Abstract
Aim:
Tumor immunotherapy aims at activating the body's own immune system to fight an existing tumor. Effective antitumor responses require tumor antigens to be presented to lymphocytes. We aimed to test the hypothesis that intratumoral administration of recombinant adenovirus encoding MIP3β would induce antitumor immunity by attracting and facilitating the interaction between lymphocytes and dendritic cells.
Methods:
A recombinant adenovirus encoding microphage inflammatory protein 3β (AdMIP3β) was constructed. The antitumor activity of AdMIP3β in BALB/c and C57BL/6 mice bearing CT26 colon adenocarcinoma and Lewis lung cancer was evaluated.
Results:
Immunotherapy with AdMIP3β resulted in significant inhibition of tumor growth and prolonged survival of tumor-bearing mice. Tumor-specific immune responses elicited by AdMIP3β include MHC class I-dependent CD8+ CTL-mediated immune response and IFN-γ response. Immunohistochemical staining demonstrated numerous CD11c+ cells and CD3+ T lymphocytes within tumor tissues of AdMIP3β-treated mice. These findings suggest that the mechanism of specific antitumor immunity induced by AdMIP3β may be involved in the chemoattraction of both T lymphocytes and DCs to the tumor site and thus facilitate the process of antigen capture and mature DC to prime naive T cells.
Conclusion:
The present study may be important in the exploration of the potential application of AdMIP3β in the treatment of a broad spectrum of tumors.
Similar content being viewed by others
Introduction
Interactions between the immune system and malignant cells play a pivotal role in tumorigenesis. Multiple mechanisms have been proposed to explain the reasons for failure to develop effective immunity against tumors. Immune ignorance, impaired antigen presentation, expression of immunosuppressive factors, tolerance induction and apoptosis resistance likely play important roles in tumor immune escape1, 2. The aim of tumor immunotherapy is to activate the body's own immune system to fight an existing tumor. Effective antitumor responses require tumor antigens to be well presented to lymphocytes3. Tolerance or activation of immune responses largely depends on antigen presentation by antigen-presenting cells (APCs)4. Dendritic cells (DCs) are the most powerful APCs. The induction of tumor immunity can be initiated by the effectors of innate immunity and further developed by cells of adaptive immunity, with DCs playing a central regulatory role5.
Chemokines are a large family of cytokines capable of regulating migration and activation of leukocytes, DCs, etc6. Members of the chemokine family are categorized into four groups depending on the spacing of their first two cysteine residues. The CC chemokines have two adjacent cysteines near their amino terminus6. MIP3β is one CC chemokine that is highly expressed in lymphoid tissues and can chemoattract naive T cells, B cells, DCs, natural killer cells (NKs) and macrophage progenitor cells7, 8, 9, 10. MIP3β mediates its effects through the specific G protein-coupled seven-transmembrane domain chemokine receptor, CCR711.
MIP3β-CCR7 interaction plays a key role in promoting encounters between recirculating T cells and dendritic cells and in the migration of activated B cells into the T zone of secondary lymphoid tissues7. Based on our understanding of the function of MIP3β in immune response, we hypothesized that intratumoral administration of recombinant adenovirus encoding MIP3β would induce antitumor immunity by attracting and facilitating the interaction between lymphocytes and DCs. To test this hypothesis, the recombinant adenovirus encoding MIP3β (AdMIP3β) was constructed. At the same time, adenovirus-null (recombinant adenovirus without any therapeutic gene-AdNull) was used as a control virus. In the present study, AdMIP3β was shown to be effective in inducing antitumor immunity, which was mediated by both CD8+ dependent CTL response and IFN-γ response. These observations may indicate a new strategy for the treatment of tumors by eliciting strong tumor-specific immune response through local expression of MIP3β.
Materials and methods
Adenovirus vectors
All Adenovirus vectors used in this study are replication-incompetent serotype 5 adenoviruses (Ad5) with a deletion in the E1 region. The construction, production, and purification of adenovirus were performed as previously described12. Briefly, the expression cassette with the EF-1α/eIF4g composite promoter containing the mouse MIP3β cDNA (Genbank Accession Number: AF059208) was inserted into the shuttle vector. The generation of recombinant adenoviral plasmids was obtained by homologous recombination between the recombinant shuttle vector plasmid and the pAdEasy-1 adenoviral plasmid in E coli. Recombinant adenoviral vector DNA, digested with Pac I, was used for transfection of 293 cells from a human embryonic kidney epithelial cell line. Transfected cells were harvested when a complete viral cytopathic effect (CPE) was apparent (after 10–14 days). The cells were subjected to three cycles of freezing and thawing, and the virus was separated from cell debris by centrifugation. The virus stock was amplified by serial passage in 293 cells and purified using cesium chloride continuous gradient ultracentrifugation. AdNull, an essentially identical vector that lacks a transgene expression cassette, was prepared in the same way. The biological titer was determined by the TCID50 method according to the manufacturer's (Qbiogene) instructions. Viral concentration was expressed in plaque-forming units (PFU).
