ABSTRACT
Immune reactions to foreign or self-antigens lead to protective immunity and, sometimes, immune disorders such as allergies and autoimmune diseases. Antigen presenting cells (APC) including epidermal Langerhans cells (LCs) play an important role in the course and outcome of the immune reactions. Epidermal powder immunization (EPI) is a technology that offers a tool to manipulate the LCs and the potential to harness the immune reactions towards prevention and treatment of infectious diseases and immune disorders.
Similar content being viewed by others
THE BIOLOGY OF LANGERHANS CELLS
LCs, like the related dendritic cells (DCs), are professional antigen presenting cells. LCs are uniquely present in the epidermis of the skin while DCs are present in the mucosa, dermis, and internal organs. Epidermal LCs form a semi-continuous network in the skin. The density of LCs in most areas of the human skin with the exception of sole and palm is approximately 500-1000 cells/mm2 (an estimated total of 109 LCs for an adult)1, 2, 3. LCs represent a very dynamic cell population. At any given time, there are LCs loaded with antigens leaving the skin and fresh LCs coming in to replenish the sites. The mean residence time in skin for a LC is about 3 w4, 5.
LCs initiate, maintain, and regulate adaptive immunities in the skin. These cells take up epicutaneous antigens, emigrate into the regional skin-draining lymph nodes, and present the processed antigens to the T cells. LCs are derived from bone marrow progenitor cells, which travel through blood to home in the epidermis and become LCs6, 7, 8. Cytokines such as interleukin-4 (IL-4), tumor-necrosis factora (TNF-α), and granulocyte macrophage colony stimulating factor (GM-CSF) are thought to drive the differentiation process of the progenitor cells9. The resident LCs in the epidermis are immature cells possessing strong phagocytic and endocytic capacity. The primary function of the immature LCs is to capture and process antigens. After picking up antigen, LCs migrate across the basal membrane and dermis into draining lymph nodes via lymphatics. Migration is mediated by proinflammatory cytokines including TNF-α and IL-1 that originate from the LCs and other epidermal cells including keratinocytes10, 11. During migration, maturation takes place. Mature LCs express high levels of MHC class I and class II antigens, costimulatory molecules (CD80 and CD86), and chemokine receptors, all of which are important for its antigen presentation function12, 13, 14. Antigen presentation to T cells by LCs takes place in the draining lymph nodes. LCs are capable of presenting antigen to both the naive T cells and antigen-specific T cells of CD4+ and CD8+ phenotypes to stimulate both antibody and cellular immune responses. Induction of CTL responses in mice may require as few as 10 LCs, whereas the induction of an antibody response may require upto 1000 LCs15, 16.
Cytokines secreted by LCs such as IL-1, IL-6, IL-12 and IL-18 play an important role in regulating the immune responses11, 17, 18. These cytokines combined with those secreted by other epidermal cells can polarize the LCs to selectively bias the development of Th1 or Th2 cells. For example, LCs promote the development of a Th1 type response in the presence of a high level of IL-1219. LCs matured in the presence of a high level of IL-10, or in the absence of IL-12, stimulate the maturation of Th2 type responses20, 21. Interaction of LCs with other cells of the immune system results in differential cellular and humoral immune responses and cytokine production (Fig 1) which are important in protection against infections, and sometimes, are associated with immune disorders including allergies22.
A schematic representation of the LCs' life circle and their function of presenting antigens introduced by the skin route. Bone marrow derived LC progenitors travel through blood and home in the epidermis. LCs take up antigens introduced via the skin route and migrate to the draining lymph nodes under the influence of TNFa and IL-1b. LCs present processed antigen to naÏve T cells and induce immune responses that are either Th1 or Th2 types, depending on the cytokine environment. IL-12 promotes a Th1 responses whereas the IL-10 and IL-4 promote a Th2 response. Th2 response is associated with allergic reactions.
