Abstract
Cholesterol efflux from lipid-loaded cells is a key athero-protective event that counteracts cholesterol uptake. The imbalance between cholesterol efflux and uptake determines the prevention or development of atherosclerosis. Many proteins and factors participate in the cholesterol efflux event. However, there are currently no systematic models of reverse cholesterol transport (RCT) that include most RCT-related factors and events. On the basis of recent research findings from other and our laboratories, we propose a novel model of one center and four systems with coupling transportation and networking regulation. This model represents a common way of cholesterol efflux; however, the systems in the model consist of different proteins/factors in different cells. In this review, we evaluate the novel model in vascular smooth muscle cells (VSMCs) and macrophages, which are the most important original cells of foam cells. This novel model consists of 1) a caveolae transport center, 2) an intracellular trafficking system of the caveolin-1 complex, 3) a transmembrane transport system of the ABC-A1 complex, 4) a transmembrane transport system of the SR-B1 complex, and 5) an extracelluar trafficking system of HDL/Apo-A1. In brief, the caveolin-1 system transports cholesterol from intracellular compartments to caveolae. Subsequently, both ABC-A1 and SR-B1 complex systems transfer cholesterol from caveolae to extracellular HDL/Apo-A1. The four systems are linked by a regulatory network. This model provides a simple and concise way to understand the dynamic process of atherosclerosis.
Similar content being viewed by others
Introduction
Over the past few decades, our understanding of the pathologic mechanisms of atherosclerosis has progressed significantly. Multiple proteins and factors are involved in the process of atherosclerosis. Many recent reviews have summarized the landmark events of atherosclerosis1, 2, 3. The blood plasma cholesterol level plays an important role in atherosclerosis. Regulation of the blood plasma cholesterol level involves cholesterol uptake, biosynthesis, transportation, metabolism, and secretion4, 5, 6, 7. This review focuses on recent advances in our understanding of reverse cholesterol transport (RCT). The general concept of RCT is to transport cholesterol from peripheral tissues and cells to the liver, transform it into bile acids, and finally eliminate it from the body. RCT may prevent the formation and development of atherosclerosis by decreasing cholesterol levels in the plasma and accumulation in the wall of the arteries. The narrower concept of RCT involves cholesterol efflux from cells. In this review, we focus on the narrower RCT from macrophages and vascular smooth muscle cells (VSMCs). Monotype-derived macrophages and VSMCs migrate to the subendothelial space and form foam cells via the uptake of lipoproteins, especially low density lipoproteins (LDLs). During the long-term period in which macrophages and VSMCs form foam cells, cholesterol is transformed to cholesterol esters. It is thought that macrophages and VSMCs are designated as lipid-loaded cells if the ratio of cholesterol esters in the total cholesterol is less than 50% in these cells; they are denoted as foam cells if the ratio is more than 50%. Foam cells have very weak or no RCT capability, whereas lipid-loaded cells have a strong (at the earlier stage) or a little (at the mid and later stages) RCT capability. Cholesterol efflux from lipid-loaded cells is a key athero-protective event against cholesterol uptake, and the imbalance between cholesterol efflux and uptake determines the prevention or development of atherosclerosis. Therefore, cholesterol efflux is an important event in preventing the transformation of lipid-loaded cells into foam cells.
In fact, many proteins and factors are involved in cholesterol efflux, as shown in Table 1, such as the caveolin family (caveolin-1, -2, -3), the ATP binding cassette transporter (ABC), apolipoprotein (Apo-A1), scavenger receptor class B1 (SR-B1), cholesterol synthetase and metabolic enzymes (eg, LCAT, CETP, ACAT, LPL), the immunophilin family, nuclear receptors and factors (PPARs, LXR, RXR, FXR), protein kinases (PKC, PKA), and sterol regulatory element-binding proteins (SREBPs). These molecules are directly or indirectly involved in RCT. Currently, there is no systematic model of RCT that evaluates the main pathways of cholesterol efflux. By summarizing recent international studies and our research, we propose a novel RCT model of “four systems and one center with coupling transportation and networking regulation”. As shown in Figure 1, the model consists of 1) an intracellular trafficking system of the caveolin-1 complex, 2) a transmembrane transport system of the ABC-A1 complex, 3) a transmembrane transport system of the SR-B1 complex, 4) an extracelluar trafficking system of HDL/Apo-A1, and 5) a caveolae transport center. In brief, the caveolin-1 complex system transports cholesterols from intracellular compartments into caveolae; both ABCA1 and SR-B1 complex systems then transfer the cholesterols from caveolae to extracellular HDL/Apo-A1, which then transfers cholesterol to the liver. The whole process is known as coupling transportation. A network regulates the four systems and the entire transportation process. This model represents a common rule of cholesterol efflux; however, the systems in the model consist of different proteins/factors in different cells. In this review, we evaluate this novel model in VSMCs and macrophages, which are the most important original cells of foam cells. This model facilitates our understanding of the mechanism responsible for reverse cholesterol transmembrane transportation.
Working model of cellular reverse cholesterol transport. A novel RCT model of “four systems and one center with coupling transportation and networking regulation”. The model consists of 1) an intracellular trafficking system of the caveolin-1 complex, 2) a transmembrane transport system of the ABC-A1 complex, 3) a transmembrane transport system of the SR-B1 complex, 4) an extracelluar trafficking system of HDL/Apo-A1, and 5) a caveolae transport center. In brief, the caveolin-1 complex system transports cholesterols from intracellular compartments into caveolae; both the ABCA1 and the SR-B1 complex systems then transfer the cholesterol from caveolae to HDL/Apo-A1, which finally transfers cholesterol to the liver. The whole process is known as coupling transportation. A network regulates the four systems and the entire transportation process.
Model of reverse cholesterol transportation
One transportation center-caveolae
Caveolae, which are 50-100 nm plasma membrane invaginations, are rich in cholesterol and phosphosphingolipids. These flask-shaped lipid-raft structures have many proposed functions in cell signaling, endocytosis, and cholesterol homeostasis. The formation and maintenance of caveolae is primarily due to caveolin, which is a main protein marker of caveolae. This protein contains a cytoplasmic C-terminus and a cytoplasmic N-terminus that are linked by a hydrophobic hairpin inserted in the membrane. Caveolins lead to local changes in the morphology of the membrane. Caveolae usually appear in nonclassical invagination structures in many cell types, such as grape-like clusters in skeletal muscle cells, H rosettes in adipocytes, and tube-like cavities in endothelial cells. Caveolae in macrophages exhibit different structure types according to the expression level of caveolin-1.
Caveolae, as cholesterol storage pools, mediate transmembrane cholesterol transportation and the endocytosis and transcytosis of lipoprotein8, 9. Recently, we found that caveolae and caveolin-1 mediate the endocytosis and transcytosis of oxidized low-density lipoprotein (ox-LDL) in endothelial cells10. Caveolae in the plasma membrane may invaginate and form caveolar vesicles that contain cholesterol or lipoprotein. Concomitantly, the caveolar vesicles may fuse with the plasma membrane and form caveolae, which mediate cholesterol efflux, as shown in Figure 1. Excluding vesicle-mediated cholesterol transportation, many receptors in caveolae, such as low density lipoprotein (LDL) receptors, SR-B1, CD36, and ABC-A1, mediate cholesterol efflux. Fielding et al11, 12 reported that caveolin-1 protein and mRNA were upregulated, and the amount of caveolae increased several-fold when fibroblasts or monocytes/macrophages were cultured together with LDL, which resulted in an approximately 15% increase in intracellular free cholesterol. Subsequent incubation of these cells with plasma HDL selectively unloaded free cholesterol from caveolae into the medium. Pretreatment of cells with caveolin-1 siRNA significantly reduced caveolae and decreased cholesterol efflux. It was reported that cholesterol efflux decreased approximately 80% after vanadate, a specific inhibitor of Ca2+-ATPase localized almost exclusively within caveolae, had destroyed caveolae in VSMCs and endothelial cells13. Additionally, we found that SR-B1 transgenic mice fed a high-lipid diet had fewer caveolae in the vein endomembrane network than wild type mice but higher incidence rates of atherosclerosis14. Additionally, extracellular trafficking systems that reach the plasma membrane, bind to SR-B1 and ABC-A1 in caveolae, and remove cholesterol stored in caveolae. If cholesterol is not stored in caveolae, extracellular trafficking systems can not transport cholesterol from cells despite reaching the plasma membrane. Therefore, caveolae are communication centers of cholesterol transmembrane exchange.
Four trafficking systems
Intracellular trafficking system of the caveolin-1 complex
Caveolin-1, the most important member of the caveolin family and a multi-functional 21–24 kDa signaling protein, is enriched in caveolae, endoplasmic reticulum and Golgi bodies and shuttles back and forth between the cytoplasm and the cell membrane via the intracellular trafficking of caveolar vesicles. Its function is highly correlated with its tyrosine phosphorylation level. Two proline residues at the two ends of a hydrophobic region of 33 amino acids cause the N-terminal and C-terminal sequences to form a hairpin structure. The hydrophobic region alone is not strong enough to cause firm caveolin-1 binding to the cell membrane; Schelgel15 reported that the N-terminal membrane attachment domain (N-MAD, residues 82–101) and the C-terminus (C-MAD, residues 135–150) of caveolin-1 are sufficient to anchor caveolins to the cell membrane. The highly conserved N-MAD forms the caveolin scaffolding domain (CSD), which can combine with and inactivate some intracellular signaling molecules, such as Src tyrosine kinase, G protein α subunit, PKC-α, PKA, and H-Has, to negatively regulate signal transduction16. CSD is essential for the interaction between caveolin-1 and lipids, but it is not clear how the competitive binding of proteins and phospholipids to the same domain in caveolin-1 is regulated.
Caveolin-1-deficient L1210J cells are unable to rapidly transfer intracellular synthetic cholesterol out of cells, but this function is recovered following the delivery of caveolin-117. The intracellular cholesterol ester decreases 50% and the accumulation of cholesterol within the cell membrane increases in HepG2 cells transfected with a caveolin-1 expression plasmid18. Our research revealed that in VSMCs treated with 50 mg/L ox-LDL, caveolin-1 expression and accumulation in the cell membrane increased at an early stage of the ox-LDL treatment and then decreased with prolonged treatment times, especially when foam cells formed. At the same time, after treatment with the same amount of ox-LDL, the intracellular lipid droplets decreased significantly in VSMCs transfected with the caveolin-1 expression plasmid compared with cells transfected without plasmid14. Recently, we found that static pressure significantly decreased caveolin-1 expression in VMSCs cultured in vitro, in pressure- and time-dependent manners19, and that cholesterol accumulation significantly increased in VSMCs [forthcoming data].
