Abstract
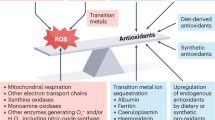
Many dietary phytochemicals exhibit health-beneficial effects including prevention of diseases such as cancer, as well as neurological, cardiovascular, inflammatory, and metabolic diseases. Evolutionarily, herbivorous and omnivorous animals have been ingesting plants. This interaction between “animal-plant” ecosystems has resulted in an elaborate system of detoxification and defense mechanisms evolved by animals including humans. Mammalian cells, including human cells, respond to these dietary phytochemicals by “non-classical receptor sensing” mechanisms of electrophilic chemical-stress typified by “thiol-modulated” cellular signaling events primarily leading to the gene expression of pharmacologically beneficial effects, but sometimes unwanted cytotoxicity also. Our laboratory has been studying two groups of dietary phytochemical cancer-chemopreventive compounds (isothiocyanates and polyphenols), which are effective in chemical-induced, as well as genetically-induced, animal carcinogenesis models. These compounds typically generate “cellular stress” and modulate gene expression of phase II detoxifying/antioxidant enzymes. Electrophiles, reactive oxygen species, and reactive nitrogen species are known to act as second messengers in the modulation of many cellular signaling pathways leading to gene expression changes and pharmacological responses. Redox-sensitive transcription factors such as nuclear factor-E2-related factor 2 (Nrf2), AP-1, NF-κB, to cite a few examples, sense and transduce changes in the cellular redox status and modulate gene expression responses to oxidative and electrophilic stresses, presumably via sulfhydryl modification of critical cysteine residues found on these proteins and/or other upstream redox-sensitive molecular targets. In the current review, we will explore dietary cancer chemopreventive phytochemicals, discuss the link between oxidative/electrophilic stresses and the redox circuitry, and consider different redox-sensitive transcription factors. We will also discuss the kelch-like erythroid Cap‘n’ Collar homologue-associated protein 1 (Keap1)-Nrf2 axis in redox signaling of induction of phase II detoxifying/antioxidant defense mechanisms, an important target and preventive strategy for normal cells against carcinogenesis, and the converse inhibition of cell growth/inflammatory signaling pathways that would confer therapeutic intervention in many types of cancers. Finally, we will summarize the Nrf2 paradigm in gene expression, the pharmacotoxicogenomic relevance of redox-sensitive Nrf2, and the redox regulation of cell death mechanisms.
Similar content being viewed by others
Article PDF
References
Chen, C, Kong, AN, Dietary chemopreventive compounds and ARE/EpRE signaling. Free Radic Biol Med 2004; 36: 1505–16.
Chen, C, Kong, AN, Dietary cancer-chemopreventive compounds: from signaling and gene expression to pharmacological effects. Trends Pharmacol Sci 2005; 26: 318–26.
Hu, R, Khor, TO, Shen, G, Jeong, WS, Hebbar, V, Chen, C, et al. Cancer chemoprevention of intestinal polyposis in ApcMin/+ mice by sulforaphane, a natural product derived from cruciferous vegetable. Carcinogenesis 2006; 27: 2038–46.
Xu, C, Huang, MT, Shen, G, Yuan, X, Lin, W, Khor, TO, et al. Inhibition of 7,12-dimethylbenz(a)anthracene-induced skin tumorigenesis in C57BL/6 mice by sulforaphane is mediated by nuclear factor E2-related factor 2. Cancer Res 2006; 66: 8293–6.
Forman, HJ, Torres, M, Fukuto, J, Redox signaling. Mol Cell Biochem 2002; 234–5: 49–62.
Hansen, JM, Go, YM, Jones, DP, Nuclear and mitochondrial compartmentation of oxidative stress and redox signaling. Annu Rev Pharmacol Toxicol 2006; 46: 215–34.
Hansen, JM, Watson, WH, Jones, DP, Compartmentation of Nrf–2 redox control: regulation of cytoplasmic activation by glutathione and DNA binding by thioredoxin-1. Toxicol Sci 2004; 82: 308–17.
Barford, D, The role of cysteine residues as redox-sensitive regulatory switches. Curr Opin Struct Biol 2004; 14: 679–86.
Sporn, MB, Liby, KT, Cancer chemoprevention: scientific promise, clinical uncertainty. Nat Clin Pract Oncol 2005; 2: 518–25.