Role of LCs/DCs in immunity and immune disorders
Infectious diseases
LCs play an important role in the immune responses to cutaneous infections caused by Herpes simplex virus types 1 and 2, poxvirus, papillomavirus, varicella virus, and other pathogens that cause skin infections23. LCs may contribute to the anti-infection immunity by at least two mechanisms, induction of cellular and humoral immunities by presenting antigen of the infectious agents to the immune system and secretion of cytokines that have direct antiviral activities24. Inoculation of HSV-1 or vaccinia viruses to a skin site depleted of LCs results in higher morbidity and mortality than a normal skin site in animal studies25. Vice versa, activation of LCs locally or systemically correlates with the resistance to infection with these viruses26. LCs not only contribute to the control of primary infections, but also play a role in the control of recurrent viral infections caused by HSV-1 23.
Allergic contact dermatitis (ACD)
ACD is mediated by responses of CD4+ T cells to the contact allergens, mostly low-molecular-weight-haptens. ACD has two phases, sensitization and elicitation. Sensitization refers to events that lead to the activation of T cells whereas the elicitation refers the events that occur when the activated T cells are re-exposed to the same allergen. The sensitization and elicitation phases of ACD are complex processes that involve different cells and molecules27, 28, 29. However, LCs are clearly shown to play an important role in the sensitization process. When applied to the skin, contact allergens bind to the proteins in the epidermis and become a complete antigen. The complete antigen is picked up by LCs or dermal DCs. Some allergens may bind directly to the MHC class II molecules of LCs since topically administered contact allergens accumulate in the LCs of the local skin. Once the LCs have taken up a contact allergen, they migrate to the draining lymph node and present the allergen to the naive CD4 T cells. Activated CD4+ T cells proliferate and home in the peripheral tissues including skin. In the elicitation phase (when exposed to the same allergen again), antigen presentation is still required, this may be accomplished by LCs and other skin cells. Some evidence suggests that allergen-specific T cells can be activated by allergen loaded LCs in the skin to release cytokines, which cause the infiltration of inflammatory cells.
Allergy and Asthma
Recent studies suggest that allergy and asthma are due to eosinophilic inflammation of the airway mediated by Th2 cytokines (IL-4, IL-5, IL-6, and IL-13 etc) (Fig 1) in response to environmental stimulants30. Airway DCs are believed to play an important role in the sensitization to asthma31, 32. The airway DCs reside in the lateral intercellular spaces of the basal epithelial cells and form a network of cells in a way similar to LCs in the skin. Inhaled allergens are captured, processed, transported to the regional lymph nodes and presented to naive T cells. The low levels or absence of IL-12 and the abundance of IL-4 and IL-10 cytokines are believed to be important in selectively biasing the development of Th2 cells. Some Th2 cytokines such as IL-3, IL-5 and GM-CSF cause airway eosinophilia, whereas other Th2 cytokines (IL-4, IL-13) cause the production of IgE antibodies by B cells. In the elicitation phase, allergen introduced cross-linking of antigen-specific IgE antibody bound to mast cells through high-affinity receptors triggers mast cells to release inflammatory mediators and subsequent sustained infiltration of eosinophils. Increased LC density has been observed in the affected tissue during the allergic inflammation, which is thought to be important in controlling and maintaining the inflammation.
Tumor
Functional impairment of LCs in the skin is believed to increase the susceptibility of tumor development upon exposure to carcinogens, UV light, or oncogenic viral infections (e.g. papillomavirus) 33, 34. In some cases, it is observed that presence of a large number of LCs in the tumor is of prognostic value35, 36. LCs are believed to contribute to the immune protection against tumors by directly secreting cytokines with anti-tumor activity and evoking systemic immune responses to tumor associated antigens (TAA).