Caveolin-1 transports cholesterol out of cells via two models: a vesicle model and a complex model. Caveolin-1 in the endoplasmic reticulum membrane promotes the formation of caveolar vesicles that contain lipids by budding and then covers the vesicle surfaces together with adipophilin20. Po et al21 found that the presence of caveolin-1 with structural mutations on the membranes of intracellular caveolar vesicles triggered the transportation of cholesterol from the cell membrane to endosomes with a decrease in cholesterol synthesis and efflux and an increase in intracellular cholesterol. Therefore, caveolar vesicle-coupled caveolins are very important in regulating the metabolic balance of cholesterol and lipid transportation. Smart's laboratory22 found that caveolin, cyclophilin A, cyclophilin 40 and HSP56 comprise the cholesterol transport complex involved in cholesterol efflux. This complex shuttles back and forth and happens disaggregation-reaggregation between the cytoplasm and cell membranes; and caveolin-1 and cyclophilin A play primary roles in the complex formation of intracellular cholesterol transportation. In brief, these studies suggest that caveolins are the key proteins mediating intracellular cholesterol transportation to cell membranes. Compared to free diffusion, the trafficking system of the caveolin-1 complex may mediate cholesterol efflux with higher efficiency, stronger directionality, and especially more accurate regulation. The caveolin-1-SR-B1 and caveolin-1-ABCA1 coupling transport model is easily regulated by many intracellular and extracellular factors.
Transmembrane transport system of the SR-B1 complex
HDL receptors and their binding proteins include scavenger receptor class B type I (SR-B1), HDL binding protein (HBP), and CD36. SR-B1 is a HDL specific receptor composed of 509 amino acid residues. SR-B1, a horse hoof-like transmembrane glycoprotein, consists of 5 domains: a large extracellular, circular domain composed of 403 amino acid residues with 9 N-terminal-linked glycosylation sites and rich in cysteines, two cytoplasmic domains (amino-terminal domain and carboxy-terminal domain), and two transmembrane domains (N-terminal domain of 28 amino acid residues and C-terminal domain of 25 residues)23.
HDL presents or accepts cholesterol while anchored to plasma membranes via its receptor, SR-B1. The density gradient of cholesterol between HDL and the cell surface determine whether HDL affords or accepts cholesterol; however, the detailed mechanism remains unclear. Further studies have shown that the extracellular domain (ECD) of SR-B1 not only binds to ligands but also forms a hydrophobic channel for the trafficking of cholesterol esters (CE). It has been reported that caveolae present in cell membranes are the initial receiving sites of SR-B1-mediated CE intake, and 80% of CE accumulates in caveolae24. After discriminating HDL, SR-B1 forms a dimer via a leucine zipper region to construct a hydrophobic channel25. CE is transported from HDL to caveolae, and it travels to caveolins through the hydrophobic channel along cholesterol concentration gradients. Caveolae then invaginate into cells and form vesicles containing SR-B1, caveolin and CE26. After CE is transported to other intracellular cholesterol pools, caveolin and SR-B1 return to the cell membrane for the next transportation process. When the monolayer membrane of globelike HDL and the outer layer of the cell membrane fuse, free cholesterol is exchanged between HDL and cells.
SR-B1 mediates not only CE selective uptake (mainly in hepatocytes) but also cholesterol efflux (mainly in perithelial cells). Therefore, SR-B1 knockout aggravates atherosclerotic pathological changes in apo-E-deficient mice. However, when the bone marrow of SR-B1+/+ mice is transplanted into apo-E-deficient mice, the atherosclerotic plaque area decreases, which suggests that SR-B1 may inhibit atherosclerotic pathological changes by promoting cholesterol efflux27.
The RCT capability of SR-B1 depends on the phospholipid content of its ligands. When phospholipase A2 is used to exhaust phosphatidylcholine (PC) in extracellular HDL, intracellular cholesterol efflux declines. Apo-A1 with poor lipid can bind to SR-B1 but does not induce cholesterol efflux28. Moreover, SR-B1-regulated cholesterol efflux is very sensitive to kinds of phospholipids. Both PC- and phosphosphingolipid-enriched HDL promote cholesterol efflux, but the former possesses a stronger capacity to induce intracellular cholesterol efflux29. These data suggest that SR-B1 may facilitate cholesterol uncoupling from the cell membrane and diffusion into maturing HDL.
Transmembrane transport system of the ABC-A1 complex
The ABC gene encodes an intracellular cholesterol-efflux regulatory protein (CERP) and mediates the transmembrane transport of many substances, such as amino acids, proteins, cholesterol, and phospholipids. Currently, the ABC superfamily consists of six families: A, B, C, D, E, and F, with 48 members. ABC-A1 and ABC-G1 are responsible for cholesterol efflux; ABC-G5 and ABC-G8 are responsible for cholesterol secretion from bile duct endothelial cells to the biliary tract. Among the entire superfamily, ABC-A1 plays the most important role in lipid metabolism. Researches on Tangier disease and familial HDL-deficient syndrome revealed the important role of ABC-A1 in regulating the concentration of HDL30, 31.
ABC-A1 is an integrated membrane protein that contains two highly conserved cytoplasmic ATP-binding cassettes (including two pairs of walkers) and two transmembrane domains with 6 transmembrane helices in each domain. Each domain constitutes the wall of the intramembrane fluidity channel that connects the extracellular space and the cytoplasm. ABC-A1 transports intracellular free unesterified cholesterol and phospholipids to extracellular Apo-A1 through the channels using the energy provided by ATP. This process represents one way to form nascent HDL and is thus called the “gate keeper” that regulates cholesterol efflux in perithelial cells32. ABC-A1 cross-links to Apo-A1 via two large extracellular rings. Fitzgerald et al32 used four different mutant constructs to modify the structure of the extracellular rings. Consequently, ABC-A1 lost the capability to form cross-links with apo-A1, which resulted in a decrease in cholesterol efflux33. The direct interaction between ABC-A1 and Apo-A1 is a necessary and decisive step in ABC-A1-mediated cholesterol efflux to HDL, and only intact activity of the triphosadenine kinase of ABC-A1 ensures that Apo-A1 binds to the cell surface to promote cholesterol efflux. Moreover, ABC-A1 is unstable because it can be easily digested by calpain, unsaturated fatty acid, and intracellular cholesterol at toxic levels after the phosphorylation of intracellular PEST (sequence rich in proline, glutamic acid, serine, and threonine). In contrast, PKA- and PKC-induced dephosphorylation of PEST and phosphorylation at Ser-1024 and Ser-2054 increase ABC-A1 stability and cholesterol efflux. ABC-A1-mediated RCT also depends on the presence of lipids and correlates directly with phospholipids within the cellular membrane and cholesterol-enriched domains such as caveolae34.
The ABC-A1-mediated RCT pathway can be described in detail using two working models. One is called the “molecular efflux” model, which is also known as the “two-step transport model”. The first step in this model is the transport of phospholipids from cholesterol-enriched caveolae to apolipoproteins to form an intermediate complex; the second step is the transport of cholesterol from caveolae to the complex so as to form a nascent HDL13. Another model is the so-called “membrane fusion” model, or the “one-step transport” model. ABC-A1 drives the diffusion of cholesterol and phospholipids into lipoproteins to form a complex of phospholipids, cholesterol and lipoproteins, which moves on cellular membranes to take cholesterol35. The first working model shows that the activity of the ABC-A1 phospholipid transferase promotes phospholipid efflux prior to cholesterol transportation during the process of ABC-A1-mediated cholesterol efflux. However, the second model aids in explaining why the deficiency of membrane phospholipid does not happen during cholesterol efflux from macrophages. Both models have their advantages.
Cholesterol accumulation decreases in macrophages located in atherosclerotic plaques in ABC-A1 transgenic animals with high HDL concentrations. In Apo-E knockout mice transfected with the ABC-A1 gene, atherosclerotic plaque areas are significantly decreased. Moreover, we also discovered that oleic acid might decrease ABC-A1 expression. Furthermore, 22-R-hydroxycholesterol stimulates ABC-A1 expression and cholesterol efflux in THP-1-derived foam cells36. These experimental data suggest that ABC-A1 may mediate cholesterol exchanges between the cellular membrane and HDL. Recently, we found that eicosapentaenoic acid negatively affects both ABC-A1 activity and ABC-A1-dependent cholesterol efflux by decreasing ABC-A1 protein levels and by reducing cAMP/PKA-mediated ABC-A1 serine phosphorylation in THP-1 macrophage-derived foam cells37. Additionally, we also found that IFN-γ potentially decreases ABC-A1 expression and cholesterol efflux in THP-1 macrophage-derived foam cells38. NO-1886, a novel compound that functions as an effective lipoprotein lipase (LPL) activator, inhibits atherosclerosis in high-fat/high-sucrose/high-cholesterol-fed Chinese Bama minipigs by increasing the mRNA and protein levels of ABC-A1 in the liver, retroperitoneal adipose tissue and aorta39.
ABC-A1 promotes not only RCT but also intracellular cholesterol transport to the plasma membrane. Neufeld et al40 found that transfection of enhanced green fluorescent proteins (EGFP)-labeled ABC-A1 adenovirus vector into fibroblasts from Tangier patients could recover the ability to transport cholesterol from endosomes to plasma membranes and ABC-A1-mediated cholesterol efflux. After normal fibroblasts transfected with the ABC-A1 adenovirus vector were incubated with fluorescence-labeled Apo-A1, both ABC-A1 and Apo-A1 in endosomes and cholesterol efflux increased, illustrating that ABC-A1 may promote endocytic Apo-A1 release in the form of de novo HDL production. ABC-A1 also affects the construction of the plasma membranes and increases the content of phospholipids and cholesterol in the outer layer of the plasma membrane41.
