ABSTRACT
Programmed cell death plays an important role in maintaining homeostasis during animal development, and has been conserved in animals as different as nematodes and humans. Recent studies of Drosophila have provided valuable information toward our understanding of genetic regulation of death. Different signals trigger the novel death regulators rpr, hid, and grim, that utilize the evolutionarily conserved iap and ark genes to modulate caspase function. Subsequent removal of dying cells also appears to be accomplished by conserved mechanisms. The similarity between Drosophila and human in cell death signaling pathways illustrate the promise of fruit flies as a model system to elucidate the mechanisms underlying regulation of programmed cell death.
Similar content being viewed by others
INTRODUCTION
During development of multicellular organisms, programmed cell death plays an important role in the maintenance of homeostasis by eliminating obsolete cells or tissues, controlling cell numbers, and removing damaged cells1. Studies of developing animals categorized physiological cell death into three classes: apoptosis, autophagy, and non-lysosomal cell death2,3. Apoptosis is characterized by a series of cellular changes including chromatin condensation, DNA fragmentation, blebbing of the plasma membrane, fragmentation of cytoplasm into apoptotic bodies, and removal by phagocytes4. Therefore, debris of dying cells is contained rather than released, and this process is thought to prevent inflammation. Apoptosis is usually observed in isolated cells, like the death of immature T lymphocytes5. Cells that die by autophagy possess large autophagic vacuoles that are involved in the destruction of cytosolic contents. This type of cell death is usually found in regions where tissues are removed into to such as the epithelial plates following closure of the palate3. While phagocytes have been associated with cells that die by autophagy, the relationship of these cells is unclear2. Non-lysosomal cell death is least characterized, and is rarely observed. The mechanisms that regulate autophagy and non-lysosomal cell death are not well understood and, thus, most of this review will emphasize apoptosis.
Pioneering studies of the nematode C. elegans led to the identification of genes that function in apoptosis6. ced-3 and ced-4 loss-of-function and ced-9 gain-of-function mutants live and possess additional cells in adult nematodes7,8. ced-9loss-of-function mutants, on the other hand, die during embryogenesis due to ectopic cell death8. This lethality is suppressed by loss-of-function mutations in ced-3 and ced-4 indicating that these two genes act downstream or in parallel to ced-9. Cell death induced by ectopic expression of ced-4 is reduced in ced-3 mutants, indicating that ced-4 is likely to function upstream of ced-3. This paradigm is further supported by biochemical studies showing that CED-9 binds to CED-4, and CED-4 physically interacts with CED-3, leading to the activation of CED-39, 10, 11, 12.
The ced-3, ced-4 and ced-9 genes are conserved in mammals13. CED-3 encodes a protein that is similar to a family of cystein proteases collectively known as caspases, which upon proteolytic activation cleave cellular substrates14. CED-4 exhibits sequence homology to Apaf-1, and has been shown to be involved in activation of pro-caspases in the presence of cytochrome c and dATP15, 16, 17. CED-9 is homologous to the Bcl-2 family of oncogenes,that include both antiapoptotic and proapoptotic members18.
Drosophila melanogaster possesses many of the signal transduction pathways that play important roles during human development19. The strength of Drosophila as a model for molecular genetic studies, and the recent completion of the Drosophila genome sequence20, establishes fruit flies as an important model organism for biomedical research. In this review, we describe the current knowledge of genetic regulation of programmed cell death in Drosophila.
Conserved regulators of cell death in Drosophila
Caspases are a family of evolutionarily conserved proteins that are central components of the apoptotic machinery. Caspases are synthesized as inactive zymogens that are comprised of two domains: a prodomain and a catalytic domain21. Following proteolytic cleavage of caspase zymogens at a conserved aspartate residue, the prodomain is separated from the catalytic domain, which becomes further processed to an active form. Currently, there are at least 14 caspases in humans, and this family of proteases is divided into initiator and effector caspases. Initiator caspases contain a long prodomain, which is responsible for protein-protein interactions, and this interaction is important for caspase activation. Initiator caspases are thought to regulate the activation of inactive effector caspases.