Technologies that target LCs for immunization
Given the importance of APCs in the initiation and regulation of immune responses, a number of strategies and techniques have been examined in the past decade to employ LCs/DCs in the prophylactic and therapeutic immunizations against infectious diseases and immune disorders. Most technologies focus on the use of DCs because they can be generated in vitro from peripheral blood monocytes or bone marrow cells37, 38. The ex vivo DC-based vaccines are widely examined for cancer immunotherapy. This strategy involves in vitro generation of DCs, antigen loading, and transfusion of cells to the patient. In comparison, far fewer technologies that exploit epidermal LCs in immunization exist. The epidermis is too thin to be accessed by traditional syringe and needle injection. Application of vaccine antigens to the surface of an intact skin has been explored. Transcutaneous immunization (TCI) is a technology that topically delivers liquid vaccines using cholera toxin adjuvant39. Antibody responses were elicited to several protein antigens including diphtheria and tetanus toxoids, cholera toxin (CT), heat-labile toxin from E. coli (LT), and bovine serum albumin in mice39. TCI of humans with LT causes morphological changes in LCs, suggesting that LCs may play a role in the antibody response40. Topical immunization techniques may not be effective with large vaccine molecules or particle vaccine formulations (e.g. inactivated whole pathogens, or alum-adsorbed vaccines) because of the barrier function of the stratum corneum. Here we will focus discussion on a unique technology, Epidermal Powder Immunization (EPI), which enables efficient delivery of vaccine antigens to the LC rich epidermis.
EPI is a novel technology that was developed to target antigens to LCs in vivo. EPI has its roots in a technology that was developed in the early1990s for genetically engineering plants and then was adapted for DNA immunization41, 42, 43. EPI delivers antigens in the form of microscopic particles to the epidermis using a needle-free powder delivery system (or PowderJect device) and elicits broad immune responses44. Here we will review our findings using this technology to deliver traditional vaccines in relation to LC targeting and its potential applications in the prevention and treatment of infectious diseases and immune disorders.
Many traditional vaccines such as proteins, peptides, polysaccharides, inactivated pathogens etc are suitable for EPI44, 45, 46, 47. Vaccine powders can be prepared by coating antigens onto 1-2 mm gold particles or embedding them into 20-50 mm particles using sugar excipients (trehalose, mannitol, sucrose, or combinations)44, 47. The driving force of the device is pressured helium gas. Actuation of the device causes the release of helium gas, which accelerates the vaccine particles to high velocity that penetrate the stratum corneum and land in the LC rich viable epidermis. In addition to targeting antigen to the LC rich epidermis, EPI offers the advantage of pain-free delivery. This is because the sensory nerve endings in the epidermis are far less dense than deeper tissues such as dermis and muscle.
A. Evidence for targeting LCs
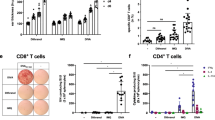
Histological data shows that EPI delivers powdered vaccines into the LC rich viable epidermis. Antigen targeting of LCs in the target tissue following EPI is dependent on whether gold or sugar formulation was used. EPI with the gold formulation results in intracellular deposition of both gold and antigen in the epidermal cells47. LCs account for about 3% of the total number of the epidermal cells (Fig 2A), thus, a fraction of particles is directly delivered to LCs. EPI delivers antigens to a skin target of approximately 110 mm2, which comprises approximately 1×105 LCs47. Delivering a 0.5 mg of gold coated with antigen results in up to 50% of the LCs at the vaccination site containing antigen-coated particles as revealed by an immunofluorescence microscopy(47) (Fig 2B). The number of antigen-loaded LCs is sufficient to evoke an immune response.
EPI targets antigens to LCs. A. Normal LCs (green) in the epidermal sheet stained using Mab to I-Ad antigen conjugated to fluorescein isothiocyanate (FITC) (60×). B. EPI using gold particles delivers antigen to the cytoplasm of LCs. EPI was performed using gold particles (1–2 μm) coated with TR-OVA (model antigen). Immediately after EPI, Epidermal sheet from the site of immunization was prepared and LCs were stained with Mab to I-Ad antigen conjugated to FITC. The gold particles inside the green-colored LCs are in yellow color, whereas the gold particles in non-fluorescent cells (keratinocytes) are in red color (60×). C. Epidermal sheet from the site of EPI using sugar particles (20-50 μm) containing TR-OVA and black tissue marking dye viewed under a light microscope (60×). Black spots indicate the footprint of vaccine particles. D. The same field shown in C viewed under an UV light. LCs (FITC-stained) become yellow once they take up antigen (60×) after the dissolution of vaccine-containing sugar particles. E. Migration of LCs from the vaccination site to the draining lymph node. The vaccination sites were treated with FITC (dissolved in DMF) by topical application 2 h prior to EPI with TR-OVA coated gold particles. Fluorescent cells in the draining lymph nodes were examined 20 h later by fluorescent microscopy under an UV light. The green cells are LCs that contain TR-OVA coated gold particles originating from the site of EPI (240×).