ABC-A1 mediates the efflux of cellular cholesterol to lipid-poor apolipoproteins but not to HDL particles that constitute the bulk of the plasma HDL. However, ABC-G1 and ABC-G4 mediate isotopic and net mass efflux of cellular cholesterol to HDL. In transfected 293 cells, ABC-G1 and ABC-G4 stimulate cholesterol efflux to both smaller (HDL-3) and larger (HDL-2) subclasses of HDL but not to lipid-poor apo-AI. Treatment of macrophages with a liver X receptor activator causes the upregulation of ABC-G1 and increases cholesterol efflux to HDL. ABC-G1 is highly expressed in macrophages and likely mediates cholesterol efflux from macrophage-derived foam cells to the major HDL fractions42. In contrast to ABC-A1, which specifically couples cholesterol efflux to the acceptor Apo-A143, 44, the efflux activity of ABC-G1 is relatively nonspecific because it can promote efflux not only to HDL but also to LDL and cyclodextrin42. ABC-G1 may traffic to the plasma membrane, and the majority of ABC-G1 has been shown to be intracellular in different cell types45, 46. Whether ABC-G1 is mainly mobilized to the cell surface to support cholesterol efflux45 or regulates intracellular cholesterol distribution remains unclear.
Extracellular HDL trafficking system
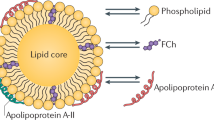
The key step in RCT is the transport of cholesterol from cells to lipoproteins, of which apolipoproteins are an important component. Different lipoproteins have dissimilar types and amounts of apolipoproteins, such as apo-A, apo-B, apo-C, and apo-E. Apo-A1 is the main transporter in HDL and is an important cholesterol receptor that may couple SR-B1 and ABC-A1 to complete the transmembrane transport of cholesterol.
Apo-A1 contains 243 amino acids comprising two amphiprotic α-helices (209-219 and 220-241) that are separated by a proline residue47. The two helices have a high affinity for lipids and can be recognized by SR-B1 and ABC-A1. The amphiprotic helices form a hydrophobic tunnel after binding to SR-B1, which promotes the diffusion of HDL-CE from cells and activates the DG/PKC signaling pathway to promote cholesterol efflux48. After binding to apo-A1, the stability and function of ABC-A1 are increased. When cells are not stimulated or are not loaded with a high content of lipids, apo-A1 activates phosphatidylcholine phospholipase C (PC-PLC) to hydrolyze phospholipids and produce diacylglycerol49. Diacylglycerol activates PKC to phosphorylate ABC-A1, thus increasing the stability of ABC-A1 and cholesterol efflux. However, when too much cholesterol enters cells, ABC-A1 forms a complex with the G protein α subunit (Gαs), which activates adenylate cyclase to produce more cAMP. This process results in the activation of PKA, which phosphorylates ABC-A1 to inhibit ABC-A1 degradation and to promote phospholipid and cholesterol efflux50. Recently, we found that the addition of NO-1886 (0.1 g/kg body weight/day) to the diet of high-fat/high-sucrose/high-cholesterol-fed Chinese Bama minipigs for 5 months significantly reduced atherosclerotic lesions and significantly increased plasma HDL-c and apolipoprotein AI levels39.
Regulatory network of the RCT model
Nuclear factors
There are many nuclear factors, such as sterol regulatory element binding proteins (SREBPs), retinoid X receptors (RXR), liver X receptors (LXRs) and peroxisome proliferator-activated receptors (PPARs), involved in regulating the expression of many RCT-related genes.
SREBPs
SREBPs, a basic-helix-loop-helix leucine zipper class of transcription factors, bind to the sterol regulatory element TCACNCCAC51. As shown in Figure 2, inactivated SREBPs consist of a regulatory subunit and a DNA binding subunit and are located in endoplasmic reticulum membranes. The regulatory subunit is hypersensitive to the concentration of intracellular free cholesterol. When the intracellular sterol level is low, SREBPs are cleaved to a regulatory subunit and a water soluble N-terminal domain (the DNA binding subunit) that is translocated to the nucleus to upregulate the expression of sterol biosynthesis-related genes that possess the sterol regulatory element (SRE) in their promoters52. Sterols in turn inhibit the cleavage of SREBPs and sterol synthesis via a negative feedback loop. SREBPs upregulate the expression levels of SR-B1, LDL receptor (LDLR), cholesterol synthesis enzyme, and HMG-CoA reductase; concomitantly, they downregulate the expression levels of ABC-A1, ABC-G1, ABC-G4, ABC-G5, ABC-G8, and SR-B1. Moreover, SREBPs are regulated by LXR and PPAR-γ53, 54. We found that curcumin inhibited ox-LDL-induced cholesterol accumulation in cultured VSMCs by increasing caveolin-1 expression via inhibition of the nuclear translocation of SREBP-155. Furthermore, Daxx mediates oxidized low-density lipoprotein-induced cholesterol accumulation in macrophages and hepatocytes by downregulating SREBP-1 and upregulating caveolin-156, 57.
Nuclear factors regulate reverse cholesterol transport. The main nuclear factors that regulate RCT include SREBPs, RXR, LXR, and PPARγ. Inactivated SREBPs consist of a regulatory subunit and a DNA binding subunit and are located in endoplasmic reticulum membranes. When the intracellular sterol level is low, SREBPs are cleaved to release a soluble N-terminal domain, also known as a DNA binding subunit, which translocates into the nucleus to upregulate the expression of sterol biosynthesis-related genes that have a sterol regulatory element (SRE) in their promoters. SREBPs also regulate caveolin-1, cholesterol synthesis enzyme, LDL receptor and HMG-CoA reductase. Moreover, SREBPs are regulated by LXR and PPAR-γ. The transcriptional activity of LXR is switched on to activate the exression of many RCT-related genes (such as ABC-A1) following the binding of the LXR and RXR hetero-dimer to the direct repeat response element (DR-4). The main activators of LXR are oxidative sterols and PPAR-γ, whereas RXR is mainly activated by retinoids. PPAR-γ regulates caveolin-1, SR-B1, ABC-A1, CD36, apo-E, and LXR.
RXR and LXR
Many nuclear factors are involved in regulating the extroversive transport of cholesterol, including retinoid X receptor (RXR), liver X receptor (LXR), and peroxisome proliferator-activated receptor (PPAR). As shown in Figure 2, LXR and RXR form a hetero-dimer, which promotes the expression of many RCT-related genes (such as ABC-A1, SR-B1, SREBP) after the dimer binds to the direct repeat response element (DR-4)58, 59. The main activators of LXR and RXR are oxidative sterols, whereas RXR is mainly activated by retinoids. Recently, we found that IFN-γ may first downregulate the expression of LXRα through the JAK/STAT1 signaling pathway and then decrease the expression of ABC-A1 in THP-1 macrophage-derived foam cells38. TGF-β1 upregulates the expression of ABC-A1, ABC-G1 and SR-BI via the LXRα signaling pathway in THP-1 macrophage-derived foam cells60. Additionally, NO-1886 increases the expression of ABC-A1 by upregulating LXRα in the livers of Chinese Bama minipigs39.
PPAR
PPAR family comprises three homeotypic isomerides, PPAR-γ, PPAR-δ, and PPAR-α, which are involved in cholesterol efflux. The natural ligands of PPAR include fatty acids and fatty acid derivatives such as linoleic acid, α-linolenic acid, arachidonic acid, docosahexenoic acid, eicosapentaenoic acid, oleic acid, and elaidic acid.
As shown in Figure 2, PPAR and RXR form a hetero-dimer, which promotes the expression of caveolin-1, SR-B1, ABCA1, ABC-G1, CD36, apo-E, and LXR61, 62, 63, 64, 65. In detail, PPAR-α may directly upregulate the expression of apo-AI and apo-AII and downregulate the expression of apo-CIII, an inhibitor of lipoprotein lipase (LPL), which increases the level of HDL and decreases the level of triglycerides66. PPAR-α may also upregulate ABCA1 expression to promote RCT via two pathways63. In one pathway, PPAR-α promotes the expression of LXR-α to upregulate ABCA1 expression. In the other pathway, PPAR-α activates the cytochrome P450 system to increase the synthesis of oxycholesterol, which is an endogenous ligand of LXR-α, and activate LXR-α. Additionally, PPAR-α promotes RCT by upregulating SR-B164.
PPAR-γ plays an important role in promoting lipocyte differentiation and adipose tissue formation. PPAR-γ upregulates the expression of intracellular fatty acid transportation-, synthesis- and esterification-related genes. PPAR-γ may promote lipid uptake by upregulating CD36 expression and regulate insulin sensitivity by upregulating adiponectin67. However, PPAR-γ does not promote the formation of foam cells. Activated PPAR-γ inhibits the formation of macrophage-derived foam cells and decreases the accumulation of triglycerides in macrophages treated with triglyceride-rich lipoproteins by downregulating SR-A and apo-B4868, 69. PPAR-γ enhances cholesterol efflux and attenuates atherosclerosis by inducing caveolin-1 expression in apo-E-deficient mice70.
Retinoic acid (RA) activates PPAR-δ and the RA receptor71. Fatty acid-binding protein 5 (FABP5) transports RA from the cytosol into the nucleus to activate PPAR-δ, and cellular retinoic acid-binding protein II (CRABPII) transports RA to the nucleus to activate RAR. The ratio of the FABP5/CRABPII concentrations determines which receptor is activated. Activated PPAR-δ induces the expression of genes that affect lipid and glucose homeostasis, including the following: ADRP, a protein associated with lipid droplets; UCP1 and 3, uncoupling proteins; ALDH9, an enzyme that catalyzes carnitine formation (carnitine participates in fatty acid oxidation); and ANGPTL4, a factor necessary for lipid and glucose metabolism72. Currently, evidence is emerging that the PPARbeta/delta isotype is a potential pharmacological target for the treatment of disorders associated with metabolic syndrome73.
Lipid transport proteins
Lipid transport proteins include sterol carrier protein-2 (SCP-2), Niemann-Pick protein C (NPC), cholesteryl ester transfer protein (CETP) and phospholipid transfer protein. Most of these proteins are indirectly involved in the four transfer systems.