Reverse transcription-PCR
To confirm the expression of MIP3β mRNA mediated by AdMIP3β, total cellular RNA was isolated from the AdMIP3β infected SKOV3 cells using TRIzol reagent (Invitrogen, Grand Island, NY) and subjected to reverse transcription-PCR for the amplification of the fragment of MIP3β using a One-Step RT-PCR kit (Takara). Upstream primer and downstream primer for the amplified fragment of MIP3β were 5′-CTAATGATGCGGAAGACTGCTG-3′ and 5′-TGTTGCCTTTGTTCTTGGCA-3′. The human GAPDH (glyceraldehyde-3-phosphate dehydrogenase) gene was used as an internal control: 5′-CGACCACTTTGTCAAGCTCA-3′ and 5′-AGGGGTCTACATGGCAACTG-3′. RT-PCR was carried out at 50 °C for 30 min and at 94 °C for 2 min. Amplification was done at 94 °C for 30 s, 56 °C for 30 s, and 72 °C for 90 s (30 cycles). The reaction was extended at 72 °C for 7 min. The RT-PCR products were analyzed in a 1.5% agarose gel, visualized under UV light, and photographed.
Chemotaxis assay in vitro
To assess the chemoattractive property of MIP3β, cell culture supernatants harvested from AdMIP3β-infected COS-7 cells, AdNull-infected COS-7 cells and non-adenovirus-infected COS-7 cells were used. The migration of spleen lymphocytes was evaluated in a 96-well Boyden microchambers chemotaxis chamber (Neuroprobe) as described previously14. The lower wells of the chamber were filled with 50 μL of concentrated supernatant obtained as described above. A polyvinylpyrrolidone-free polycarbonate filter (5 μm pore size) was layered onto the wells. Then, 50 μL of cell suspension (5×106 spleen lymphocytes/mL) was added to the upper compartment of the migration chamber. The chemotaxis plate was incubated for 4 h at 37 °C in a humidified CO2 incubator. Cells that had migrated through the polycarbonate membrane were fixed in 70% methanol, stained with 1% Coomassie brilliant blue for 5 min and counted in three randomly selected high power fields under a microscope. Each assay was performed in triplicate and the results were expressed as the mean value of triplicates±SD.
Design of animal experiments
The CT26 colon carcinoma (CT26) model and Lewis lung carcinoma (LL/2) model were established in BALB/c and C57BL/6 female mice at 6 to 8 weeks of age, respectively. Tumor cells (5×105 CT26 or 2×106 LL/2 in 100 μL PBS) were injected into the right flanks of syngeneic mice. When the tumors were palpable on day 7 after tumor cell inoculation, mice were randomly divided into three groups of ten mice each. Group 1 mice received intratumoral administration of 5×108 PFU AdMIP3β in 100 μL PBS every 3 days, for a total of 6 doses. Group 2 mice received intratumoral administration of 5×108 PFU AdNull in 100 μL PBS every 3 days, for a total of 6 doses. Group 3 mice received intratumoral administration of 100 μL PBS every 3 days, for a total of 6 times. All studies involving mice were approved by the West China Hospital Cancer Center's Animal Care and Use Committee. The tumor size was monitored by measuring the longest dimension (length) and shortest dimension (width) at intervals of four days, and the tumor volume was calculated by the following formula: Tumor volume (mm3)=0.52×length (mm)×width2 (mm2). When the animals appeared moribund, the mice were killed, and this time point was defined as death for survival analysis.
Mice treated with recombinant adenovirus have been investigated, in particular for potential toxicity, for 3 months. Complete blood count and enzyme analyses were carried out. Gross measures such as weight loss, ruffling of fur, life span, behavior, and feeding were investigated. Tissues from the heart, liver, spleen, lung, kidney, brain, bone marrow, and other organs were also fixed in 10% neutral buffered formalin solution and embedded in paraffin. Sections of 5 μm were stained with hematoxylin and eosin (H&E).
Immunohistochemistry
The mice were sacrificed on day 7 after completion of treatment and the tumors were dissected. Sections from each paraffin embedded tissue sample for each group were stained with hematoxylin and eosin (H&E). For detection of T lymphocytes and MIP3β, sections were incubated with the appropriate diluted polyclonal goat antimouse CD3 (Santa Cruz Biotechnology, Inc) and monoclonal goat anti-mouse MIP3β (R&D Systems) overnight at 4 °C. After sections were washed, the secondary biotinylated antibody was added. Sections were then incubated with avidin-biotin-horseradish peroxidase complex (Vectastain Elite ABC), following the manufacturer's instructions. Slides were examined under a light microscope. To identify DC, frozen sections were fixed in acetone, washed with PBS, and incubated with hamster fluorescein isothiocyanate (FITC)-conjugated antibody against mouse CD11c (Serotec). Slides were examined under a fluorescence microscope.