EPI with a sugar formulation results in a different antigen distribution pattern than the gold formulation. The footprint of the sugar particles can be located by a tissue marking black dye, which binds to the cells with minimal diffusion after particle dissolution48. The discrete dark spots are visible under a light microscope (Fig 2C). Following sugar particle dissolution, the Tax Red labeled ovalbumin (TR-OVA) diffuses into the surrounding cells and appears to be picked up by LCs. Nearly all LCs in the target tissue appear to contain antigens (Fig 2D). Antigen and antigen carrying LCs are present at the site of immunization for up to 5 d although the concentrations are greatly reduced after the first 48 h (unpublished data).
EPI inherently activates LCs in the target site as evidenced by enlarged cell body and brighter staining with an I-Ad specific monoclonal antibody (enhanced expression of class II antigens)48. LC activation is probably mediated by cytokines produced by epidermal cells when triggered by the particle penetration. In vitro culture of epidermal sheets prepared from the immunization site result in the migration of LCs into the culture medium. In vivo, antigen-carrying LCs can be shown in the regional draining lymph nodes starting 20 h after the immunization and lasting for several days (Fig 2E). More recent studies indicate that transfer of LCs isolated from the immunization site to naive mice induces antigen-specific antibody responses, confirming that LCs are the antigen presenting cells (unpublished data).
B. Breadth of the immune responses induced by EPI
B1. Serum antibody responses: EPI elicits strong serum antibody responses in mice to a variety of antigens including inactivated influenza viruses, diphtheria toxoid, hepatitis B surface antigen (HBsAg), and HIV gp120. Using the same vaccine, EPI elicited significantly higher serum titers than syringe and needle injection via common routes (e.g. subcutaneous, intramuscular, or intraperitoneal). Dose range studies indicate that EPI may require a considerably smaller antigen dose than needle injection to achieve maximal immunity44, 48. The serum antibody titers are as long-lasting as those elicited by needle injection45. Recently, we showed that EPI elicits high serum antibody titers to influenza vaccine and HBsAg in monkeys, which have an immune system resembling that of humans.
B2. Mucosal antibodies. The majority of infectious agents gain entry to the human body through mucosal tissues, thus mucosal antibodies, i.e. secretory IgA (SIgA), in the mucosal surface are vitally important for protection. The immune system of humans and animals is compartmentalized in such a way that mucosal antibodies are only induced by direct application of vaccines to mucosal sites, whereas vaccination by a non-mucosal route (i.e. intramuscularly) normally does not elicit a mucosal immunity. However, recent data has shown that the skin immune system appears to cross talk with the mucosal immune system.
Following EPI of mice with an inactivated influenza vaccine, antigen-specific SIgA antibodies were detected in mucosal lavages of the small intestine, trachea, and vaginal tract46. The local origin of the SIgA antibodies was further shown by measuring antibodies released from cultured tracheal and small intestinal fragments and by detecting antigen-specific IgA secreting cells in the lamina propria using ELISPOT assays. These antibodies appear to be important for protection against experimental challenge. An adjuvant appears to be required for EPI to induce a mucosal antibody response. CT, LT, a synthetic oligodeoxynucleotide containing immunostimulatory CpG motifs (CpG DNA), and many other adjuvants were found to enhance the mucosal antibody responses to co-delivered antigens.