SCP-2
Sterol carrier proteins (also known as nonspecific lipid transfer proteins) comprise a family of proteins that transfer steroids and likely also phospholipids and gangliosides between cellular membranes. SCP-2 binds cholesterol with high affinity (Kd near 4 nmol/L), binds plasma membrane caveolin-1, and enhances rapid (detectable in <1 min) directional cholesterol transfer from the plasma membrane to intracellular sites74. The human SCP2 is a basic protein that is believed to participate in the intracellular transport of cholesterol and various other lipids. SCP-2 stimulates the uptake of fatty acid and cholesterol and causes significant activation of acetyl coenzyme A: cholesterol acyltransferase (ACAT)68. The overexpression of SCP-2 may finally inhibit HDL-mediated cholesterol efflux. SCP-2 plays a significant role in HDL-mediated cholesterol efflux by regulating the sizes of the rapid vs slow cholesterol efflux pools75, 76.
NPC
NPC comprises two isomers, NPC1 and NPC2, which monitor the levels of intracellular cholesterol and regulate the transport of cholesterol from late endosomes/lysosomes to other compartment or cell membranes. This process maintains intracellular cholesterol homeostasis through alterations in the transport patterns of vesicles or direct participation in cholesterol transmembrane transportation. NPC1 may promote cholesterol efflux by regulating ABCA1. We found that NO-1886 upregulates NPC1 expression through the LXRα signaling pathway in THP-1 macrophage-derived foam cells77.
CETP
CETP plays an important role in RCT by regulating the extracellular transport system. CETP promotes the exchange of cholesterol esters (CE) and triglycerides78 among lipoproteins. Examples of this are the transportation of CE from HDL to VLDL, chylomicra (CM) or their cruel granules, and eventually to LDL, as well as the transportation of TG from VLDL or CM to HDL. CETP transports CE to TG-rich lipoprotein, followed by CE efflux to granules and to LDL. Liver receptors complete the process of CE uptake79.
PLTP
PLTP, which is one of at least two lipid transfer proteins found in human plasma, primarily regulates the extracellular transfer system of HDL. PLTP interacts with Apo-A1 and Apo-Apo-A280. It transfers phospholipids from triglyceride-rich lipoproteins to HDL. In addition to regulating the size of HDL particles, this protein may be involved in cholesterol metabolism. At least two transcript variants encoding different isoforms have been discovered for this gene.
Cholesterol synthesis enzymes
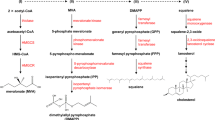
Cholesterol synthesis enzymes include cytochrome P450, family 27, subfamily A, polypeptide 1 (CYP27A1), lipoprotein lipase (LPL), lecithin:cholesterol acyltransferase (LCAT) and acyl-CoA:cholesterol acyltransferase (ACAT). They are mainly responsible for cholesterol synthesis and metabolism.
CYP27A1
Mitochondrial sterol 27-hydroxylase (CYP27A1), a cytochrome P450 oxidase, is expressed in the liver, peripheral tissues, and macrophages and catalyzes oxidative cleavage of the sterol side chain in the bile acid biosynthetic pathway in the liver and 27-hydroxylation of cholesterol in most tissues81. The 27-hydroxycholesterol (27-HOC) activates LXRα and induces the cholesterol efflux transporters ABC-A1 and ABC-G1 in macrophages, and therefore CYP27A1 may increase intracellular 27-HOC levels, and induce ABC-A1 and ABC-G1 expression, and finally stimulate cholesterol efflux82. Partial or complete deficiency of CYP27A1 leads to abnormal cholesterol and cholestanol (reduced form of cholesterol) accumulation in multiple cells and tissues and is often manifested by premature atherosclerosis83. Conversely, CYP27A1 introduction into cells stimulates cholesterol efflux, and therefore, it may facilitate protection against atherosclerosis84.
LPL
LPL, an enzyme that decomposes endogenous TG in the blood circulation (especially TG in the CM and VLDL), transfers cholesterol, phospholipids and apolipoproteins between lipoproteins. Free fatty acids, which are one of the decomposition products of TG, provide energy for tissues or are re-esterified to form TG, which is stored in adipose tissues. Catalyzed by LPL, VLDL is transformed to LDL, and CM is utilized to produce de novo HDL following the loss of CM surface lipids85, 86. We found that NO-1886, which is an effective activator of lipoprotein lipase, could increase the activity of LPL in rats fed a diet rich in cholesterol, leading to a decrease in plasma TG and an increase in HDL-c and thus inhibiting the pathological changes associated with atherosclerosis in the coronary artery87, 88.
LCAT
LCAT catalyzes the transesterification of phospholipid acyl chains to unesterified cholesterol (UC) to synthesize CE. Activation of LCAT by Apo-AI on nascent (discoidal) HDL is essential for the formation of mature (spheroidal) HDL during the antiatherogenic process of reverse cholesterol transport89. LCAT converts a disk-like and a small globular de novo HDL (HDL3) to mature globular HDL (HDL2), and therefore, the concentration of free cholesterol in HDL decreases. In addition, a concentration gradient of cholesterol from the cell membrane to the plasma lipoprotein forms, which results in reverse cholesterol transportation.
ACAT
ACAT is an intracellular protein located in the endoplasmic reticulum that forms cholesterol esters from cholesterol. ACAT, which is also known as sterol O-acyltransferase (SOAT), belongs to the class of enzymes known as acyltransferases that transfer fatty acyl groups between molecules. ACAT, which is an important enzyme in bile acid biosynthesis, catalyzes the intracellular esterification of cholesterol. ACAT-mediated esterification of cholesterol limits its solubility in cell membrane lipids and thus promotes the accumulation of cholesterol esters in fat droplets within the cytoplasm. This process is important because it prevents the toxic accumulation of free cholesterol in various cell membrane fractions90. Most of the cholesterol absorbed during intestinal transport undergoes ACAT-mediated esterification prior to incorporation into chylomicrons. ACAT also plays an important role in the formation of foam cells and atherosclerosis by participating in the accumulation of cholesterol esters in macrophages and vascular tissue91. The rate-controlling enzyme in cholesterol catabolism, hepatic cholesterol 7-hydroxylase, is believed to be regulated in part by ACAT.
Other regulatory proteins
Immunophilin family
Cyclophilins are proteins that bind to cyclosporine, an immunosuppressant that is often used to suppress rejection after internal organ transplantation. These proteins possess peptidyl prolyl isomerase activity, which catalyzes the isomerism of peptide bonds from trans to cis form at proline residues and facilitates protein folding. Cyclophilins, including cyclophilin A, cyclophilin B, cyclophilin D, and cyclophilin 40, are responsible for cell growth, proliferation, migration and intracellular ion homeostasis, and they regulate stress-related genes and chaperone activity92, 93, 94. Cyclophilin A and cyclophilin 40 are involved in intracellular cholesterol transportation. Cyclophilin A, an 18 kDa protein, contains one domain that binds to caveolins and another domain that binds to heat shock proteins95. Cyclophilin A, a peptidel prolyl cis-trans isomerase (PPIase) that catalyzes the cis-trans isomerization of prolines and participates in protein folding, is an important inhibitor of calcineurin96. Activated calcineurin suppresses the expression and activity of PPAR-γ, a regulator of the expression of SR-B1, ABC-A1 and CD36. Cyclosporin A, an inhibitor of cyclophilin A, suppresses apo-A1/HDL-induced RCT97. Therefore, cyclophilin A participates not only in the formation of the caveolin-1-cholesterol transport complex but also in the regulation of SR-B1- and ABC-A1-mediated cholesterol transmembrane transportation.
Adipophilin
Adipophilin, or adipose differentiation-related protein (ADRP)98, is the most important membrane protein that coats lipid droplets in foam cells. Incubation of macrophages with very low-density lipoprotein (VLDL) dramatically increases cellular triglyceride content to similar extents in control and adipophilin-overexpressing cells. The lipid droplet content of macrophages is increased by overexpression of adipophilin and/or VLDL loading. In contrast, the inhibition of adipophilin expression using siRNA prevents lipid droplet formation and significantly reduces intracellular triglyceride content. Adipophilin elevates cellular lipid levels via inhibition of β-oxidation and stimulation of long-chain fatty acid incorporation into triglycerides. Adipophilin expression in THP-1 macrophages alters the cellular content of different lipids and increases the size of lipid droplets, consistent with the role of adipophilin in human foam cell formation99. It has been recently reported that adipophilin blocks lipid efflux and increases lipid accumulation in THP-1 macrophages independently of the expression of lipid efflux-related genes100. Adipophilin expression is regulated by the PPAR-γ/RXR dimer101.
Phospholipids
Margarita et al102 found that cholesterol efflux from cells increased when the cells were treated with neutral lipidosomes containing a phospholipid bilayer as well as apolipoprotein, although the lipidosomes did not bind to SR-B1 and ABC-A1 in the cell membrane. This cholesterol efflux to lipoprotein was blocked by an anti-phospholipid antibody that prevents the fusion of lipidosomes with cells, suggesting that phospholipids play an important role in cholesterol efflux.
cAMP, PKC and PKA are also involved in regulating the extroversive transport of cholesterol mediated by the ABC-A1 signaling pathway103, 104. We have found that eicosapentaenoic acid reduces ABC-A1 serine phosphorylation through the cAMP/PKA signaling pathway37.
Discussion
By summarizing the latest international studies together with our findings, we propose the novel working model of “four systems and one center with coupling transportation and networking regulation”, which consists of the intracellular trafficking system of the caveolin-1 complex, the transmembrane transport system of the ABC-A1 complex, the transmembrane transport system of the SR-B1 complex, the extracellular trafficking system of HDL/Apo-A1 and the caveolar transport center. Thus, this model clarifies the most important proteins and their interrelations in RCT. This model represents a common rule for almost all cells; however, the systems contained in this model comprise different proteins/factors in different cells. For example, CD36 plays the role of SR-B1, and ABC-G1 plays the role of ABC-A1 in some cells. In this review, we focus on VSMCs and macrophages, which are the most important original cells of foam cells.