In vitro cytotoxicity assay
LDH release assay was performed according to the manufacturer's protocol for the CytoTox 96 assay kits (Promega). Briefly, splenocytes obtained from the AdMIP3β-administered mice or control mice were treated with ammonium chloride-potassium lysing buffer to deplete erythrocytes. To prepare T-cell enriched fractions, these splenocytes were further incubated in complete medium for 90 min, and non-adherent cells were removed gently and incubated on nylon wool columns13. A total of 100 μL of effector cells and target cells (LL/2) were assigned at different E:T ratios to each well of a 96-well plate and incubated for 5 h at 37 °C. Cytotoxicity was quantified by measurement of the cytosolic enzyme, lactate dehydrogenase (LDH), in the culture supernatant. The percentage of specific lysis was expressed as specific lysis=[(experimental release)–(spontaneous release)/(target maximum–target spontaneous release)]×100. Spontaneous LDH release in the absence of CTL was less than 10% of the maximal cellular release by detergent lysis.
In the cytotoxicity inhibition assay, effector cells or tumor cells were pretreated with mAb at room temperature for 30 min, washed, and tested. mAbs used included anti-CD4 (10 μg/mL), anti-CD8 (10 μg/mL), anti-H-2Kb/H-2Db (50 μg/mL), anti-I-A/I-E (50 μg/mL) (BD PharMingen). The above concentrations of mAb were effective in mediating their activity in preliminary experiments. Control cytotoxicity assay was performed in the presence of control mAb or isotype IgG.
ELISPOT assay for detection of IFN-γ secreting T cells
Enzyme-linked immunospot (ELISPOT) assay for IFN-γ secreting cells was performed as previously described with slight modifications15, 16. Briefly, bone marrow-derived DCs were isolated by flushing femur bone activities. Red blood cells were lysed using ammonium chloride lysis buffer. Cells were cultured with 10% FCS RPMI-1640 added with 10 ng/mL GM-CSF and 10 ng/mL IL-4. DCs were harvested and enriched using 14.5% metrizamide (Sigma) complete medium gradients on day 7 and the purity was confirmed by co-expression of MHC-II and CD11c by FCM. DCs were incubated with tumor lysates for 16 hours at 37 °C. T cells isolated from AdMIP3β-treated mice and control mice were restimulated with tumor cell lysate-pulsed DCs at a responder-to-stimulator ratio of 20:1 in capture antibody-coated (anti-mouse IFN-γ antibody, R&D SYSTEMs) 96-well nitrocellulose plates and incubated for 5 h at 37 °C. The plates were then washed and incubated with a secondary biotinylated anti-IFN-γ mAb at 37 °C for 1 h. After being washed, the plates were developed with avidin-alkaline phosphatase and BCIP/NBT. The IFN-γ secreting T cells were dectected as dark blue spots and were enumerated under a microscope.
Purification of CD4+ or CD8+ T cells in vitro and their adoptive transfer in vivo
Purified CD4+ or CD8+ T lymphocytes from nylon wool-purified splenic T cells were obtained by treatment with specific Abs plus complement, as previously reported13. Briefly, nylon wool-purified splenic T cells (107 cells/mL) in complete medium were incubated on ice for 45 min with previously determined optimal amounts of purified mAb to mouse CD4 (10 μg/mL, clone GK 1.5; ATCC) or CD8 (10 μg/mL, clone2.43; ATCC). The cells were then washed with RPMI 1640 medium supplemented with 5% FBS. The lymphocytes were then incubated at 37 °C for 1 h in the presence of complement (rabbit Low-Tox-M; Cedarlane Laboratories, Hornby, Ontario, Canada). Viable cells were recovered by centrifugation through a 1.119 g/cm3 Ficoll gradient and washed twice in complete medium before being counted (>95% viable by trypan blue exclusion). Depletion of immune cell subsets was confirmed by flow cytometry (Coulter Elite ESP; Coulter, Miami, FL) using FITC-labeled anti-CD4 and anti-CD8 (BD PharMingen)13. In all cases, the specific cellular depletions were ≥99% effective. One day after the adoptive transfer of 2×107 cells, the mice were challenged with 1×105 tumor cells.
In vivo depletion of immune cell subsets
Immune cell subsets were depleted as described13. Briefly, tumor-burdened mice (10 mice/group) were injected ip with 500 μg of anti-CD4 (clone GK 1.5, rat IgG), anti-CD8 (clone 2.43, rat IgG) or isotype controls 1 day before the first AdMIP3β administration and then twice weekly for 3 weeks. Peripheral blood was collected from two representative mice from each group, and lymphocytes were analyzed by flow cytometry (Coulter Elite ESP, Miami, FL) for the depletion of the appropriate cell subset. As a result, the depletion of CD4+ and CD8+ cells was consistently >98%.