B3. Cytotoxic T lymphocyte response: CTLs play a vital role in host defense against viral and intracellular bacterial infections. However, non-replicating vaccines administered by intramuscular injection using a syringe and needle elicit predominantly humoral responses but not CTL responses. Proteins in the cytosol are processed and presented under the restriction of MHC class I molecules and induce CTL responses. EPI using gold particles delivers proteins to the cytosol of LCs and other epidermal cells. CTL responses were observed to HBsAg, HIV gp120, and an influenza virus nucleoprotein peptide in mice when loaded on to gold particles and administered by EPI47, 48. The CTL responses were similar to the responses elicited by DNA vaccines and live-attenuated vaccines. EPI with sugar powder formulations does not normally elicit a CTL response44. This is because antigens in the sugar formulation are delivered extracellularly. These antigens are taken up by LCs via phagocytosis and endocytosis.
B4. T helper responses and immunomodulation with adjuvants: The T helper cell responses (Th1 vs Th2) following EPI have been examined using a number of antigens. The T helper cell responses are dependent on the antigen dose and whether adjuvant is used. In the absence of an adjuvant, both diphtheria toxoid and HBsAg induce a Th2 response as reflected by the production of low level of IFN-γ and a high IgG1/IgG2a antibody ratio45. When decreasing antigen doses were tested, the Th2 response shifted toward the Th1 response (unpublished data). The T helper cell responses can be shifted towards Th1 or Th2 when appropriate adjuvants are used. For example, CpG DNA, QS-21 and LT adjuvants cause a shift to the Th1 response whereas the aluminum adjuvants and PCPP promote a Th2 response45.
C. Potential applications
C1. Infectious diseases. EPI can elicit high levels of serum antibody, which are important for prophylactic immunization against infection by extracellular bacteria and many viruses. Mucosal antibodies are particularly important because these antibodies may prevent pathogens from gaining entry to the deeper tissue. EPI may have immunotherapy potential for chronic infections caused by viruses and intracellular bacteria since EPI with antigen coated gold particles elicit a CTL response, which may help to cure established infections by eliminating infected cells. It is also important to note that EPI may allow the use of new and more potent adjuvants in routine human vaccination, which otherwise may not be safe when administered into a deeper tissue. This is because adjuvants administered by EPI appear to be gradually released from the immunization site to deeper tissue over a period of days, thus reducing the peak serum concentration and systemic toxicity. Furthermore, periodic sloughing and regeneration of the epidermis remove any residual adjuvant in the skin that may cause chronic local reactions, a problem seen with deeper tissue injection.
C2. Cancer immunotherapy: Immunizations with whole tumor cells or lysates, and recently with tumor-associated (TAA) antigens by traditional methods have produced some promising clinical results49. Antigen presentation is a critical regulatory element for the induction of humoral and cellular immune responses to tumors. DCs are now being widely explored for immunotherapy against a variety of tumors. The commonly used technique for DC-based cancer immunotherapy involves ex vivo cultivation of autologous DCs from the peripheral blood cells, in vitro antigen loading, and the transfer of DCs to the patients.
EPI represents an in vivo technique for developing DC-based immunotherapy. It directly delivers antigens to the cytosol of the LCs and elicits both antibody and cellular immune responses. EPI is suitable for delivering tumor cell lysates, purified antigen, and other immunostimulating agents (e.g. GM-CSF). It is technically much more suitable for commercialization when compared with ex vivo DC-based approaches.
C3. Immunotherapy for allergic diseases. Allergic diseases including ACD, asthma, and hay fever are commonly treated with corticosteroids and anti-histamine drugs. Immunotherapy by subcutaneous injection of allergen over a prolonged period has been used to treat several types of allergies with variable clinical effects. It is effective in inducing prolonged remission of insect venom anaphylaxis, but is only moderately effective against hay fever, and even less effective against asthma. Although the mechanism of immune therapy in humans is not fully understood, studies using animal models suggest that immunization strategies promoting a Th1 response can decrease the formation of IgE antibodies and eosinophilia50. Alternatively, formation of IgG antibodies will neutralize the allergens before being captured by the IgE antibodies. EPI using antigen and an appropriate adjuvant (CpG DNA, saponin, etc) promoted strong Th1 responses in an animal model, suggesting that it may be possible to reprogram the immune system of the sensitized individual and offers a more effective means of allergy immunotherapy. Given the concept that the immune system is compartmentalized, administering treatment to skin by EPI may lead to maximal therapeutic effect in the skin and related mucosal tissues, which are normally the affected targets of allergies.