The intracellular trafficking system of the caveolin-1 complex generally consists of caveolin-1, cyclophilin A, cyclophilin 40, HSP56 and other unknown proteins, and it participates in cholesterol trafficking from the cytosol to cell membranes. Caveolin-1 expression is upregulated by PPAR but downregulated by SREBPs62, 105, 106. Recently, it has been shown that annexin A2 (ANX-A2), which is part of family of calcium- and phospholipid-binding proteins, may also be involved in the formation of the caveolin-1 complex during the process of cholesterol efflux107. The caveolin-1 complex transfers cholesterol to transmembrane transport systems of the SR-B1 and ABC-A1 complexes. First, caveolin-1 and cyclophilin A bind to SR-B1 or ABC-A1108 and present cholesterol to the two transmembrane transport systems. Finally, they go back to the intracellular transport complex. The receptors of the SR-B1 and ABC-A1 systems are lipid-rich apolipoproteins such as HDL. The SR-B1 system functions bidirectionally via uptake of cholesterol ester and efflux of free cholesterol. The direction of transportation is determined by the transmembrane concentration gradient of cholesterol. SR-B1 is regulated by PPAR/RXR, LXR/RXR, and SREBPs54, 64. However, ABC-A1 mainly transfers cholesterol uni-directionally outside of cells using the energy provided by ATP. ABC-A1 is upregulated by PPAR/RXR, LXR/RXR, cAMP, and HDL and downregulated by SREBPs38, 63, 104, 109. In general, there is a one-way and a two-way transmembrane transport system in almost all cells, but the components of the systems differ in different cells. The extracellular trafficking system of HDL, which consists of HDL, LCAT, CETP, and LPL, mainly accepts cholesterol transported by the transmembrane transport systems. The cholesterol is finally transported to the liver via the HDL system.
Caveolae present in the cell membrane function as storage and transport centers of cholesterol. Caveolin-1, ABC-A1, and SR-B1 in the three transport systems mentioned above are rich in caveolae. The transmembrane transportation of cholesterol mediated by caveolin-1, ABC-A1, SR-B1, and HDL takes place in caveolae, which are the most important centers of cholesterol transportation. In brief, the coupled trafficking of cholesterol happens among the four transport systems in caveolae. The intracellular transport system of the caveolin-1 complex transports cholesterol from the cytosol to cell membranes and stores the cholesterol in caveolae. Then the transmembrane transport systems of SR-B1 and ABC-A1 transport the cholesterol to HDL, and finally to the liver for metabolism. In addition, we propose that cholesterol exchange may happen within the cell membrane between the SR-B1 and ABC-A1 complex, especially when cholesterol in one transmembrane transport system is more than that in another transmembrane transport system, as shown in Figure 1.
Both the caveolin-1/SR-B1/HDL and the caveolin-1/ABC-A1/HDL pathways co-regulate the transmembrane transportation of cholesterol. Many factors regulate these two pathways, such as the HDL size, the cholesterol content of the HDL and the blood serum concentration of total cholesterol. When the content of cholesterol in the plasma membrane exceeds 20%, SR-B1-dependent cholesterol efflux is blocked; when the ABC-A1 level decreases by 80%, ABC-A1-dependent cholesterol efflux is blocked110. Yancey et al104 reported that when endothelial lipase was overexpressed in apo-A1 transgenic mice, the phospholipid/apo-A1 ratio, total cholesterol and HDL in the serum decreased by 60%, 89%, and 91%, respectively. In addition, ABC-A1-mediated cholesterol efflux increased by 63%, and SR-B1-induced cholesterol efflux decreased by 90%. In contrast, when phosphatidylserine phospholipase was overexpressed, the reverse results were observed. Apo-A1 is involved not only in the transmembrane transportation of cholesterol but also in the transport of intracellular and extracellular cholesterol. After binding to the HDL receptor, the complex consisting of Apo-A1 and phospholipids triggers the transfer of free cholesterol from the intracellular cholesterol pool to the cell membrane97.
The regulation network controlling the four transport systems is very complex. Many proteins are involved in regulating the four transport systems of cholesterol efflux, including nuclear factors, lipid transportation proteins, cholesterol synthesis enzymes, the immunophilin family, adipophilin, phospholipids, etc. Different proteins participate in regulating cholesterol efflux in different cells.
This model of reverse cholesterol transportation illustrates almost all of the cholesterol efflux pathways and facilitates our understanding of the mechanism responsible for cholesterol efflux from lipid-loaded cells. This model could promote the development of targets to prevent and treat atherosclerosis.
References
Lusis AJ, Fogelman AM, Fonarow GC . Genetic basis of atherosclerosis: part II: clinical implications. Circulation 2004; 110: 2066–71.
Hansson GK . Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med 2005; 352: 1685–95.
Lusis AJ, Fogelman AM, Fonarow GC . Genetic basis of atherosclerosis: part I: new genes and pathways. Circulation 2004; 110: 1868–73.
Ory DS . Nuclear receptor signaling in the control of cholesterol homeostasis: have the orphans found a home? Circ Res 2004; 95: 660–70.
Payne AH, Hales DB . Overview of steroidogenic enzymes in the pathway from cholesterol to active steroid hormones. Endocr Rev 2004; 25: 947–70.
von Bergmann K, Sudhop T, Lutjohann D . Cholesterol and plant sterol absorption: recent insights. Am J Cardiol 2005; 96: 10D–14D.
Martin S, Parton RG . Caveolin, cholesterol, and lipid bodies. Semin Cell Dev Biol 2005; 16: 163–74.
Fielding CJ, Fielding PE . Caveolae and intracellular trafficking of cholesterol. Adv Drug Deliv Rev 2001; 49: 251–64.
Fielding PE, Fielding CJ . Intracellular transport of low density lipoprotein derived free cholesterol begins at clathrin-coated pits and terminates at cell surface caveolae. Biochemistry 1996; 35: 14932–8.
Sun SW, Zu XY, Tuo QH, Zhu BY, Chen LX, Lei XY, et al. Caveolae and caveolin-1 mediated the endocytosis and transcytosis of oxidized low density lipoprotein in endothelial cells. Acta Pharmacol Sin 2010; 31: 1336–42.
Fielding CJ, Bist A, Fielding PE . Caveolin mRNA levels are up-regulated by free cholesterol and down-regulated by oxysterols in fibroblast monolayers. Proc Natl Acad Sci USA 1997; 94: 3753–8.
Fielding PE, Fielding CJ . Plasma membrane caveolae mediate the efflux of cellular free cholesterol. Biochemistry 1995; 34: 14288–92.
Fielding PE, Nagao K, Hakamata H, Chimini G, Fielding CJ . A two-step mechanism for free cholesterol and phospholipid efflux from human vascular cells to apolipoprotein A-1. Biochemistry 2000; 39: 14113–20.
Yan PK, Liao DF, Yang YZ . Influence of caveolin-1 on cholesterol efflux in vascular smooth muscle cells. Chin J Arterioscler 2002; 10: 379–83.
Schlegel A, Lisanti MP . A molecular dissection of caveolin-1 membrane attachment and oligomerization. Two separate regions of the caveolin-1 C-terminal domain mediate membrane binding and oligomer/oligomer interactions in vivo. J Biol Chem 2000; 275: 21605–17.
Razani B, Woodman SE, Lisanti MP . Caveolae: from cell biology to animal physiology. Pharmacol Rev 2002; 54: 431–67.
Uittenbogaard A, Smart EJ . Palmitoylation of caveolin-1 is required for cholesterol binding, chaperone complex formation, and rapid transport of cholesterol to caveolae. J Biol Chem 2000; 275: 25595–9.
Fu Y, Hoang A, Escher G, Parton RG, Krozowski Z, Sviridov D . Expression of caveolin-1 enhances cholesterol efflux in hepatic cells. J Biol Chem 2004; 279: 14140–6.
Luo DX, Cheng J, Xiong Y, Li J, Xia C, Xu C, et al. Static pressure drives proliferation of vascular smooth muscle cells via caveolin-1/ERK1/2 pathway. Biochem Biophys Res Commun 2010; 391: 1693–7.
Brown DA . Lipid droplets: proteins floating on a pool of fat. Curr Biol 2001; 11: R446–9.
Pol A, Luetterforst R, Lindsay M, Heino S, Ikonen E, Parton RG . A caveolin dominant negative mutant associates with lipid bodies and induces intracellular cholesterol imbalance. J Cell Biol 2001; 152: 1057–70.
Uittenbogaard A, Ying Y, Smart EJ . Characterization of a cytosolic heat-shock protein-caveolin chaperone complex. Involvement in cholesterol trafficking. J Biol Chem 1998; 273: 6525–32.
Rhainds D, Brissette L . The role of scavenger receptor class B type I (SR-BI) in lipid trafficking. defining the rules for lipid traders. Int J Biochem Cell Biol 2004; 36: 39–77.
Mulcahy JV, Riddell DR, Owen JS . Human scavenger receptor class B type II (SR-BII) and cellular cholesterol efflux. Biochem J 2004; 377: 741–7.
Gu X, Trigatti B, Xu S, Acton S, Babitt J, Krieger M . The efficient cellular uptake of high density lipoprotein lipids via scavenger receptor class B type I requires not only receptor-mediated surface binding but also receptor-specific lipid transfer mediated by its extracellular domain. J Biol Chem 1998; 273: 26338–48.
Frank PG, Marcel YL, Connelly MA, Lublin DM, Franklin V, Williams DL, et al. Stabilization of caveolin-1 by cellular cholesterol and scavenger receptor class B type I. Biochemistry 2002; 41: 11931–40.
Zhang W, Yancey PG, Su YR, Babaev VR, Zhang Y, Fazio S, et al. Inactivation of macrophage scavenger receptor class B type I promotes atherosclerotic lesion development in apolipoprotein E-deficient mice. Circulation 2003; 108: 2258–63.
de la Llera-Moya M, Rothblat GH, Connelly MA, Kellner-Weibel G, Sakr SW, Phillips MC, et al. Scavenger receptor BI (SR-BI) mediates free cholesterol flux independently of HDL tethering to the cell surface. J Lipid Res 1999; 40: 575–80.
Yancey PG, Bortnick AE, Kellner-Weibel G, de la Llera-Moya M, Phillips MC, Rothblat GH . Importance of different pathways of cellular cholesterol efflux. Arterioscler Thromb Vasc Biol 2003; 23: 712–9.
Soro-Paavonen A, Westerbacka J, Ehnholm C, Taskinen MR . Metabolic syndrome aggravates the increased endothelial activation and low-grade inflammation in subjects with familial low HDL. Ann Med 2006; 38: 229–38.
Stefkova J, Poledne R, Hubacek JA . ATP-binding cassette (ABC) transporters in human metabolism and diseases. Physiol Res 2004; 53: 235–43.
Oram JF, Lawn RM . ABCA1. The gatekeeper for eliminating excess tissue cholesterol. J Lipid Res 2001; 42: 1173–9.