Lotem, J, Netanely, D, Domany, E, Sachs, L, Human cancers overexpress genes that are specific to a variety of normal human tissues. Proc Natl Acad Sci USA 2005; 102: 18 556–61.
Surh, YJ, Cancer chemoprevention with dietary phytochemicals. Nat Rev Cancer 2003; 3: 768–80.
Conney, AH, Tailoring cancer chemoprevention regimens to the individual. J Cell Biochem 2004; 91: 277–86.
Chemoprevention Working Group. Prevention of cancer in the next millennium: Report of the Chemoprevention Working Group to the American Association for Cancer Research. Cancer Res 1999; 59: 4743–58.
Chen, C, Shen, G, Hebbar, V, Hu, R, Owuor, ED, Kong, AN, Epigal-locatechin-3-gallate-induced stress signals in HT-29 human colon adenocarcinoma cells. Carcinogenesis 2003; 24: 1369–78.
Hu, R, Kim, BR, Chen, C, Hebbar, V, Kong, AN, The roles of JNK and apoptotic signaling pathways in PEITC-mediated responses in human HT-29 colon adenocarcinoma cells. Carcinogenesis 2003; 24: 1361–7.
Jeong, WS, Kim, IW, Hu, R, Kong, AN, Modulatory properties of various natural chemopreventive agents on the activation of NF-kappaB signaling pathway. Pharm Res 2004; 21: 661–70.
Keum, YS, Yu, S, Chang, PP, Yuan, X, Kim, JH, Xu, C, et al. Mechanism of action of sulforaphane: inhibition of p38 mitogen-activated protein kinase isoforms contributing to the induction of antioxidant response element-mediated heme oxygenase-1 in human hepatoma HepG2 cells. Cancer Res 2006; 66: 8804–13.
Xu, C, Yuan, X, Pan, Z, Shen, G, Kim, JH, Yu, S, et al. Mechanism of action of isothiocyanates: the induction of ARE-regulated genes is associated with activation of ERK and JNK and the phosphorylation and nuclear translocation of Nrf2. Mol Cancer Ther 2006; 5: 1918–26.
Kim, JH, Xu, C, Keum, YS, Reddy, B, Conney, A, Kong, AN, Inhibition of EGFR signaling in human prostate cancer PC-3 cells by combination treatment with beta-phenylethyl isothiocyanate and curcumin. Carcinogenesis 2006; 27: 475–82.
Khor, TO, Keum, YS, Lin, W, Kim, JH, Hu, R, Shen, G, et al. Combined inhibitory effects of curcumin and phenethyl isothiocyanate on the growth of human PC-3 prostate xenografts in immunodeficient mice. Cancer Res 2006; 66: 613–21.
Myzak, MC, Dashwood, WM, Orner, GA, Ho, E, Dashwood, RH, Sulforaphane inhibits histone deacetylase in vivo and suppresses tumorigenesis in Apc-minus mice. Faseb J 2006; 20: 506–8.
Kim, BR, Hu, R, Keum, YS, Hebbar, V, Shen, G, Nair, SS, et al. Effects of glutathione on antioxidant response element-mediated gene expression and apoptosis elicited by sulforaphane. Cancer Res 2003; 63: 7520–5.
Khor, TO, Huang, MT, Kwon, KH, Reddy, BS, Kong, AN, Nrf2-deficient mice have an increased susceptibility to dextran sulfate sodium-induced colitis. Cancer Res 2006; 66: 11580–4.
Dinkova-Kostova, AT, Liby, KT, Stephenson, KK, Holtzclaw, WD, Gao, X, Suh, N, et al. Extremely potent triterpenoid inducers of the phase 2 response: correlations of protection against oxidant and inflammatory stress. Proc Natl Acad Sci USA 2005; 102: 4584–9.
Moriarty-Craige, SE, Jones, DP, Extracellular thiols and thiol/disulfide redox in metabolism. Annu Rev Nutr 2004; 24: 481–509.
Georgiou, G, How to flip the (redox) switch. Cell 2002; 111: 607–10.
Jacob, C, Lancaster, JR, Giles, GI, Reactive sulphur species in oxidative signal transduction. Biochem Soc Trans 2004; 32: 1015–7.
Karin, M, The regulation of AP-1 activity by mitogen-activated protein kinases. J Biol Chem 1995; 270: 16 483–6.
