortical isopeptide immunoreactivity as well as Alzheimer type pathology. J Alzheimers Dis 2008; 13: 53–66.
Lesort M, Attanavanich K, Zhang J, Johnson GV . Distinct nuclear localization and activity of tissue transglutaminase. J Biol Chem 1998; 273: 11991–4.
Aeschlimann D, Mosher D, Paulsson M . Tissue transglutaminase and factor XIII in cartilage and bone remodeling. Seminars in thrombosis and hemostasised. Thieme Medical Publishers, Inc; 1996. p 437–43.
Greenberg CS, Birckbichler PJ, Rice RH . Transglutaminases: multifunctional cross-linking enzymes that stabilize tissues. FASEB J 1991; 5: 3071–7.
Folk J . The trimethylacetyl-transglutaminase complex. Method Enzymol 1981; 87: 36–42.
Lorand L, Conrad SM . Transglutaminases. In: Transglutaminase. Springer; 1984. p 9–35.
Zhang J, Lesort M, Guttmann RP, Johnson GV . Modulation of the in situ activity of tissue transglutaminase by calcium and GTP. J Biol Chem 1998; 273: 2288–95.
Zhang W, Johnson BR, Suri DE, Martinez J, Bjornsson TD . Immunohistochemical demonstration of tissue transglutaminase in amyloid plaques. Acta Neuropathol 1998; 96: 395–400.
Singer SM, Zainelli GM, Norlund MA, Lee JM, Muma NA . Transglutaminase bonds in neurofibrillary tangles and paired helical filament tau early in Alzheimer's disease. Neurochem Int 2002; 40: 17–30.
Zhang J, Wang S, Huang W, Bennett D, Dickson D, Wang D, et al. Tissue transglutaminase and its product isopeptide are increased in Alzheimer's disease and APPswe/PS1dE9 double transgenic mice brains. Mol Neurobiol 2016; 53: 5066–78.
Yang Y, Geldmacher DS, Herrup K . DNA replication precedes neuronal cell death in Alzheimer's disease. J Neurosci 2001; 21: 2661–8.
Luo L, O'Leary DD . Axon retraction and degeneration in development and disease. Annu Rev Neurosci 2005; 28: 127–56.
Martin Seamus J, Henry Conor M, Cullen Sean P . A perspective on mammalian caspases as positive and negative regulators of inflammation. Mol Cell 2012; 46: 387–97.
Taylor RC, Cullen SP, Martin SJ . Apoptosis: controlled demolition at the cellular level. Nat Rev Mol Cell Biol 2008; 9: 231–41.
Khoury CM, Yang Z, Ismail S, Greenwood MT . Characterization of a novel alternatively spliced human transcript encoding an N-terminally truncated Vps24 protein that suppresses the effects of Bax in an ESCRT independent manner in yeast. Gene 2007; 391: 233–41.
Lu J, Moochhala S, Kaur C, Ling EA . Changes in apoptosis-related protein (p53, Bax, Bcl-2, and Fos) expression with DNA fragmentation in the central nervous system in rats after closed head injury. Neurosci Lett 2000; 290: 89–92.
Hou Q, Cymbalyuk E, Hsu SC, Xu M, Hsu YT . Apoptosis modulatory activities of transiently expressed Bcl-2: roles in cytochrome c release and Bax regulation. Apoptosis 2003; 8: 617–29.
Lebedeva IV, Sarkar D, Su ZZ, Kitada S, Dent P, Stein C, et al. Bcl-2 and Bcl-xL differentially protect human prostate cancer cells from induction of apoptosis by melanoma differentiation associated gene-7, mda-7/IL-24. Oncogene 2003; 22: 8758–73.
Wang R, Malter JS, Wang DS . N-acetylcysteine prevents 4-hydroxynonenal-and amyloid-β-induced modification and inactivation of neprilysin in SH-SY5Y cells. J Alzheimers Dis 2010; 19: 179–89.
Shen JN, Wang DS, Wang R . The protection of acetylcholinesterase inhibitor on β-amyloid-induced injury of neurite outgrowth via regulating axon guidance related genes expression in neuronal cells. Int J Clin Exp Pathol 2012; 5: 900.