Fitzgerald ML, Morris AL, Rhee JS, Andersson LP, Mendez AJ, Freeman MW . Naturally occurring mutations in the largest extracellular loops of ABCA1 can disrupt its direct interaction with apolipoprotein A-I. J Biol Chem 2002; 277: 33178–87.
Drobnik W, Borsukova H, Bottcher A, Pfeiffer A, Liebisch G, Schutz GJ, et al. Apo AI/ABCA1-dependent and HDL3-mediated lipid efflux from compositionally distinct cholesterol-based microdomains. Traffic 2002; 3: 268–78.
Gillotte KL, Zaiou M, Lund-Katz S, Anantharamaiah GM, Holvoet P, Dhoest A, et al. Apolipoprotein-mediated plasma membrane microsolubilization. Role of lipid affinity and membrane penetration in the efflux of cellular cholesterol and phospholipid. J Biol Chem 1999; 274: 2021–8.
Tang CK, Yang JH, Yi GH, Wang Z, Liu LS, Wan ZY, et al. Effects of oleate on ATP binding cassette transporter A1 expression and cholesterol efflux in THP-1 macrophage-derived foam cells. Sheng Wu Hua Xue Yu Sheng Wu Wu Li Xue Bao (Shanghai) 2003; 35: 1077–82.
Hu YW, Ma X, Li XX, Liu XH, Xiao J, Mo ZC, et al. Eicosapentaenoic acid reduces ABCA1 serine phosphorylation and impairs ABCA1-dependent cholesterol efflux through cyclic AMP/protein kinase A signaling pathway in THP-1 macrophage-derived foam cells. Atherosclerosis 2009; 204: e35–43.
Hao XR, Cao DL, Hu YW, Li XX, Liu XH, Xiao J, et al. IFN-gamma down-regulates ABCA1 expression by inhibiting LXRalpha in a JAK/STAT signaling pathway-dependent manner. Atherosclerosis 2009; 203: 417–28.
Zhang C, Yin W, Liao D, Huang L, Tang C, Tsutsumi K, et al. NO-1886 upregulates ATP binding cassette transporter A1 and inhibits diet-induced atherosclerosis in Chinese Bama minipigs. J Lipid Res 2006; 47: 2055–63.
Neufeld EB, Stonik JA, Demosky SJ Jr, Knapper CL, Combs CA, Cooney A, et al. The ABCA1 transporter modulates late endocytic trafficking: insights from the correction of the genetic defect in Tangier disease. J Biol Chem 2004; 279: 15571–8.
Smith JD, Waelde C, Horwitz A, Zheng P . Evaluation of the role of phosphatidylserine translocase activity in ABCA1-mediated lipid efflux. J Biol Chem 2002; 277: 17797–803.
Wang N, Lan D, Chen W, Matsuura F, Tall AR . ATP-binding cassette transporters G1 and G4 mediate cellular cholesterol efflux to high-density lipoproteins. Proc Natl Acad Sci USA 2004; 101: 9774–9.
Vedhachalam C, Ghering AB, Davidson WS, Lund-Katz S, Rothblat GH, Phillips MC . ABCA1-induced cell surface binding sites for ApoA-I. Arterioscler Thromb Vasc Biol 2007; 27: 1603–9.
Vedhachalam C, Duong PT, Nickel M, Nguyen D, Dhanasekaran P, Saito H, et al. Mechanism of ATP-binding cassette transporter A1-mediated cellular lipid efflux to apolipoprotein A-I and formation of high density lipoprotein particles. J Biol Chem 2007; 282: 25123–30.
Wang N, Ranalletta M, Matsuura F, Peng F, Tall AR . LXR-induced redistribution of ABCG1 to plasma membrane in macrophages enhances cholesterol mass efflux to HDL. Arterioscler Thromb Vasc Biol 2006; 26: 1310–6.
Tarr PT, Edwards PA . ABCG1 and ABCG4 are coexpressed in neurons and astrocytes of the CNS and regulate cholesterol homeostasis through SREBP-2. J Lipid Res 2008; 49: 169–82.
Bocharov AV, Baranova IN, Vishnyakova TG, Remaley AT, Csako G, Thomas F, et al. Targeting of scavenger receptor class B type I by synthetic amphipathic alpha-helical-containing peptides blocks lipopolysaccharide (LPS) uptake and LPS-induced pro-inflammatory cytokine responses in THP-1 monocyte cells. J Biol Chem 2004; 279: 36072–82.
Yamauchi Y, Hayashi M, Abe-Dohmae S, Yokoyama S . Apolipoprotein A-I activates protein kinase C alpha signaling to phosphorylate and stabilize ATP binding cassette transporter A1 for the high density lipoprotein assembly. J Biol Chem 2003; 278: 47890–7.
Sviridov D, Fidge N, Beaumier-Gallon G, Fielding C . Apolipoprotein A-I stimulates the transport of intracellular cholesterol to cell-surface cholesterol-rich domains (caveolae). Biochem J 2001; 358: 79–86.
Haidar B, Denis M, Marcil M, Krimbou L, Genest J Jr . Apolipoprotein A-I activates cellular cAMP signaling through the ABCA1 transporter. J Biol Chem 2004; 279: 9963–9.
Yokoyama C, Wang X, Briggs MR, Admon A, Wu J, Hua X, et al. SREBP-1, a basic-helix-loop-helix-leucine zipper protein that controls transcription of the low density lipoprotein receptor gene. Cell 1993; 75: 187–97.
Wang X, Sato R, Brown MS, Hua X, Goldstein JL . SREBP-1, a membrane-bound transcription factor released by sterol-regulated proteolysis. Cell 1994; 77: 53–62.
Sato R . SREBPs: protein interaction and SREBPs. FEBS J 2009; 276: 622–7.
Shimano H . SREBPs: novel aspects of SREBPs in the regulation of lipid synthesis. FEBS J 2009; 276: 615.
Yuan HY, Kuang SY, Zheng X, Ling HY, Yang YB, Yan PK, et al. Curcumin inhibits cellular cholesterol accumulation by regulating SREBP-1/caveolin-1 signaling pathway in vascular smooth muscle cells. Acta Pharmacol Sin 2008; 29: 555–63.
Tuo QH, Liang L, Zhu BY, Cao X, Liao DF . Effect of Daxx on cholesterol accumulation in hepatic cells. World J Gastroenterol 2008; 14: 435–40.
He QZ, Zeng HC, Tuo QH, Zhu BY, Rang WQ, Tang XQ, et al. Daxx mediates oxidized low-density lipoprotein-induced cholesterol accumulation and apoptosis in macrophages by upregulating caveolin-1 expression. Prog Biochem Biophysic 2010; in publish.
Venkateswaran A, Laffitte BA, Joseph SB, Mak PA, Wilpitz DC, Edwards PA, et al. Control of cellular cholesterol efflux by the nuclear oxysterol receptor LXR alpha. Proc Natl Acad Sci USA 2000; 97: 12097–102.
Repa JJ, Turley SD, Lobaccaro JA, Medina J, Li L, Lustig K, et al. Regulation of absorption and ABC1-mediated efflux of cholesterol by RXR heterodimers. Science 2000; 289: 1524–9.
Hu YW, Wang Q, Ma X, Li XX, Liu XH, Xiao J, et al. TGF-beta1 up-regulates expression of ABCA1, ABCG1 and SR-BI through liver X receptor alpha signaling pathway in THP-1 macrophage-derived foam cells. J Atheroscler Thromb.
Ricote M, Li AC, Willson TM, Kelly CJ, Glass CK . The peroxisome proliferator-activated receptor-gamma is a negative regulator of macrophage activation. Nature 1998; 391: 79–82.
Llaverias G, Vazquez-Carrera M, Sanchez RM, Noe V, Ciudad CJ, Laguna JC, et al. Rosiglitazone upregulates caveolin-1 expression in THP-1 cells through a PPAR-dependent mechanism. J Lipid Res 2004; 45: 2015–24.
Ogata M, Tsujita M, Hossain MA, Akita N, Gonzalez FJ, Staels B, et al. On the mechanism for PPAR agonists to enhance ABCA1 gene expression. Atherosclerosis 2009; 205: 413–9.
Ahmed RA, Murao K, Imachi H, Yu X, Li J, Wong NC, et al. Human scavenger receptor class B type 1 is regulated by activators of peroxisome proliferators-activated receptor-gamma in hepatocytes. Endocrine 2009; 35: 233–42.
Tao H, Aakula S, Abumrad NN, Hajri T . Peroxisome proliferator-activated receptor gamma regulates the expression and function of very low density lipoprotein receptor. Am J Physiol Endocrinol Metab 2010; 298: E68–79.
Ziouzenkova O, Plutzky J . Retinoid metabolism and nuclear receptor responses: New insights into coordinated regulation of the PPAR-RXR complex. FEBS Lett 2008; 582: 32–8.
Lim HJ, Lee S, Lee KS, Park JH, Jang Y, Lee EJ, et al. PPARgamma activation induces CD36 expression and stimulates foam cell like changes in rVSMCs. Prostaglandins Other Lipid Mediat 2006; 80: 165–74.
Haraguchi G, Kobayashi Y, Brown ML, Tanaka A, Isobe M, Gianturco SH, et al. PPAR(alpha) and PPAR(gamma) activators suppress the monocyte-macrophage apoB-48 receptor. J Lipid Res 2003; 44: 1224–31.
Ditiatkovski M, Toh BH, Bobik A . GM-CSF deficiency reduces macrophage PPAR-gamma expression and aggravates atherosclerosis in ApoE-deficient mice. Arterioscler Thromb Vasc Biol 2006; 26: 2337–44.
Hu Q, Zhang XJ, Liu CX, Wang XP, Zhang Y . PPARgamma1-induced caveolin-1 enhances cholesterol efflux and attenuates atherosclerosis in apolipoprotein E-deficient mice. J Vasc Res 2010; 47: 69–79.
Graf GA, Li WP, Gerard RD, Gelissen I, White A, Cohen JC, et al. Coexpression of ATP-binding cassette proteins ABCG5 and ABCG8 permits their transport to the apical surface. J Clin Invest 2002; 110: 659–69.
Wolf G . Retinoic acid activation of peroxisome proliferation-activated receptor delta represses obesity and insulin resistance. Nutr Rev 2010; 68: 67–70.
Coll T, Rodriguez-Calvo R, Barroso E, Serrano L, Eyre E, Palomer X, et al. Peroxisome proliferator-activated receptor (PPAR) beta/delta: a new potential therapeutic target for the treatment of metabolic syndrome. Curr Mol Pharmacol 2009; 2: 46–55.