Smeal, T, Binetruy, B, Mercola, D, Grover-Bardwick, A, Heidecker, G, Rapp, UR, et al. Oncoprotein-mediated signalling cascade stimulates c-Jun activity by phosphorylation of serines 63 and 73. Mol Cell Biol 1992; 12: 3507–13.
Abate, C, Patel, L, Rauscher, FJ III, Curran, T, Redox regulation of fos and jun DNA-binding activity in vitro. Science 1990; 249: 1157–61.
Xanthoudakis, S, Curran, T, Redox regulation of AP-1: a link between transcription factor signaling and DNA repair. Adv Exp Med Biol 1996; 387: 69–75.
Hirota, K, Matsui, M, Iwata, S, Nishiyama, A, Mori, K, Yodoi, J, AP-1 transcriptional activity is regulated by a direct association between thioredoxin and Ref-1. Proc Natl Acad Sci USA 1997; 94: 3633–8.
Matthews, JR, Kaszubska, W, Turcatti, G, Wells, TN, Hay, RT, Role of cysteine62 in DNA recognition by the P50 subunit of NF-kappa B. Nucleic Acids Res 1993; 21: 1727–34.
Galter, D, Mihm, S, Droge, W, Distinct effects of glutathione disulphide on the nuclear transcription factor kappa B and the activator protein-1. Eur J Biochem 1994; 221: 639–48.
Hirota, K, Murata, M, Sachi, Y, Nakamura, H, Takeuchi, J, Mori, K, et al. Distinct roles of thioredoxin in the cytoplasm and in the nucleus. A two-step mechanism of redox regulation of transcription factor NF-kappaB. J Biol Chem 1999; 274: 27 891–7.
Alam, J, Stewart, D, Touchard, C, Boinapally, S, Choi, AM, Cook, JL, Nrf2, a Cap ‘n’ Collar transcription factor, regulates induction of the heme oxygenase-1 gene. J Biol Chem 1999; 274: 26 071–8.
McMahon, M, Itoh, K, Yamamoto, M, Chanas, SA, Henderson, CJ, McLellan, LI, et al. The Cap ‘n’ Collar basic leucine zipper transcription factor Nrf2 (NF-E2 p45-related factor 2) controls both constitutive and inducible expression of intestinal detoxification and glutathione biosynthetic enzymes. Cancer Res 2001; 61: 3299–307.
Thimmulappa, RK, Mai, KH, Srisuma, S, Kensler, TW, Yamamoto, M, Biswal, S, Identification of Nrf2-regulated genes induced by the chemopreventive agent sulforaphane by oligonucleotide microarray. Cancer Res 2002; 62: 5196–203.
Prochaska, HJ, De Long, MJ, Talalay, P, On the mechanisms of induction of cancer-protective enzymes: a unifying proposal. Proc Natl Acad Sci USA 1985; 82: 8232–6.
Li, W, Jain, MR, Chen, C, Yue, X, Hebbar, V, Zhou, R, et al. Nrf2 possesses a redox-insensitive nuclear export signal overlapping with the leucine zipper motif. J Biol Chem 2005; 280: 28 430–8.
Shen, G, Hebbar, V, Nair, S, Xu, C, Li, W, Lin, W, et al. Regulation of Nrf2 transactivation domain activity. The differential effects of mitogen-activated protein kinase cascades and synergistic stimulatory effect of Raf and CREB-binding protein. J Biol Chem 2004; 279: 23 052–60.
Keum, YS, Owuor, ED, Kim, BR, Hu, R, Kong, AN, Involvement of Nrf2 and JNK1 in the activation of antioxidant responsive element (ARE) by chemopreventive agent phenethyl isothiocyanate (PEITC). Pharm Res 2003; 20: 1351–6.
Yu, X, Kensler, T, Nrf2 as a target for cancer chemoprevention. Mutat Res 2005; 591: 93–102.
Burton, NC, Kensler, TW, Guilarte, TR, In vivo modulation of the Parkinsonian phenotype by Nrf2. Neurotoxicology 2006; 27: 1094–100.
Itoh, K, Wakabayashi, N, Katoh, Y, Ishii, T, Igarashi, K, Engel, JD, et al. Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev 1999; 13: 76–86.