Statistical analyses
For comparison of individual time points, analysis of variance (ANOVA) and unpaired Student t tests were used.
Results
Expression and biological activity of murine MIP3β in vitro
The recombinant adenovirus encoding MIP3β (AdMIP3β) was constructed. The expression cassette with the EF-1α/e1F4g composite promoter containing the mouse MIP3β cDNA was cloned into the E1 site in the adenovirus genome (Figure 1A). The ability of AdMIP3β vector to produce functional MIP3β protein was confirmed in vitro by RT-PCR, Western blot analysis and chemotaxis assay. RT-PCR analysis revealed specific expression of MIP3β in SKOV3 cells infected with AdMIP3β (MOI=50), whereas MIP3β mRNA was not detected in AdNull-infected SKOV3 cells and non-adenovirus-infected SKOV3 cells (Figure 1B). To confirm the biological activity of secreted MIP3β, chemotaxis for lymphocytes was assayed. The concentrated supernatant of COS-7 cells infected with AdMIP3β showed markedly increased chemotactic activity for lymphocytes compared with controls (Figure 1C).
Schematic presentation of recombinant AdMIP3β and characterization of MIP3β expression by recombinant AdMIP3β and analysis of its chemotactic activity. Complete expression cassette for MIP3β was cloned into the E1 site in the adenovirus genome by homologous recombination (A). Total cellular RNA was isolated from the AdMIP3β infected SKOV3 cells using TRIzol reagent and subjected to RT-PCR for amplification of the MIP3β fragment. RT-PCR analysis revealed specific expression of MIP3β in SKOV3 cells infected with AdMIP3β (B, lane 1), whereas MIP3β mRNA was not dectected in AdNull-infected SKOV3 cells (B, lane 2) and non-adenovirus-infected SKOV3 cells (B, lane 3). Chemotaxis activity of recombinant MIP3β was demonstrated in lymphocytes with the addition of concentrated conditioned medium obtained from AdMIP3β-infected COS-7 cells. Procedures for cell staining were described under Materials and Methods. The number of cells that migrated to the lower surface was microscopically counted in five randomly chosen high power fields. The experiment was repeated three times and triplicate wells for each sample were set up for each experiment. The results were shown as means±SD. The conditioned medium obtained from AdMIP3β-infected COS-7 cells showed apparent chemotactic activity, which was inhibited when anti-MIP3β was given (C). bP<0.05 vs control groups.
Tumor growth inhibition studies
Intratumoral injection of AdMIP3β inhibited the growth of both CT26 murine tumor and LL/2 murine tumor. Several preliminary experiments have been conducted to explore the optimal dose and schedule of the administration of AdMIP3β. The preliminary data showed that the tumor inhibition effect resulting from the treatment with 1×109 PFU corresponded to that of 5×108 PFU, whereas treatment with a dose of 1×107 PFU showed a lesser effect. In addition, the tumor growth inhibition in mice treated with only one dose of 5×108 PFU resulted in significantly decreased tumor growth inhibition compared with the adopted schedule and dose of treatment with intratumoral injection of 5×108 PFU AdMIP3β per mouse every 3 days, for a total of 6 doses (data not shown). As shown in Figure 2, tumors grew progressively in AdNull or PBS-treated mice, but there was apparent inhibition of tumor growth in AdMIP3β-treated mice. The survival of the mice injected with AdMIP3β was also significantly longer than that of AdNull or PBS-injected mice (P<0.01, by log-rank test).
Tumor growth inhibition by AdMIP3β induced anti-tumor immunity. Mice (10 per group) treated with intratumoral injection of AdMIP3β (♦), AdNull (▴) and PBS (□)every 3 days, for a total of 6 doses, starting at d 7 after the sc introduction of 5×105 CT26 cells (A) or 2×106 LL/2 cells (B) into mice. Results are expressed as means±SD. bP<0.05 vs control groups. There was apparent protection from tumor growth in AdMIP3β-treated mice compared with AdNull/PBS-treated mice.
Mice treated with AdMIP3β have been investigated for potential long-term toxicity. No adverse consequences were indicated in gross measures such as weight loss, ruffling of fur, and life span. Furthermore, there were no side effects found with regard to hematology and liver function. Microscopic examination of the liver, kidney, lung, spleen, brain, heart, and bone marrow showed no pathologic changes.