CONCLUSIONS
Given the central role of LCs and DCs in the initiation and regulation of the immune response to infectious pathogens, allergens, and tumor antigens, immunization strategies that target LCs/DCs may lead to more effective therapies against these diseases. Developing technologies for effective in vivo or in vitro targeting of LCs/DCs are ultimately important. EPI delivers antigen to the LC rich skin and generates robust immune responses in animal models. The quality of the immune responses (TH1 vs. TH2, mucosal vs. systemic, cellular vs. humoral) generated by EPI can potentially be tailored to specific therapeutic goals by choosing appropriate carrier particles and co-administration of an immunomodulator/adjuvant.
References
Chen H, Yuan J, Wang Y, and Silvers WK . Distribution of ATPase-positive Langerhans cells in normal adult skin. Br J Dermatol 1985; 113:707–11.
Berman B, Chen VL, France DS, Dotz WI, Petroni, G . Anatomical mapping of epidermal Langerhans cell density in adults. Br J Dermatol 1983; 109(5):553–8.
Bos JD, Zonneveld I, Das PK, Krieg SR, van der Loos CM, Kapsenberg ML . The skin immune system (SIS): distribution and immunophenotype of lymphocyte subpopulations in normal human skin. J Invest Dermatol 1987; 88(5):569–73.
Teunissen MB . Dynamic nature and function of epidermal Langerhans cells in vivo and in vitro: a review, with emphasis on human Langerhans cells. Histochem J 1992; 24(10):697–716.
Romani N, Ratzinger G, Pfaller K, Salvenmoser W, Stossel H, Koch F, Stoitzner P . Migration of dendritic cells into lymphatics-the Langerhans cell example: routes, regulation, and relevance. Int Rev Cytol 2001; 207:237–70.
Gordyal P, Isaacson PG . Immunocytochemical characterization of monocyte colonies of human bone marrow: a clue to the origin of Langerhans cells and interdigitating reticulum cells. J Pathol 1985; 146(3):189–95.
Dezutter-Dambuyant C, Schmitt D, Faure M, Cordier G, Thivolet J . Detection of OKT6-positive cells (without visible Birbeck granules) in normal peripheral blood. Immunol Lett 1984; 8(3):121–6.
Jakob T, Ring J, Udey MC . Multistep navigation of Langerhans/dendritic cells in and out of the skin. J Allergy Clin Immunol 2001; 108(5):688–96.
Pieri L, Domenici L, Romagnoli P . Langerhans cells differentiation: a three-act play. Ital J Anat Embryol 2001; 106(1):47–69.
Romani N, Ratzinger G, Pfaller K, Salvenmoser W, Stossel H, Koch F, Stoitzner P . Migration of dendritic cells into lymphatics-the Langerhans cell example: routes, regulation, and relevance. Int Rev Cytol 2001; 207:237–70.
Wang B, Amerio P, Sauder DN . Role of cytokines in epidermal Langerhans cell migration. J Leukoc Biol 1999; 66(1):33–9.
Cruz PD Jr, Bergstresser PR . Antigen processing and presentation by epidermal Langerhans cells. Induction of immunity or unresponsiveness. Dermatol Clin 1990; 8(4):633–47.
Romani N, Koide S, Crowley M, Witmer-Pack M, Livingstone AM, Fathman CG, Inaba K, Steinman RM . Presentation of exogenous protein antigens by dendritic cells to T cell clones. Intact protein is presented best by immature, epidermal Langerhans cells. J Exp Med 1989; 169(3):1169–78.
Schuler G, Steinman RM . Murine epidermal Langerhans cells mature into potent immunostimulatory dendritic cells in vitro. J Exp Med 1985; 161(3):526–46.