Pahlman S, Ruusala AI, Abrahamsson L, Mattsson ME, Esscher T . Retinoic acid-induced differentiation of cultured human neuroblastoma cells: a comparison with phorbolester-induced differentiation. Cell Differ 1984; 14: 135–44.
Verhaar R, Jongenelen CA, Gerard M, Baekelandt V, Van Dam AM, Wilhelmus MM, et al. Blockade of enzyme activity inhibits tissue transglutaminase-mediated transamidation of alpha-synuclein in a cellular model of Parkinson's disease. Neurochem Int 2011; 58: 785–93.
Freund KF, Doshi KP, Gaul SL, Claremon DA, Remy DC, Baldwin JJ, et al. Transglutaminase inhibition by 2-[(2-oxopropyl) thio] imidazolium derivatives: mechanism of factor XIIIa inactivation. Biochemistry 1994; 33: 10109–19.
Maiuri L, Ciacci C, Ricciardelli I, Vacca L, Raia V, Rispo A, et al. Unexpected role of surface transglutaminase type II in celiac disease. Gastroenterology 2005; 129: 1400–13.
Skill NJ, Johnson TS, Coutts IG, Saint RE, Fisher M, Huang L, et al. Inhibition of transglutaminase activity reduces extracellular matrix accumulation induced by high glucose levels in proximal tubular epithelial cells. J Biol Chem 2004; 279: 47754–62.
Egorina E, Sovershaev M, Østerud B . In cell Western assay: a new approach to visualize tissue factor in human monocytes. J Thromb Haemost 2006; 4: 614–20.
Yin X, Zhou X, Xue Z, Tian L, Zhou Y, Yang L, et al. Prokaryotic expression and functional analysis of the Mb1514 gene in Mycobacterium bovis. Mol Cell Biochem 2014; 385: 43–52.
Huang X, Yi C, Fan Y, Zhang Y, Zhao L, Liang Z, et al. Magnetic Fe3O4 nanoparticles grafted with single-chain antibody (scFv) and docetaxel loaded β-cyclodextrin potential for ovarian cancer dual-targeting therapy. Mat Sci Eng C 2014; 42: 325–32.
Fleminger S, Oliver D, Lovestone S, Rabe-Hesketh S, Giora A . Head injury as a risk factor for Alzheimer's disease: the evidence 10 years on; a partial replication. J Neurol Neurosurg Psychiatry 2003; 74: 857–62.
Szczygielski J, Mautes A, Steudel W, Falkai P, Bayer T, Wirths O . Traumatic brain injury: cause or risk of Alzheimer's disease? A review of experimental studies. J Neural Transm ( (Vienna) 2005; 112: 1547–64.
Sastre M, Klockgether T, Heneka MT . Contribution of inflammatory processes to Alzheimer's disease: molecular mechanisms. Int J Dev Neurosci 2006; 24: 167–76.
Rogers JT, Lahiri DK . Metal and inflammatory targets for Alzheimer's disease. Curr Drug Targets 2004; 5: 535–51.
Rosano C, Newman AB . Cardiovascular disease and risk of Alzheimer's disease. Neurol Res 2006; 28: 612–20.
Koistinaho M, Koistinaho J . Interactions between Alzheimer's disease and cerebral ischemia — focus on inflammation. Brain Res Rev 2005; 48: 240–50.
Sun X, Chen WD, Wang YD . β-Amyloid: the key peptide in the pathogenesis of Alzheimer's disease. Front Pharmacol 2015; 6: 221.
Viola KL, Klein WL . Amyloid β oligomers in Alzheimer's disease pathogenesis, treatment, and diagnosis. Acta Neuropathol 2015; 129: 183–206.
Zueva I, Semenov V, Mukhamedyarov M, Lushchekina S, Kharlamova A, Petukhova E, et al. 6-Methyluracil derivatives as acetylcholinesterase inhibitors for treatment of Alzheimer's disease. Int J Risk Saf Med 2015; 27: S69–S71.