Cellular immune response in AdMIP3β induced antitumor activity
The tumor killing activity of CTLs was examined. T cells isolated from the spleens of AdMIP3β-treated mice recognized and killed syngeneic tumor cells, thus exhibiting higher cytotoxicity against syngeneic tumor cells than those from control groups (Figure 3A). The cytotoxicity could be blocked by anti-CD8 or anti-MHC class I mAb, but not by anti-CD4 or anti-MHC-II in vitro, which suggests that the observed killing activity may result from MHC class I-dependent CD8+ CTL (Figure 3B).
Cellular immune response in AdMIP3β-induced antitumor activity. (A) Representative experiment of CTL-mediated cytotoxicity in vitro. T lymphocytes from mice treated with AdMIP3β (•), AdNull (▴) and PBS (□) were tested against LL/2 cells at different E:T ratios by an LDH release assay. T cells derived from the spleens of AdMIP3β-treated mice showed higher cytotoxicity against LL/2 cells than did T cells from control mice. The specific cytotoxicity could be blocked by anti-CD8 or anti-MHC-I but not by anti-CD4 or anti-MHC-II (B). (C) Abrogation of antitumor activity by in vivo depletion of the immune cell subsets. Depletion of immune cell subsets was performed as described. Depletion of CD8+T lymphocytes showed apparent abrogation of antitumor activity in AdMIP3β-treated mice, whereas the depletion of CD4+T lymphocytes showed partial abrogation of the antitumor effect. The depletion of both CD8+ and CD4+T lymphocytes showed synergistic abrogation of antitumor activity. Data represented are from day 25 after tumor cell injection. Similar results can be found at other time points. (D) Antitumor immunity by adoptive transfer of T cell subsets. Adoptive transfer of CD8+ T cells or CD4+ T cells from mice treated with AdMIP3β showed apparent anti-tumor activity. However, the transfer of T lymphocytes from mice treated with AdNull and PBS had no effect. bP<0.05 vs control groups.
Furthermore, mice depleted of CD8+ T lymphocytes by the injection of anti-CD8 mAb and treated with AdMIP3β were less protected from tumor challenge. Treatment with anti-CD4 mAb showed partial abrogation, whereas treatment with normal rat immunoglobulin (Ig) failed to abrogate the antitumor activity (Figure 3C). Although in vivo depletion of both CD8+ and CD4+T lymphocytes showed synergistic abrogation of antitumor activity, elements of antitumor immunity require further investigation.
Adoptive transfer of CD4-depleted (CD8+) or CD8-depleted (CD4+) T lymphocytes isolated from mice treated with AdMIP3β showed antitumor activity against syngeneic tumor cell implantation. The transfer of T lymphocyte subsets from mice treated with AdNull or mock infected mice had no anti-tumor effect (Figure 3D).
IFN-γ response
IFN-γ, the hallmark of Th1 type cytokine, is mainly produced by antigen epitope activated CD4+ Th1 or CD8+ T lymphocytes17, 18. Thus, IFN-γ specific ELISPOT assay could be utilized to assess the specificity and magnitude of T cell responses. The spleen cells isolated from the AdMIP3β-treated tumor-bearing mice or controls were re-stimulated with tumor cell lysate-pulsed DCs and then subjected to ELISPOT assay for evaluating IFN-γ producing cells. The IFN-γ secreting T cells were detected as dark blue spots and were enumerated under a microscope. As shown in Figure 4, increased IFN-γ secreting cells were found in T cells isolated from AdMIP3β-treated mice compared with controls (P<0.01).
Quantification of IFN-γ secreting cells by ELISPOT assay. Spleen cells from mice treated with AdMIP3β, AdNull or PBS were prepared one week after completion of treatment. Data presented are means±SD. Spleen cells from AdMIP3β-treated mice showed a significantly increased number of IFN-γ secreting cells (Spot forming cells, SFC) than those of control mice. bP<0.05 vs control groups.
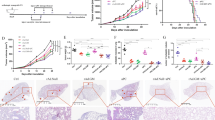
Intratumoral injection of AdMIP3β resulted in T lymphocyte and DC infiltration accompanied by distinct necrosis and apoptosis within tumor tissues. MIP3β chemoattracts both T cells and DCs7, 8. We therefore tested whether intratumoral administration of AdMIP3β could induce the infiltration of T cells and DCs within tumors. Immunohistochemical staining of MIP3β in tumor tissues showed comprehensive expression of MIP3β in local tumor tissues of AdMIP3β-treated mice (Figure 5A), but negative staining for tumor tissues of control mice (Figure 5B–5C). Also, H&E staining of sections of paraffin embedded tumor tissues revealed that treatment with AdMIP3β resulted in apparent lymphocyte infiltration accompanied by distinct necrosis and apoptosis compared with those from control mice (Figure 5D, 5E, 5F). Immunohistochemical staining of lymphocytes showed increased infiltration of CD3 positive lymphocytes within tumor tissues of AdMIP3β-treated mice (Figure 5G). In contrast, AdNull- or PBS-treated mice showed few CD3 positive T cells and CD19 positive B cells (Figure 5H–5I). Furthermore, detection of DC in tumor tissues by immunofluorescent staining with FITC-conjugated CD11c revealed that treatment with AdMIP3β resulted enriched infiltration of CD11c+ cells within the tumor tissues compared with those of control mice (Figure 5J–5L). These data suggested that intratumoral administration of AdMIP3β could induce anti-tumor immunity characterized by tumor tissue-infiltrated T lymphocytes as well as CD11c+ DCs.