Parr RD, Martin GG, Hostetler HA, Schroeder ME, Mir KD, Kier AB, et al. A new N-terminal recognition domain in caveolin-1 interacts with sterol carrier protein-2 (SCP-2). Biochemistry 2007; 46: 8301–14.
Storey SM, Atshaves BP, McIntosh AL, Landrock KK, Martin GG, Huang H, et al. Effect of sterol carrier protein-2 gene ablation on HDL-mediated cholesterol efflux from primary cultured mouse hepatocytes. Am J Physiol Gastrointest Liver Physiol 2010; 299: G244–54.
Schroeder F, Huang H, McIntosh AL, Atshaves BP, Martin GG, Kier AB . Caveolin, sterol carrier protein-2, membrane cholesterol-rich microdomains and intracellular cholesterol trafficking. Subcell Biochem 2010; 51: 279–318.
Ma X, Hu YW, Mo ZC, Li XX, Liu XH, Xiao J, et al. NO-1886 up-regulates Niemann-Pick C1 protein (NPC1) expression through liver X receptor alpha signaling pathway in THP-1 macrophage-derived foam cells. Cardiovasc Drugs Ther 2009; 23: 199–206.
Repetto S, Bado M, Broda P, Lucania G, Masetti E, Sotgia F, et al. Increased number of caveolae and caveolin-3 overexpression in Duchenne muscular dystrophy. Biochem Biophys Res Commun 1999; 261: 547–50.
Oliver WR Jr, Shenk JL, Snaith MR, Russell CS, Plunket KD, Bodkin NL, et al. A selective peroxisome proliferator-activated receptor delta agonist promotes reverse cholesterol transport. Proc Natl Acad Sci U S A 2001; 98: 5306–11.
Kujiraoka T, Nanjee MN, Oka T, Ito M, Nagano M, Cooke CJ, et al. Effects of intravenous apolipoprotein A-I/phosphatidylcholine discs on LCAT, PLTP, and CETP in plasma and peripheral lymph in humans. Arterioscler Thromb Vasc Biol 2003; 23: 1653–9.
Pikuleva IA . Cholesterol-metabolizing cytochromes P450: implications for cholesterol lowering. Expert Opin Drug Metab Toxicol 2008; 4: 1403–14.
Li T, Chen W, Chiang JY . PXR induces CYP27A1 and regulates cholesterol metabolism in the intestine. J Lipid Res 2007; 48: 373–84.
Bjorkhem I BK, Leitersdorf E . Inborn errors in bile acid biosynthesis and storage of sterols other than cholesterol. In: The metabolic and molecular bases of inherited disease. New York: McGraw-Hill; 1995. p2073–2099.
Escher G, Krozowski Z, Croft KD, Sviridov D . Expression of sterol 27-hydroxylase (CYP27A1) enhances cholesterol efflux. J Biol Chem 2003; 278: 11015–9.
Hu L, van der Hoogt CC, Espirito Santo SM, Out R, Kypreos KE, van Vlijmen BJ, et al. The hepatic uptake of VLDL in lrp−ldlr−/−vldlr−/− mice is regulated by LPL activity and involves proteoglycans and SR-BI. J Lipid Res 2008; 49: 1553–61.
Lichtenstein L, Berbee JF, van Dijk SJ, van Dijk KW, Bensadoun A, Kema IP, et al. Angptl4 upregulates cholesterol synthesis in liver via inhibition of LPL- and HL-dependent hepatic cholesterol uptake. Arterioscler Thromb Vasc Biol 2007; 27: 2420–7.
Yin W, Liao D, Wang Z, Xi S, Tsutsumi K, Koike T, et al. NO-1886 inhibits size of adipocytes, suppresses plasma levels of tumor necrosis factor-alpha and free fatty acids, improves glucose metabolism in high-fat/high-sucrose-fed miniature pigs. Pharmacol Res 2004; 49: 199–206.
Yin W, Liao D, Kusunoki M, Xi S, Tsutsumi K, Wang Z, et al. NO-1886 decreases ectopic lipid deposition and protects pancreatic beta cells in diet-induced diabetic swine. J Endocrinol 2004; 180: 399–408.
Jones MK, Catte A, Li L, Segrest JP . Dynamics of activation of lecithin:cholesterol acyltransferase by apolipoprotein A-I. Biochemistry 2009; 48: 11196–210.
Kusunoki J, Aragane K, Kitamine T, Yamaura T, Ohnishi H . Effect of F-1394, a potent and selective inhibitor of acyl-CoA:cholesterol acyltransferase (ACAT), on esterification of cholesterol and basolateral secretion of cholesteryl ester in Caco-2 cells. Nippon Yakurigaku Zasshi 1997; 110: 357–65.
Lei L, Xiong Y, Chen J, Yang JB, Wang Y, Yang XY, et al. TNF-alpha stimulates the ACAT1 expression in differentiating monocytes to promote the CE-laden cell formation. J Lipid Res 2009; 50: 1057–67.
Deligny A, Denys A, Marcant A, Melchior A, Mazurier J, van Kuppevelt TH, et al. Synthesis of heparan sulfate with cyclophilin B-binding properties is determined by cell type-specific expression of sulfotransferases. J Biol Chem 285: 1701–15.
Carreira RS, Lee Y, Ghochani M, Gustafsson AB, Gottlieb RA . Cyclophilin D is required for mitochondrial removal by autophagy in cardiac cells. Autophagy 2010; 6: 462–72.
Bonfils C, Bec N, Larroque C, Del Rio M, Gongora C, Pugniere M, et al. Cyclophilin A as negative regulator of apoptosis by sequestering cytochrome c. Biochem Biophys Res Commun 2010; 393; 325–30.
Kallen J, Walkinshaw MD . The X-ray structure of a tetrapeptide bound to the active site of human cyclophilin A. FEBS Lett 1992; 300: 286–90.
Takahashi N, Hayano T, Suzuki M . Peptidyl-prolyl cis-trans isomerase is the cyclosporin A-binding protein cyclophilin. Nature 1989; 337: 473–5.
Ito J, Nagayasu Y, Kato K, Sato R, Yokoyama S . Apolipoprotein A-I induces translocation of cholesterol, phospholipid, and caveolin-1 to cytosol in rat astrocytes. J Biol Chem 2002; 277: 7929–35.
Persson J, Degerman E, Nilsson J, Lindholm MW . Perilipin and adipophilin expression in lipid loaded macrophages. Biochem Biophys Res Commun 2007; 363: 1020–6.
Larigauderie G, Cuaz-Perolin C, Younes AB, Furman C, Lasselin C, Copin C, et al. Adipophilin increases triglyceride storage in human macrophages by stimulation of biosynthesis and inhibition of beta-oxidation. Febs J 2006; 273: 3498–510.
Larigauderie G, Furman C, Jaye M, Lasselin C, Copin C, Fruchart JC, et al. Adipophilin enhances lipid accumulation and prevents lipid efflux from THP-1 macrophages: potential role in atherogenesis. Arterioscler Thromb Vasc Biol 2004; 24: 504–10.
Bildirici I, Roh CR, Schaiff WT, Lewkowski BM, Nelson DM, Sadovsky Y . The lipid droplet-associated protein adipophilin is expressed in human trophoblasts and is regulated by peroxisomal proliferator-activated receptor-gamma/retinoid X receptor. J Clin Endocrinol Metab 2003; 88: 6056–62.
Zhao Y, Sparks DL, Marcel YL . Specific phospholipid association with apolipoprotein A-I stimulates cholesterol efflux from human fibroblasts. Studies with reconstituted sonicated lipoproteins. J Biol Chem 1996; 271: 25145–51.
Liu XH, Xiao J, Mo ZC, Yin K, Jiang J, Cui LB, et al. Contribution of D4-F to ABCA1 expression and cholesterol efflux in THP-1 macrophage-derived foam cells. J Cardiovasc Pharmacol 2010 July 9. doi: 10.1097/FJC.0b013e3181edaf69
Yancey PG, Kawashiri MA, Moore R, Glick JM, Williams DL, Connelly MA, et al. In vivo modulation of HDL phospholipid has opposing effects on SR-BI- and ABCA1-mediated cholesterol efflux. J Lipid Res 2004; 45: 337–46.
Ronis MJ, Chen Y, Badeaux J, Badger TM . Dietary soy protein isolate attenuates metabolic syndrome in rats via effects on PPAR, LXR, and SREBP signaling. J Nutr 2009; 139: 1431–8.
Pettinelli P, Del Pozo T, Araya J, Rodrigo R, Araya AV, Smok G, et al. Enhancement in liver SREBP-1c/PPAR-alpha ratio and steatosis in obese patients: correlations with insulin resistance and n-3 long-chain polyunsaturated fatty acid depletion. Biochim Biophys Acta 2009; 1792: 1080–6.
Smart EJ, De Rose RA, Farber SA . Annexin 2-caveolin 1 complex is a target of ezetimibe and regulates intestinal cholesterol transport. Proc Natl Acad Sci U S A 2004; 101: 3450–5.
Babitt J, Trigatti B, Rigotti A, Smart EJ, Anderson RG, Xu S, et al. Murine SR-BI, a high density lipoprotein receptor that mediates selective lipid uptake, is N-glycosylated and fatty acylated and colocalizes with plasma membrane caveolae. J Biol Chem 1997; 272: 13242–9.
Uehara Y, Miura S, von Eckardstein A, Abe S, Fujii A, Matsuo Y, et al. Unsaturated fatty acids suppress the expression of the ATP-binding cassette transporter G1 (ABCG1) and ABCA1 genes via an LXR/RXR responsive element. Atherosclerosis 2007; 191: 11–21.
Feng B, Tabas I . ABCA1-mediated cholesterol efflux is defective in free cholesterol-loaded macrophages. Mechanism involves enhanced ABCA1 degradation in a process requiring full NPC1 activity. J Biol Chem 2002; 277: 43271–80.
Sumi K, Tanaka T, Uchida A, Magoori K, Urashima Y, Ohashi R, et al. Cooperative interaction between hepatocyte nuclear factor 4 alpha and GATA transcription factors regulates ATP-binding cassette sterol transporters ABCG5 and ABCG8. Mol Cell Biol 2007; 27: 4248–60.