Harrison C, Layton C, Hau Z, Bullock A, Johnson T, MacNeil S . Transglutaminase inhibitors induce hyperproliferation and parakeratosis in tissue-engineered skin. Br J Dermatol 2007; 156: 247–57.
Heggland I, Storkaas IS, Soligard HT, Kobro-Flatmoen A, Witter MP . Stereological estimation of neuron number and plaque load in the hippocampal region of a transgenic rat model of Alzheimer's disease. Eur J Neurosci 2015; 41: 1245–62.
Bayer AU, Keller ON, Ferrari F, Maag KP . Association of glaucoma with neurodegenerative diseases with apoptotic cell death: Alzheimer's disease and Parkinson's disease. Am J Ophthalmol 2002; 133: 135–7.
Yang Y, Mufson EJ, Herrup K . Neuronal cell death is preceded by cell cycle events at all stages of Alzheimer's disease. J Neurosci 2003; 23: 2557–63.
Shelat PB, Chalimoniuk M, Wang JH, Strosznajder JB, Lee JC, Sun AY, et al. Amyloid beta peptide and NMDA induce ROS from NADPH oxidase and AA release from cytosolic phospholipase A2 in cortical neurons. J Neurochem 2008; 106: 45–55.
Gabr SA, Berika MY, Alghadir AH . Apoptosis and clinical severity in patients with psoriasis and HCV infection. Indian J Dermatol 2014; 59: 230.
Tucholski J, Johnson GV . Tissue transglutaminase differentially modulates apoptosis in a stimuli-dependent manner. J Neurochem 2002; 81: 780–91.
Lee HJ, Lee CH . Transglutaminase-2 is involved in expression of osteoprotegerin in MG-63 osteosarcoma cells. Biomol Ther 2013; 21: 204.
Nurminskaya MV, Belkin AM . Cellular functions of tissue transglutaminase. Int Rev Cel Mol Biol 2012; 294: 1.
Cho SY, Lee JH, Bae HD, Jeong EM, Jang GY, Kim CW, et al. Transglutaminase 2 inhibits apoptosis induced by calcium overload through down-regulation of Bax. Exp Mol Med 2010; 42: 639–50.
Wang W, Li X, Han XZ, Meng FB, Wang ZX, Zhai YQ, et al. Transglutaminase-2 is Involved in cell apoptosis of osteosarcoma cell line U2OS under hypoxia condition. Cell Biochem Biophys 2015; 72: 283–8.
Bernardi P, Rasola A . Calcium and cell death: the mitochondrial connection. In: Carafoli E, Brini M, editors. Calcium signalling and disease: molecular pathology of calcium. Springer; 2007. p 481–506.
Jones R, Nicholas B, Mian S, Davies P, Griffin M . Reduced expression of tissue transglutaminase in a human endothelial cell line leads to changes in cell spreading, cell adhesion and reduced polymerisation of fibronectin. J Cell Sci 1997; 110: 2461–72.
Martinez J, Rich E, Barsigian C . Transglutaminase-mediated cross-linking of fibrinogen by human umbilical vein endothelial cells. J Biol Chem 1989; 264: 20502–8.
Wang Z, Telci D, Griffin M . Importance of syndecan-4 and syndecan-2 in osteoblast cell adhesion and survival mediated by a tissue transglutaminase-fibronectin complex. Exp Cell Res 2011; 317: 367–81.
Telci D, Griffin M . Tissue transglutaminase (TG2) — a wound response enzyme. Front Biosci 2005; 11: 867–82.
Verderio E, Johnson T, Griffin M . Tissue transglutaminase in normal and abnormal wound healing: review article. Amino Acids 2004; 26: 387–404.
Acknowledgements
This work was supported by the grants to Rui WANG from the National Natural Science Foundation of China (No 81072627 and 81230090), the Shanghai Committee of Science and Technology (No 12431900901), and the 111 Project of the Chinese Ministry of Education (No B07023).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, J., Ding, Yr. & Wang, R. Inhibition of tissue transglutaminase promotes Aβ-induced apoptosis in SH-SY5Y cells. Acta Pharmacol Sin 37, 1534–1542 (2016). https://doi.org/10.1038/aps.2016.95
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/aps.2016.95