Intratumoral injection of AdMIP3β resulted in MIP3β expression in tumor tissues and infiltration of lymphocytes and DCs accompanied by distinct necrosis and apoptosis within tumor tissues. The expression of MIP3β in tumor tissues was examined by immunohistochemical staining with goat anti-mouse MIP3β antibody as described under Materials and Methods. Local tumor tissues of AdMIP3β-treated mice exhibited a wide expression of MIP3β accompanied by lymphocyte infiltration (A), whereas the expression of MIP3β in tissues of control groups was not recognized (B and C). H&E staining of paraffin embedded tumor tissue sections showed distinct necrosis and apoptosis within the tumor tissues of AdMIP3β-treated mice (D) compared with tissues from the control groups (E, F). Immunohistochemical staining of lymphocytes demonstrated apparent CD3+ T lymphocyte infiltration within tumor tissues of AdMIP3β-treated mice (G), but not in control groups (H, I). Detection of DCs in tumor tissues by immunofluorescent staining with hamster FITC-conjugated CD11c showed enriched DC infiltration within the tumor tissues from the AdMIP3β treatment group (J) compared with those of the AdNull treatment group (K) and PBS group (L).
Discussion
Chemokines are a large family of cytokines that are involved in immune and inflammatory responses due to their capability of regulating the migration and activation of leukocytes, DCs, and certain other cells6. Among the many immunotherapeutic strategies currently evaluated, immunotherapy of tumors with chemokines represented a promising approach to trigger the recruitment of initiators or effectors of the immune response within tumor tissues19, 20, 21. Priming of tumor specific CTL responses frequently, if not always, requires the tumor antigen presented by APC, especially DC. MIP3β is one of the CC chemokines that is highly expressed in lymphoid tissues and chemoattracts naive T cells, B cells, DCs, NKs, and macrophage progenitor cells7, 8, 9, 10. Therefore, the present study was designed to explore the feasibility of immunotherapy of tumors with the recombinant adenovirus encoding MIP3β.
Several observations were made in the present study concerning the antitumor immunity induced by treatment with AdMIP3β. The present study was, to our knowledge, the first to demonstrate that the intratumoral injection of recombinant adenovirus encoding MIP3β could induce a tumor-specific immune response in mice. We found that a specific CTL response could be induced by intratumoral administration of AdMIP3β. The depletion of CD8+ T lymphocytes showed apparent abrogation of the antitumor activity in vivo. Also, antitumor activity can be acquired by adoptive transfer of CD8+ T lymphocytes. These findings suggest that MHC class I-dependent CD8+ CTL-mediated immune response may be mainly responsible for the observed antitumor activity. Also, the findings of IFN-γ response in the present study and the effect of depletion or adoptive transfer of CD4+ T lymphocytes suggest its partial role in antitumor immunity. Although the depletion of both CD8+ T lymphocytes and CD4+ T lymphocytes resulted in synergistic abrogation of antitumor activity, some protection remained, which requires further investigation.
The mechanism of specific antitumor immunity induced by treatment with AdMIP3β in the present study may be involved in the chemoattraction of both T lymphocytes and DCs to the tumor site, thus facilitating the process of antigen capture and mature DC priming naive T cells. Immunohistochemical staining did demonstrate an increased number of CD11c+ cells and CD3+T lymphocytes within tumors of AdMIP3β-treated mice compared with control tumors. The necrosis and apoptosis in AdMIP3β-treated mice were suggested to be induced by CD8+ CTL-mediated immune response and IFN-γ response. CTL-mediated cytotoxicity depends on either the granule exocytosis pathway or the Fas pathway, whereas IFN-γ induces apoptosis through decreased Bcl-2 and increased Bax expression22, 23.
It has been previously reported that tumor cells expressing ELC (MIP3β) showed delayed progression24. Recent studies have also shown that intratumoral injection of recombinant ELC/MIP3β resulted in an IFN-γ-dependent reduction in tumor burden25. In our study, we found that tumor specific CTL response and IFN-γ releasing response could be induced by AdMIP3β. The antitumor effects were comparable with those of previous studies24, 25. However, our approach is different in several ways compared with the reports above. First, MIP3β with biological activity in our study is expressed and released by tumor cells infected with AdMIP3β. It must be noted that the recombinant proteins produced in vitro and used in the previous study may lack proper glycosylation, and their administration can lead to the induction of neutralizing antibodies, resulting in reduced efficacy26. Second, the process of producing a recombinant protein is both difficult and expensive. Third, the vector used in this experiment is replication-deficient and E1-deleted adenovirus, which possesses the advantages of high transduction efficiency and low pathogenicity.