Dhakshinamoorthy, S, Jaiswal, AK, Functional characterization and role of INrf2 in antioxidant response element-mediated expression and antioxidant induction of NAD(P)H:quinone oxi-doreductase1 gene. Oncogene 2001; 20: 3906–17.
Zhang, DD, Hannink, M, Distinct cysteine residues in Keap1 are required for Keap1-dependent ubiquitination of Nrf2 and for stabilization of Nrf2 by chemopreventive agents and oxidative stress. Mol Cell Biol 2003; 23: 8137–51.
Zhang, DD, Lo, SC, Cross, JV, Templeton, DJ, Hannink, M, Keap1 is a redox-regulated substrate adaptor protein for a Cul3-dependent ubiquitin ligase complex. Mol Cell Biol 2004; 24: 10 941–53.
Velichkova, M, Hasson, T, Keap1 regulates the oxidation-sensitive shuttling of Nrf2 into and out of the nucleus via a Crm1-dependent nuclear export mechanism. Mol Cell Biol 2005; 25: 4501–13.
Wakabayashi, N, Dinkova-Kostova, AT, Holtzclaw, WD, Kang, MI, Kobayashi, A, Yamamoto, M, et al. Protection against electrophile and oxidant stress by induction of the phase 2 response: fate of cysteines of the Keap1 sensor modified by inducers. Proc Natl Acad Sci USA 2004; 101: 2040–5.
Li, W, Yu, SW, Kong, AN, Nrf2 possesses a redox-sensitive nuclear exporting signal in the Neh5 transactivation domain. J Biol Chem 2006; 281: 27 251–63.
Dhakshinamoorthy, S, Jaiswal, AK, Small maf (MafG and MafK) proteins negatively regulate antioxidant response element-mediated expression and antioxidant induction of the NAD(P)H: quinone oxidoreductase1 gene. J Biol Chem 2000; 275: 40 134–41.
Nair, S, Xu, C, Shen, G, Hebbar, V, Gopalakrishnan, A, Hu, R, et al. Pharmacogenomics of phenolic antioxidant butylated hydroxyanisole (BHA) in the small intestine and liver of Nrf2 knockout and C57BL/6J mice. Pharm Res 2006; 23: 2621–37.
Jain, AK, Bloom, DA, Jaiswal, AK, Nuclear import and export signals in control of Nrf2. J Biol Chem 2005; 280: 29 158–68.
Bloom, D, Dhakshinamoorthy, S, Jaiswal, AK, Site-directed mutagenesis of cysteine to serine in the DNA binding region of Nrf2 decreases its capacity to upregulate antioxidant response element-mediated expression and antioxidant induction of NAD(P) H:quinone oxidoreductase1 gene. Oncogene 2002; 21: 2191–200.
Shen, G, Xu, C, Hu, R, Jain, MR, Nair, S, Lin, W, et al. Comparison of (-)-epigallocatechin-3-gallate elicited liver and small intestine gene expression profiles between C57BL/6J mice and C57BL/6J/Nrf2 (-/-) mice. Pharm Res 2005; 22: 1805–20.
Shen, G, Xu, C, Hu, R, Jain, MR, Gopalkrishnan, A, Nair, S, et al. Modulation of nuclear factor E2-related factor 2-mediated gene expression in mice liver and small intestine by cancer chemopreventive agent curcumin. Mol Cancer Ther 2006; 5: 39–51.
Hu, R, Xu, C, Shen, G, Jain, MR, Khor, TO, Gopalkrishnan, A, et al. Gene expression profiles induced by cancer chemopreventive isothiocyanate sulforaphane in the liver of C57BL/6J mice and C57BL/6J/Nrf2 (-/-) mice. Cancer Lett 2006; 243: 170–92.
Hu, R, Xu, C, Shen, G, Jain, MR, Khor, TO, Gopalkrishnan, A, et al. Identification of Nrf2-regulated genes induced by chemopreventive isothiocyanate PEITC by oligonucleotide microarray. Life Sci 2006; 79: 1944–55.
Nair, S, Xu, C, Shen, G, Hebbar, V, Gopalakrishnan, A, Hu, R, et al. Toxicogenomics of endoplasmic reticulum stress inducer tunica-mycin in the small intestine and liver of Nrf2 knockout and C57BL/6J Mice. Toxicol Lett 2006; 168: 21–39.
