Abstract
Aim:
Contraction of cardiac myocytes is controlled by the generation and amplification of intracellular Ca2+ signals. The key step of this process is the coupling between sarcolemma L-type Ca2+ channels (LCCs) and ryanodine receptors (RyRs) in the sarcoplasmic reticulum (SR). β-Adrenergic stimulation is an important regulatory mechanism for this coupling process. But the details underlied the global level, which require local Ca2+ release study are still unclear. The present study is to explore the effects of β-adrenergic stimulation on local Ca2+ release.
Methods:
Using confocal microscopy combined with loose-seal patch-clamp approaches, effects of isoprenaline (1 μmol-L−1), a β-adrenergic agonist, on local SR Ca2+ release triggered by Ca2+ influx through LCCs in intact rat cardiac myocytes were investigated.
Results:
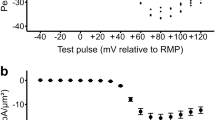
Isoprenaline increased the intensity of ensemble averaged local Ca2+ transients, the peak of which displayed a typical bell-shaped voltage-dependence over the membrane voltages ranging from ∼-40mV to ∼+35mV. Further analysis showed that this enhancement could be explained by the increased coupling fidelity (which refers the increased probability of RyRs activation upon depolarization), and the increased amplitude of evoked Ca2+ sparks (due to more Ca2+ releases through local RyRs). In addition, isoprenaline decreased the first latency, which displayed a typical “U”-shaped voltage-dependence, showing the available acceleration and synchronization of β-adrenergic stimulation on intracellular calcium release.
Conclusions:
Isoprenaline enhances local Ca2+ release in cardiac myocytes. These results underscore the importance of regulation of β-adrenergic stimulation on local intermolecular signals between LCCs and RyRs in heart cells.
Similar content being viewed by others
Article PDF
References
Cheng H, Lederer WJ, Cannell MB . Calcium sparks: elementary events underlying excitation-contraction coupling in heart muscle. Science 1993; 262: 740–4.
Lindegger N, Niggli E . Paradoxical SR Ca2+ release in guinea-pig cardiac myocytes after beta-adrenergic stimulation revealed by two-photon photolysis of caged Ca2+. J Physiol 2005; 565: 801–13.
Cheng H, Wang SQ . Calcium signaling between sarcolemmal calcium channels and ryanodine receptors in heart cells. Front Biosci 2002; 7: d1867–78.
Bers DM . Cardiac excitation-contraction coupling. Nature 2002; 415: 198–205.
Wang H, Oestreich EA, Maekawa N, Bullard TA, Vikstrom KL, Dirksen RT, et al. Phospholipase C epsilon modulates beta-adrenergic receptor-dependent cardiac contraction and inhibits cardiac hypertrophy. Circ Res 2005; 97: 1305–13.
Ziolo MT, Maier LS, Piacentino V Bossuyt J, Houser SR, Bers DM . Myocyte nitric oxide synthase 2 contributes to blunted beta-adrenergic response in failing human hearts by decreasing Ca2+ transients. Circulation 2004; 109: 1886–91.
Song LS, Wang SQ, Xiao RP, Spurgeon H, Lakatta EG, Cheng H . β-Adrenergic stimulation synchronizes intracellular Ca2+ release during excitation-contraction coupling in cardiac myocytes. Circ Res 2001; 88: 794–801.
Katra RP, Laurita KR . Cellular mechanism of calcium-mediated triggered activity in the heart. Circ Res 2005; 96: 535–42.
Shen JX, Wang S, Song LS, Han T, Cheng H . Polymorphism of Ca2+ sparks evoked from in-focus Ca2+ release units in cardiac myocytes. Biophys J 2004; 86: 182–90.
Wang SQ, Song LS, Lakatta EG, Cheng H . Ca2+ signalling between single L-type Ca2+ channels and ryanodine receptors in heart cells. Nature 2001; 410: 592–6.
Cheng H, Song LS, Shirokova N, Gonzalez A, Lakatta EG, Rios E, et al. Amplitude distribution of calcium sparks in confocal images: theory and studies with an automatic detection method. Biophys J 1999; 76: 606–17.
Bers DM Cardiac inotropy and Ca mismanagement, in excitation-contraction coupling and cardiac contractile force. Boston: Kluwer Academic Publishers; 2001. p 273–331.
Ziolo MT, Katoh H, Bers DM . Positive and negative effects of nitric oxide on Ca2+ sparks: influence of beta -adrenergic stimulation. Am J Physiol Heart Circ Physiol 2001; 281: H2295–303.
Sah R, Ramirez RJ, Backx PH . Modulation of Ca2+ release in cardiac myocytes by changes in repolarization rate: role of phase-1 action potential repolarization in excitation-contraction coupling. Circ Res 2002; 90: 165–73.
Brochet DXP, Yang D, Maio AD, Lederer WJ, Franzini-Armstrong C, Cheng H . Ca2+ blinks: rapid nanoscopic store calcium signaling. Proc Natl Acad Sci USA 2005; 102: 3099–104.
Valdivia HH, Kaplan JH, Ellis-Davies GC, Lederer WJ . Rapid adaptation of cardiac ryanodine receptors: modulation by Mg2+ and phosphorylation. Science 1995; 267: 1997–2000.
Marx SO, Gaburjakova J, Gaburjakova M, Henrikson C, Ondrias K, Marks AR . Coupled gating between cardiac calcium release channels (ryanodine receptors). Circ Res 2001; 88: 1151–8.
Lu X, Xu L, Meissner G . Phosphorylation of dihydropyridine receptor II-III loop peptide regulates skeletal muscle calcium release channel function. J Biol Chem 1995; 270: 18459–64.
Ginsburg KS, Bers DM . Modulation of excitation-contraction coupling by isoproterenol in cardiomyocytes with controlled SR Ca2+ load and Ca2+ current trigger. J Physiol (Lond) 2004; 556: 463–80.
Shannon TR, Chu G, Kranias EG, Bers DM . Phospholamban decreases the energetic efficiency of the sarcoplasmic reticulum Ca pump. J Biol Chem 2001; 276: 7195–201.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China (No 30470900), Natural Science Foundation of Guangdong Province (No 04020253) and SRF for ROCS, SEM.
Rights and permissions
About this article
Cite this article
Shen, Jx. Isoprenaline enhances local Ca2+ release in cardiac myocytes. Acta Pharmacol Sin 27, 927–932 (2006). https://doi.org/10.1111/j.1745-7254.2006.00383.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00383.x