As the most potent APC capable of activating both resting and naive T cells, DCs remain a promising strategy for cancer therapy. Various strategies have been proposed for a DC vaccine, including DC pulsed with tumor-associated antigens (TAAs), DC fused with whole tumor cells, and DC transduced with mRNA or viral vectors27, 28, 29, 30, 31. However, preparation of patient-specific DC vaccine ex vivo is time-consuming and costly, which poses a considerable challenge for up-scale production in clinical settings. In the present study, intratumoral injection of AdMIP3β resulted in local expression of MIP3β and local recruitment of DCs and T lymphocytes, thus initiating an effective tumor-specific immune response. This new paradigm of harnessing endogenous DCs and T lymphocytes for cancer therapy possesses several advantages over previous strategies. First, it could chemoattract DCs and T lymphocytes to tumor sites, both of which are essential to elicit effective antitumor immune response. Second, it bypasses the complexing manipulation of DCs and T lymphocyts ex vivo. Third, this approach overcomes the fact that there have been only a few tumor specific antigens (TSA) identified by facilitating DCs and T lymophcytes to the site of the tumor antigen reservoir.
All together, the findings in the present study may provide a new immunotherapy strategy of inducing tumor specific immune response by chemoattracting both DCs and T lymphocytes to the tumor site. Because of the effective chemoattractivity of MIP3β and the general rules of anti-tumor immunoresponse, this paradigm may be further used in a broader spectrum of tumors.
Author contribution
Yu-quan WEI and Xia ZHAO designed research; Jian-mei HOU performed research; Gang LI, Bing YAO, and Ru ZHANG contributed new analytical tools and reagents; Hong-xin DENG, Ling TIAN, and Jin-liang YANG analyzed data; Jian-mei HOU wrote the paper.
References
Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD . Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol 2002; 3: 991–8.
Igney FH, Krammer PH . Immune escape of tumors: apoptosis resistance and tumor counterattack. J Leukoc Biol 2002; 71: 907–20.
Gunzer M, Janich S, Varga G, Grabbe S . Dendritc cells and tumor immunity. Semin Immunol 2001; 13: 291–302.
Pardoll DM . Therapeutic vaccination for cancer. Clin Immunol 2000; 95: 44–62.
Banchereau J, Briere F, Caux C, Davoust J, Lebecque S, Liu YJ, et al. Immunobiology of dendritic cells. Annu Rev Immunol 2000; 18: 767–811.
Schall TJ, Bacon KB . Chemokines, leukocyte trafficking and inflammation. Curr Opin Immunol 1994; 6: 865–73.
Ngo VN, Tang HL, Cyster JG . Epstein-Barr virus-induced molecule 1 ligand chemokine is expressed by dendritic cells in lymphoid tissues and strongly attracts naive T cells and activated B cells. J Exp Med 1998; 188: 181–91.
Yanagawa Y, Onoe K . CCL19 induces rapid dendritic extension of murine dendritic cells. Blood 2002; 100: 1948–56.
Kim CH, Pelus LM, Appelbaum E, Johanson K, Anzai N, Broxmeyer HE . CCR7 ligands, SLC/6Ckine/Exodus2/TCA4 and CKβ-11/MIP-3β/ELC, are chemoattractants for CD56(+)CD16(–) NK cells and late stage lymphoid progenitors. Cell Immunol 1999; 193: 226–35.
Kim CH, Pelus LM, White JR, Broxmeyer HE . Macrophage-inflammatory protein-3β/EBI1-ligand chemokine/CKbeta-11, a CC chemokine, is a chemoattractant with a specificity for macrophage progenitors among myeloid progenitor cells. J Immunol 1998; 161: 2580–5.
Yoshida R, Imai T, Hieshima K, Kusuda J, Baba M, Kitaura M, et al. Molecular cloning of a novel human CC chemokine EBI1-ligand chemokine that is a specific functional ligand for EBI1, CCR7. J Biol Chem 1997; 272: 13803–9.
He TC, Zhou S, da Costa LT, Yu J, Kinzler KW, Vogelstein B . A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci USA 1998; 95: 2509–14.
Lu Y, Wei YQ, Tian L, Zhao X, Yang L, Hu B, et al. Immunogene therapy of tumors with vaccine based on xenogeneic epidermal growth factor receptor. J Immunol 2003; 170: 3162–70.