Katoh, Y, Itoh, K, Yoshida, E, Miyagishi, M, Fukamizu, A, Yamamoto, M, Two domains of Nrf2 cooperatively bind CBP, a CREB binding protein, and synergistically activate transcription. Genes Cells 2001; 6: 857–68.
Lin, W, Shen, G, Yuan, X, Jain, MR, Yu, S, Zhang, A, et al. Regulation of Nrf2 transactivation domain activity by p160 RAC3/ SRC3 and other nuclear co-regulators. J Biochem Mol Biol 2006; 39: 304–10.
Chen, H, Tini, M, Evans, RM, HATs on and beyond chromatin. Curr Opin Cell Biol 2001; 13: 218–24.
Ceribelli, M, Alcalay, M, Vigano, MA, Mantovani, R, Repression of new p53 targets revealed by ChIP on chip experiments. Cell Cycle 2006; 5: 1102–10.
Talalay, P, Chemoprotection against cancer by induction of phase 2 enzymes. Biofactors 2000; 12: 5–11.
Zhang, J, Ohta, T, Maruyama, A, Hosoya, T, Nishikawa, K, Maher, JM, et al. BRG1 interacts with Nrf2 to selectively mediate HO-1 induction in response to oxidative stress. Mol Cell Biol 2006; 26: 7942–52.
Wang, W, Chan, JY, Nrf1 is targeted to the endoplasmic reticulum membrane by an N-terminal transmembrane domain. Inhibition of nuclear translocation and transacting function. J Biol Chem 2006; 281: 19 676–87.
Zhang, K, Kaufman, RJ, Protein folding in the endoplasmic reticulum and the unfolded protein response. Handb Exp Pharmacol 2006; 69–91.
Hetz, C, Bernasconi, P, Fisher, J, Lee, AH, Bassik, MC, Antonsson, B, et al. Proapoptotic BAX and BAK modulate the unfolded protein response by a direct interaction with IRE1alpha. Science 2006; 312: 572–6.
Srivastava, RK, Sollott, SJ, Khan, L, Hansford, R, Lakatta, EG, Longo, DL, Bcl-2 and Bcl-X(L) block thapsigargin-induced nitric oxide generation, c-Jun NH(2)-terminal kinase activity, and apoptosis. Mol Cell Biol 1999; 19: 5659–74.
Kyriakis, JM, Banerjee, P, Nikolakaki, E, Dai, T, Rubie, EA, Ahmad, MF, et al. The stress-activated protein kinase subfamily of c-Jun kinases. Nature 1994; 369: 156–60.
Urano, F, Wang, X, Bertolotti, A, Zhang, Y, Chung, P, Harding, HP, et al. Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE1. Science 2000; 287: 664–6.
Yu, R, Jiao, JJ, Duh, JL, Tan, TH, Kong, AN, Phenethyl isothiocyanate, a natural chemopreventive agent, activates c-Jun N-terminal kinase 1. Cancer Res 1996; 56: 2954–9.
Garber, K, Researchers target unfolded protein response in cancerous tumor growth. J Natl Cancer Inst 2006; 98: 512–4.
Jones, DP, Extracellular redox state: refining the definition of oxidative stress in aging. Rejuvenation Res 2006; 9: 169–81.
Catarzi, S, Biagioni, C, Giannoni, E, Favilli, F, Marcucci, T, Iantomasi, T, et al. Redox regulation of platelet-derived-growth-factor-receptor: role of NADPH-oxidase and c-Src tyrosine kinase. Biochim Biophys Acta 2005; 1745: 166–75.
Cheng, YJ, Liu, MY, Modulation of tumor necrosis factor-alpha and oxidative stress through protein kinase C and P42/44 mitogen-activated protein kinase in lead increases lipopolysaccharide-induced liver damage in rats. Shock 2005; 24: 188–93.
Engers, R, Springer, E, Kehren, V, Simic, T, Young, DA, Beier, J, et al. Rac upregulates tissue inhibitor of metalloproteinase–1 expression by redox-dependent activation of extracellular signal-regulated kinase signaling. Febs J 2006; 273: 4754–69.
Erker, L, Schubert, R, Yakushiji, H, Barlow, C, Larson, D, Mitchell, JB, et al. Cancer chemoprevention by the antioxidant tempol acts partially via the p53 tumor suppressor. Hum Mol Genet 2005; 14: 1699–708.