Timares L, Takashima A, Johnston SA : Quantitative analysis of the immunopotency of genetically transfected dentritic cells. Proc Natl Acad Sci USA 1998; 96:13147–52.
McKinney EC, Streilein LM : On the extraordinary capacity of allogeneic epidermal Langerhans cells to prime cytotoxic T cells in vivo. J Immunol 1989; 143:1560–4.
Schroder JM . Cytokine networks in the skin. J Invest Dermatol 1995; 105(1 Suppl):20S–4S.
Hochrein H, Shortman K, Vremec D, Scott B, Hertzog P, O'Keeffe M . Differential production of IL-12, IFN-alpha, and IFN-gamma by mouse dendritic cell subsets. J Immunol 2001; 166(9):5448–55.
Kalinski P, Hilkens CM, Snijders A, Snijdewint FG, Kapsenberg ML . IL-12-deficient dendritic cells, generated in the presence of prostaglandin E2, promote type 2 cytokine production in maturing human naive T helper cells. J Immunol 1997; 159(1):28–35.
De Smedt T, Van Mechelen M, De Becker G, Urbain J, Leo O, Moser M . Effect of interleukin-10 on dendritic cell maturation and function. Eur J Immunol 1997; 27(5):1229–35.
Reider N, Reider D, Ebner S, Holzmann S, Herold M, Fritsch P, Romani N . Dendritic cells contribute to the development of atopy by an insufficiency in IL-12 production. J Allergy Clin Immunol 2002; 109:89–95.
Banchereau, J Steinman RM . Dendritic cells and the control of immunity. Nature. 1998; 392(6673):245–52.
Sprecher E, Becker Y . Role of Langerhans cells and other dendritic cells in viral diseases. Arch Virol 1993; 132(1–2):1–28.
Williams NA, Hill TJ, Hooper DC . Murine epidermal antigen-presenting cells in primary and secondary T-cell proliferative responses to herpes simplex virus in vitro. Immunology. 1991; 72(1):34–9.
Otani T, Mori R . The effects of ultraviolet irradiation of the skin on herpes simplex virus infection: alteration in immune function mediated by epidermal cells and in the course of infection. Arch Virol 1987; 96(1–2):1–15.
Sprecher E, Becker Y . Langerhans cell density and activity in mouse skin and lymph nodes affect herpes simplex type 1 (HSV-1) pathogenicity. Arch Virol 1989; 107(3–4):191–205.
Xu H, Bjarnason B, Elmets CA . Sensitization versus elicitation in allergic contact dermatitis: potential differences at cellular and molecular levels. Am J Contact Dermat 2000; 11(4):228–34.
Kimber I, Dearman RJ, Cumberbatch M, Huby RJ . Langerhans cells and chemical allergy. Curr Opin Immunol 1998; 10(6):614–9.
Tsuruta D, Kaneda K, Teramae H, Ishii M . In vivo activation of Langerhans cells and dendritic epidermal T cells in the elicitation phase of murine contact hypersensitivity. Br J Dermatol 1999; 140(3):392–9.
Holgate ST . The epidemic of allergy and asthma. Nature 1999; 402 (6760 Suppl):B2–4.
von Bubnoff D, Geiger E, Bieber T . Antigen-presenting cells in allergy. J Allergy Clin Immunol 2001; 108(3):329–39.
Lambrecht BN, Hoogsteden HC, Pauwels RA . Dendritic cells as regulators of the immune response to inhaled allergen: recent findings in animal models of asthma. Int Arch Allergy Immunol 2001; 124(4):432–46.
Meunier L, Raison-Peyron N, Meynadier J . UV-induced immunosuppression and skin cancers. Rev Med Interne 1998; 19(4):247–54.
Muller HK, Dandie GW, Ragg SJ, Woods GM . Langerhans cell alterations in cutaneous carcinogenesis. In Vivo 1993; 7(3):293–6.