Li G, Tian L, Hou JM, Ding ZY, He QM, Feng P, et al. Improved therapeutic effectiveness by combining recombinant CXC chemokine ligand 10 with cisplatin in solid tumors. Clin Cancer Res 2005; 11: 4217–24.
Benichou G, Valujskikh A, Heeger PS . Contributions of direct and indirect T cell alloreactivity during allograft rejection in mice. J Immunol 1999; 162: 352–8.
Fields RC, Shimizu K, Mule JJ . Murine dendritic cells pulsed with whole tumor lysates mediate potent antitumor immune responses in vitro and in vivo. Proc Natl Acad Sci USA 1998; 95: 9482–7.
Romagnani S . The Th1/Th2 paradigm. Immunol Today 1997; 18: 263–6.
Mosmann TR, Sad S . The expanding universe of T-cell subsets: Th1, Th2 and more. Immunol Today 1996; 17: 138–46.
Terando A, Roessler B, Mule JJ . Chemokine gene modification of human dendritic cell-based tumor vaccines using a recombinant adenoviral vector. Cancer Gene Ther 2004; 11: 165–73.
Dunussi-Joannopoulos K, Zuberek K, Runyon K, Hawley RG, Wong A, Erickson J, et al. Efficacious immunomodulatory activity of the chemokine stromal cell-derived factor 1 (SDF-1): local secretion of SDF-1 at the tumor site serves as T-cell chemoattractant and mediates T-cell-dependent antitumor responses. Blood 2002; 100: 1551–8.
Vicari AP, Ait-Yahia S, Chemin K, Mueller A, Zlotnik A, Caux C . Antitumor effects of the mouse chemokine 6Ckine/SLC through angiostatic and immunological mechanisms. J Immunol 2000; 165: 1992–2000.
Russell JH, Ley TJ . Lymphocyte-mediated cytotoxicity. Annu Rev Immunol 2002; 20: 323–70.
Zhou Y, Weyman CM, Liu H, Almasan A, Zhou A . IFN-gamma induces apoptosis in HL-60 cells through decreased Bcl-2 and increased Bak expression. J Interferon Cytokine Res 2008; 28: 65–72.
Braun SE, Chen K, Foster RG, Kim CH, Hromas R, Kaplan MH, et al. The CC chemokine CKbeta-11/MIP-3beta/ELC/Exodus 3 mediates tumor rejection of murine breast cancer cells through NK cells. J Immunol 2000; 164: 4025–31.
Hillinger S, Yang SC, Zhu L, Huang M, Duckett R, Atianzar K, et al. EBV-induced molecule 1 ligand chemokine (ELC/CCL19) promotes IFN-gamma-dependent antitumor responses in a lung cancer model. J Immunol 2003; 171: 6457–65.
Chada S, Ramesh R, Mhashilkar AM . Cytokine- and chemokine-based gene therapy for cancer. Curr Opin Mol Ther 2003; 5: 463–74.
Kikuchi T, Worgall S, Singh R, Moore MA, Crystal RG . Dendritic cells genetically modified to express CD40 ligand and pulsed with antigen can initiate antigen-specific humoral immunity independent of CD4+ T cells. Nat Med 2000; 6: 1154–9.
Chang AE, Redman BG, Whitfield JR, Nickoloff BJ, Braun TM, Lee PP, et al. A phase I trial of tumor lysate-pulsed dendritic cells in the treatment of advanced cancer. Clin Cancer Res 2002; 8: 1021–32.
Rubinstein MP, Kadima AN, Salem ML, Nguyen CL, Gillanders WE, Cole DJ . Systemic administration of IL-15 augments the antigen-specific primary CD8+ T cell response following vaccination with peptide-pulsed dendritic cells. J Immunol 2002; 169: 4928–35.
Boczkowski D, Nair SK, Nam JH, Lyerly HK, Gilboa E . Induction of tumor immunity and cytotoxic T lymphocyte responses using dendritic cells transfected with messenger RNA amplified from tumor cells. Cancer Res 2000; 60: 1028–34.
Kaplan JM, Yu Q, Piraino ST, Pennington SE, Shankara S, Woodworth LA, et al. Induction of antitumor immunity with dendritic cells transduced with adenovirus vector-encoding endogenous tumor-associated antigens. J Immunol 1999; 163: 699–707.
Acknowledgements
This work was supported by the National Basic Research Program of China (No 2004CB518800, 2004CB518807), the projects of National Natural Science Foundation of China, and the National 863 Project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hou, Jm., Zhao, X., Tian, L. et al. Immunotherapy of tumors with recombinant adenovirus encoding macrophage inflammatory protein 3β induces tumor-specific immune response in immunocompetent tumor-bearing mice. Acta Pharmacol Sin 30, 355–363 (2009). https://doi.org/10.1038/aps.2009.4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/aps.2009.4