Duan LP, Wang HH, Wang DQ . Cholesterol absorption is mainly regulated by the jejunal and ileal ATP-binding cassette sterol efflux transporters Abcg5 and Abcg8 in mice. J Lipid Res 2004; 45: 1312–23.
Kusuhara H, Sugiyama Y . ATP-binding cassette, subfamily G (ABCG family). Pflugers Arch 2007; 453: 735–44.
Calpe-Berdiel L, Rotllan N, Fievet C, Roig R, Blanco-Vaca F, Escola-Gil JC . Liver X receptor-mediated activation of reverse cholesterol transport from macrophages to feces in vivo requires ABCG5/G8. J Lipid Res 2008; 49: 1904–11.
Seizer P, Schiemann S, Merz T, Daub K, Bigalke B, Stellos K, et al. CD36 and macrophage scavenger receptor a modulate foam cell formation via inhibition of lipid-laden platelet phagocytosis. Semin Thromb Hemost 2010; 36: 157–62.
Stein S, Lohmann C, Schafer N, Hofmann J, Rohrer L, Besler C, et al. SIRT1 decreases Lox-1-mediated foam cell formation in atherogenesis. Eur Heart J 2010. doi: 10.1093/eurheartj/ehq107.
Nakano A, Inoue N, Sato Y, Nishimichi N, Takikawa K, Fujita Y, et al. LOX-1 mediates vascular lipid retention under hypertensive state. J Hypertens 2010; 28: 1273–80.
Pipe EA, Gobert CP, Capes SE, Darlington GA, Lampe JW, Duncan AM . Soy protein reduces serum LDL cholesterol and the LDL cholesterol:HDL cholesterol and apolipoprotein B:apolipoprotein A-I ratios in adults with type 2 diabetes. J Nutr 2009; 139: 1700–6.
Parish S, Peto R, Palmer A, Clarke R, Lewington S, Offer A, et al. The joint effects of apolipoprotein B, apolipoprotein A1, LDL cholesterol, and HDL cholesterol on risk: 3510 cases of acute myocardial infarction and 9805 controls. Eur Heart J 2009; 30: 2137–46.
Qi L, Liu S, Rifai N, Hunter D, Hu FB . Associations of the apolipoprotein A1/C3/A4/A5 gene cluster with triglyceride and HDL cholesterol levels in women with type 2 diabetes. Atherosclerosis 2007; 192: 204–10.
Horton JD, Shimomura I, Ikemoto S, Bashmakov Y, Hammer RE . Overexpression of sterol regulatory element-binding protein-1a in mouse adipose tissue produces adipocyte hypertrophy, increased fatty acid secretion, and fatty liver. J Biol Chem 2003; 278: 36652–60.
Tamehiro N, Shigemoto-Mogami Y, Kakeya T, Okuhira K, Suzuki K, Sato R, et al. Sterol regulatory element-binding protein-2- and liver X receptor-driven dual promoter regulation of hepatic ABC transporter A1 gene expression: mechanism underlying the unique response to cellular cholesterol status. J Biol Chem 2007; 282: 21090–9.
Shin DJ, Osborne TF . Thyroid hormone regulation and cholesterol metabolism are connected through sterol regulatory element-binding protein-2 (SREBP-2). J Biol Chem 2003; 278: 34114–8.
Masson D, Staels B, Gautier T, Desrumaux C, Athias A, Le Guern N, et al. Cholesteryl ester transfer protein modulates the effect of liver X receptor agonists on cholesterol transport and excretion in the mouse. J Lipid Res 2004; 45: 543–50.
Crestani M, De Fabiani E, Caruso D, Mitro N, Gilardi F, Vigil Chacon AB, et al. LXR (liver X receptor) and HNF-4 (hepatocyte nuclear factor-4): key regulators in reverse cholesterol transport. Biochem Soc Trans 2004; 32: 92–6.
Briand F, Treguier M, Andre A, Grillot D, Issandou M, Ouguerram K, et al. Liver X receptor activation promotes macrophage-to-feces reverse cholesterol transport in a dyslipidemic hamster model. J Lipid Res 2010; 51: 763–70.
Furnsinn C, Willson TM, Brunmair B . Peroxisome proliferator-activated receptor-delta, a regulator of oxidative capacity, fuel switching and cholesterol transport. Diabetologia 2007; 50: 8–17.
Ko MH, Puglielli L . The sterol carrier protein SCP-x/pro-SCP-2 gene has transcriptional activity and regulates the Alzheimer disease gamma-secretase. J Biol Chem 2007; 282: 19742–52.
Dyer DH, Wessely V, Forest KT, Lan Q . Three-dimensional structure/function analysis of SCP-2-like2 reveals differences among SCP-2 family members. J Lipid Res 2008; 49: 644–53.
Vikstedt R, Metso J, Hakala J, Olkkonen VM, Ehnholm C, Jauhiainen M . Cholesterol efflux from macrophage foam cells is enhanced by active phospholipid transfer protein through generation of two types of acceptor particles. Biochemistry 2007; 46: 11979–86.
Saarela J, Metso J, Schneider WJ, Jauhiainen M . Avian phospholipid transfer protein causes HDL conversion without affecting cholesterol efflux from macrophages. Biochim Biophys Acta 2009; 1791: 781–9.
Pahl MV, Ni Z, Sepassi L, Moradi H, Vaziri ND . Plasma phospholipid transfer protein, cholesteryl ester transfer protein and lecithin:cholesterol acyltransferase in end-stage renal disease (ESRD). Nephrol Dial Transplant 2009; 24: 2541–6.
Attia N, Nakbi A, Smaoui M, Chaaba R, Moulin P, Hammami S, et al. Increased phospholipid transfer protein activity associated with the impaired cellular cholesterol efflux in type 2 diabetic subjects with coronary artery disease. Tohoku J Exp Med 2007; 213: 129–37.
Tchoua U, D'Souza W, Mukhamedova N, Blum D, Niesor E, Mizrahi J, et al. The effect of cholesteryl ester transfer protein overexpression and inhibition on reverse cholesterol transport. Cardiovasc Res 2008; 77: 732–9.
Rensen PC, Havekes LM . Cholesteryl ester transfer protein inhibition: effect on reverse cholesterol transport? Arterioscler Thromb Vasc Biol 2006; 26: 681–4.
Erickson RP, Larson-Thome K, Weberg L, Szybinska A, Mossakowska M, Styczynska M, et al. Variation in NPC1, the gene encoding Niemann-Pick C1, a protein involved in intracellular cholesterol transport, is associated with Alzheimer disease and/or aging in the Polish population. Neurosci Lett 2008; 447: 153–7.
Shoji Y, Takahashi K, Ohta M, Kasai M, Kunishiro K, Kanda M, et al. Novel indoline-based acyl-CoA: cholesterol acyltransferase inhibitor: Effects of introducing a methanesulfonamide group on physicochemical properties and biological activities. Bioorg Med Chem 2009; 17: 6020–31.
Tanigawa H, Billheimer JT, Tohyama J, Fuki IV, Ng DS, Rothblat GH, et al. Lecithin: cholesterol acyltransferase expression has minimal effects on macrophage reverse cholesterol transport in vivo. Circulation 2009; 120: 160–9.
Franco M, Castro G, Romero L, Regalado JC, Medina A, Huesca-Gomez C, et al. Decreased activity of lecithin:cholesterol acyltransferase and hepatic lipase in chronic hypothyroid rats: implications for reverse cholesterol transport. Mol Cell Biochem 2003; 246: 51–6.
Gupta RP, Patrick K, Bell NH . Mutational analysis of CYP27A1: assessment of 27-hydroxylation of cholesterol and 25-hydroxylation of vitamin D. Metabolism 2007; 56: 1248–55.
von Bahr S, Movin T, Papadogiannakis N, Pikuleva I, Ronnow P, Diczfalusy U, et al. Mechanism of accumulation of cholesterol and cholestanol in tendons and the role of sterol 27-hydroxylase (CYP27A1). Arterioscler Thromb Vasc Biol 2002; 22: 1129–35.
Nuotio K, Isoviita PM, Saksi J, Ijas P, Pitkaniemi J, Sonninen R, et al. Adipophilin expression is increased in symptomatic carotid atherosclerosis: correlation with red blood cells and cholesterol crystals. Stroke 2007; 38: 1791–8.
Hascalovici JR, Song W, Vaya J, Khatib S, Fuhrman B, Aviram M, et al. Impact of heme oxygenase-1 on cholesterol synthesis, cholesterol efflux and oxysterol formation in cultured astroglia. J Neurochem 2009; 108: 72–81.
Wong J, Quinn CM, Brown AJ . SREBP-2 positively regulates transcription of the cholesterol efflux gene, ABCA1, by generating oxysterol ligands for LXR. Biochem J 2006; 400: 485–91.
Patel R, Soulages JL, Wells MA, Arrese EL . cAMP-dependent protein kinase of Manduca sexta phosphorylates but does not activate the fat body triglyceride lipase. Insect Biochem Mol Biol 2004; 34: 1269–79.
Hauet T, Yao ZX, Bose HS, Wall CT, Han Z, Li W, et al. Peripheral-type benzodiazepine receptor-mediated action of steroidogenic acute regulatory protein on cholesterol entry into leydig cell mitochondria. Mol Endocrinol 2005; 19: 540–4.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (30770868, 30971170), the Natural Science Foundation of Hunan Province, China (No 06JJ5055) and the Science & Technology Department of Hunan Province (No 04FJ4064). The authors acknowledge and express appreciation to Rhona KELLY for editing the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Luo, Dx., Cao, Dl., Xiong, Y. et al. A novel model of cholesterol efflux from lipid-loaded cells. Acta Pharmacol Sin 31, 1243–1257 (2010). https://doi.org/10.1038/aps.2010.93
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/aps.2010.93
Keywords
This article is cited by
-
Multi-omics analysis reveals changes in tryptophan and cholesterol metabolism before and after sexual maturation in captive macaques
BMC Genomics (2023)
-
Ezetimibe suppresses cholesterol accumulation in lipid-loaded vascular smooth muscle cells in vitro via MAPK signaling
Acta Pharmacologica Sinica (2014)
-
Tissue cholesterol content alterations in streptozotocin-induced diabetic rats
Acta Pharmacologica Sinica (2012)
-
Frontiers of vascular biology and disease research
Acta Pharmacologica Sinica (2010)