Miyagi J, Kinjo T, Tsuhako K, Higa M, Iwamasa T, Kamada Y, Hirayasu T . Extremely high Langerhans cell infiltration contributes to the favourable prognosis of HPV-infected squamous cell carcinoma and adenocarcinoma of the lung. Histopathology. 2001; 38(4):355–67.
Johnson SK, Kerr KM, Chapman AD, Kennedy MM, King G, Cockburn JS, Jeffrey RR . Immune cell infiltrates and prognosis in primary carcinoma of the lung. Lung Cancer 2000; 27(1):27–35.
Kumamoto T, Morita A, Takashima A . Recent advances in dendritic cell vaccines for cancer treatment. J Dermatol 2001; 28(11):658–62.
Nouri-Shirazi M, Banchereau J, Fay J, Palucka K . Dendritic cell based tumor vaccines. Immunol Lett 2000; 74(1):5–10.
Hammond SA, Guebre-Xabier M, Yu J, Glenn GM . Transcutaneous immunization: an emerging route of immunization and potent immunostimulation strategy. Crit Rev Ther Drug Carrier Syst 2001; 18(5):503–26.
Glenn GM, Taylor DN, Li X, Frankel S, Montemarano A, Alving CR . Transcutaneous immunization: a human vaccine delivery strategy using a patch. Nat Med 2000; 6(12):1403–6.
Johnston SA, Anziano PQ, Shark K, Sanford JC, Butow RA . Mitochondrial transformation in yeast by bombardment with microprojectiles. Science. 1988; 240(4858):1538–41.
Ye GN, Daniell H, Sanford JC . Optimization of delivery of foreign DNA into higher-plant chloroplasts. Plant Mol Biol 1990; 15(6):809–19.
Williams RS, Johnston SA, Riedy M, DeVit MJ, McElligott SG, Sanford JC . Introduction of foreign genes into tissues of living mice by DNA-coated microprojectiles. Proc Natl Acad Sci USA 1991; 88(7):2726–30.
Chen D, Endres RL, Erickson CA, Weis KF, McGregor MW, Kawaoka Y, Payne LG . Epidermal immunization by a needle-free powder delivery technology: Immunogenicity of influenza vaccine and protection in mice. Nat Med 2000; 6:1187–90.
Chen D, Erickson CA, Endres RL, Periwal SB, Chu Q, Shu C, Maa YF, Payne LG . Adjuvantation of epidermal powder immunization. Vaccine 2001; 19:2908–17.
Chen D, Periwal SB, Larrivee K, Zuleger C, Erickson CA, Endres RL, Payne LG . Serum and Mucosal Immune Responses to an Inactivated Influenza Virus Vaccine Induced by Epidermal Powder Immunization. J Virol 2001; 75:7956–65.
Chen D, Weis KF, Chu Q, Erickson C, Endres R, Lively CR, Osorio J, Payne LG . Epidermal powder immunization induces both cytotoxic T lymphocyte and antibody responses to protein antigens of influenza and hepatitis B viruses. J Virol 2001; 75:1630–40.
Chen D, Zuleger C, Chu Q, Maa YF, Osorio J, Payne, LG . Epidermal Powder Immunization with a Recombinant HIV gp120 Targets Langerhans Cells and Induces both Antibody and Cellular Immune Responses. Submitted to AIDS Research and Human Retroviruses.
Moingeon P . Cancer vaccines. Vaccine 2001; 19:1305–26.
Wohlleben G, Erb KJ . Barnes P . Atopic disorders: a vaccine around the corner? Trends Immunol 2001; 22(11):618–26.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
CHEN, D., PAYNE, L. Targeting epidermal Langerhans cells by epidermal powder immunization. Cell Res 12, 97–104 (2002). https://doi.org/10.1038/sj.cr.7290115
Issue Date:
DOI: https://doi.org/10.1038/sj.cr.7290115
Keywords
This article is cited by
-
Exploitation of Langerhans cells for in vivo DNA vaccine delivery into the lymph nodes
Gene Therapy (2014)
-
Genetic vaccination approaches against malaria based on the circumsporozoite protein
Wiener klinische Wochenschrift (2006)