Trachootham, D, Zhou, Y, Zhang, H, Demizu, Y, Chen, Z, Pelicano, H, et al. Selective killing of oncogenically transformed cells through a ROS-mediated mechanism by beta-phenylethyl isothiocyanate. Cancer Cell 2006; 10: 241–52.
Xu, C, Shen, G, Chen, C, Gelinas, C, Kong, AN, Suppression of NF-kappaB and NF-kappaB-regulated gene expression by sulforaphane and PEITC through IkappaBalpha, IKK pathway in human prostate cancer PC-3 cells. Oncogene 2005; 24: 4486–95.
Shen, HM, Pervaiz, S, TNF receptor superfamily-induced cell death: redox-dependent execution. Faseb J 2006; 20: 1589–98.
Han, D, Hanawa, N, Saberi, B, Kaplowitz, N, Hydrogen peroxide and redox modulation sensitize primary mouse hepatocytes to TNF-induced apoptosis. Free Radic Biol Med 2006; 41: 627–39.
Arai, RJ, Masutani, H, Yodoi, J, Debbas, V, Laurindo, FR, Stern, A, et al. Nitric oxide induces thioredoxin-1 nuclear translocation: possible association with the p21Ras survival pathway. Biochem Biophys Res Commun 2006; 348: 1254–60.
Shen, G, Xu, C, Chen, C, Hebbar, V, Kong, AN, p53-independent G1 cell cycle arrest of human colon carcinoma cells HT-29 by sulforaphane is associated with induction of p21CIP1 and inhibition of expression of cyclin D1. Cancer Chemother Pharmacol 2006; 57: 317–27.
Luo, T, Xia, Z, A small dose of hydrogen peroxide enhances tumor necrosis factor-alpha toxicity in inducing human vascular endothelial cell apoptosis: reversal with propofol. Anesth Analg 2006; 103: 110–6.
Ge, Y, Montano, I, Rustici, G, Freebern, WJ, Haggerty, CM, Cui, W, et al. Selective leukemic cell killing by a novel functional class of thalidomide analogs. Blood 2006; 108: 4126–35.
Macho, A, Calzado, MA, Munoz-Blanco, J, Gomez-Diaz, C, Gajate, C, Mollinedo, F, et al. Selective induction of apoptosis by capsaicin in transformed cells: the role of reactive oxygen species and calcium. Cell Death Differ 1999; 6: 155–65.
Reynaert, NL, Wouters, EF, Janssen-Heininger, YM, Modulation of glutaredoxin-1 expression in a mouse model of allergic airway disease. Am J Respir Cell Mol Biol 2007; 36: 147–51.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported in part by grants to Ah-Ng Tony Kong from the National Institute of Health (No RO1-CA094828, RO1-CA092515, RO1-CA073674, and RO1-CA118947).
Rights and permissions
About this article
Cite this article
Nair, S., Li, W. & Kong, AN. Natural dietary anti-cancer chemopreventive compounds: redox-mediated differential signaling mechanisms in cytoprotection of normal cells versus cytotoxicity in tumor cells. Acta Pharmacol Sin 28, 459–472 (2007). https://doi.org/10.1111/j.1745-7254.2007.00549.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00549.x
Keywords
This article is cited by
-
(E)-N-(2-(3, 5-Dimethoxystyryl) phenyl) furan-2-carboxamide (BK3C231) induces cytoprotection in CCD18-Co human colon fibroblast cells through Nrf2/ARE pathway activation
Scientific Reports (2021)
-
Derrischalcone suppresses cholangiocarcinoma cells through targeting ROS-mediated mitochondrial cell death, Akt/mTOR, and FAK pathways
Naunyn-Schmiedeberg's Archives of Pharmacology (2021)
-
Emerging Insights into Anticancer Chemopreventive Activities of Nutraceutical Moringa oleifera: Molecular Mechanisms, Signal Transduction and In Vivo Efficacy
Current Pharmacology Reports (2020)
-
Relationship between conversion rate of glucosinolates to isothiocyanates/indoles and genotoxicity of individual parts of Brassica vegetables
European Food Research and Technology (2019)
-
Effect of olive cultivar on bioaccessibility and antioxidant activity of phenolic fraction of virgin olive oil
European Journal of Nutrition (2018)