In Drosophila, there are five known caspases, DRONC, DREDD/DCP-2, DCP-1, DrICE, and DECAY22, 23, 24, 25, 26, 27, and three additional caspases have been predicted based on genomic sequence. DRONC and DREDD possess a long prodomain and, therefore, resemble human initiator caspases 8 and 9. DCP-1, DrICE, and DECAY contain a short prodomain and are similar to human effector caspases like caspases 3 and 7. Chromosomal deletions of the dredd or dronc loci can dominantly modulate the amount of apoptosis suggesting that the relative level of these genes regulates programmed cell death22,28. Furthermore, dredd and dronc are regulated at both the transcriptional and post-translational levels. dredd mRNA accumulates in a pattern that mimics the pattern of cell death during embryogenesis, and fails to accumulate in mutant embryos that lack cell death22. Consistent with this notion, dredd mRNA is ectopically accumulated in cells that are induced to die by death signals. Although dronc is ubiquitously expressed during embryogenesis, the level of dronc mRNA is dramatically up-regulated in larval salivary glands and midguts when they undergo hormone-triggered programmed cell death. Furthermore, dronc mRNA can be prematurely induced upon exposure to hormone, and the transcription of this gene is abolished in mutants that possess defects in larval salivary gland cell death24, 29. DREDD and DRONC are also subject to post-translational regulation. Upon the induction of apoptosis, DREDD zymogens undergo initial proteolytic cleavage in the presence of the broad-spectrum caspase inhibitor p35 and anti-caspase peptides22. This argues that this process may be regulated by another type of protease or by a caspase that is insensitive to these inhibitors. Furthermore, the prodomain of DRONC has recently been shown to interact with the Drosophila Inhibitor of Apoptosis Protein 1 (DIAP1), and this interaction is necessary for DIAP1 to exert its inhibitory effects on DRONC28.
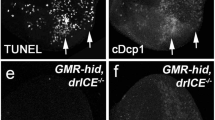
Effector caspases proteolytically cleave different cellular components, and are thought to be responsible for apoptotic morphological changes30. Among the three characterized Drosophila effector caspases, loss-of-function mutations have only been identified in dcp-1 to date27. Homozygous dcp-1 mutants die during larval development and possess melanotic tumors. dcp-1 mutant oocytes exhibit defects in nuclear lamin breakdown, which may be directly attributed to the loss of DCP-1 protease activity, as purified DCP-1 protein is capable of cleaving lamin31. In addition, these dcp-1 mutant oocytes display defects in the reorganization of actin cytoskeletal proteins. These oocyte cytoskeletal changes are similar to events that have been reported in cells undergoing apoptosis. Caspases also indirectly target proteins for degradation via the activation of other proteolytic enzymes. One example is the degradation of chromosomal DNA into nucleosomal units, an apoptotic hallmark. In mammals, DNA fragmentation is mediated by a caspase-activated DNase, CAD32. In living cells, CAD forms a complex with its inhibitor ICAD that prevents DNase activity. Upon proteolysis of ICAD by caspases, CAD DNase activity is released. Drosophila possesses homologs of both CAD and ICAD, named DCAD and DICAD, and they are biochemically similar to their mammalian homologs33,34.
The Drosophila A paf-1-related killer ( ark) possesses sequence similarity to human Apaf-135, 36, 37. Unlike CED-4 in nematodes, Ark and Apaf-1 possess a WD-repeat domain in their carboxyl termini. This domain is required for interacting with Cytochrome c, which is involved in caspase activation38. ark loss-of-function mutants exhibit defects in apoptosis, form melanotic tumors, and possess enlarged central nervous systems and defective adult wings35, 36, 37. Homozygous ark mutants suppress ectopic induction of apoptosis by the Drosophila cell death genes reaper ( rpr), head involution defective ( hid), and grim in developing adult eyes suggesting that ark function is required for these genes to activate a death program35, 36, 37. The pattern of ark mRNA expression is similar to the pattern of naturally occurring cell death during embryogenesis. ark mRNA can be ectopically induced upon exposure to X-ray and UV irradiation, and the distribution of ark mRNA closely matches the pattern of ectopic cell death35. Furthermore, Ark physically interacts with DREDD and DRONC, but not DrICE, consistent with its similarity with human Apaf-136. It is unclear if this interaction is required for the activation of DREDD and DRONC under physiological conditions. Similar to mammalian Apaf-1, ark is thought to be involved in the amplification of death signals, as some cell death still occurs in homozygous ark mutants.
Two orthologs of ced-9/ Bcl-2 named drob-1/debcl/dborg-1 and dborg-2 have been identified in Drosophila39, 40, 41, 42. Both of these genes encode proteins that exhibit sequence homology to the pro-apoptotic type of mammalian Bcl-2 proteins. Consistent with this similarity, ectopic expression of drob-1 is sufficient to partially ablate Drosophila adult compound eyes by apoptosis39, 40. No specific mutations exist in these Drosophila Bcl-2-like genes. However, RNAi experiments suggest that drob-1 may function in developmentally regulated cell death during embryogenesis42. drob-1 might also play a role in radiation-induced cell death, as drob-1 expression potentiates UV irradiation-induced apoptosis42. drob-1-induced cell death is partially suppressed by the caspase inhibitor p35, suggesting that this gene may act upstream of caspases.
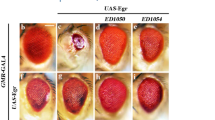
Three IAPs, DIAP1, DIAP2, and Deterin have been identified43,44. Similar to IAPs in other species, these fly proteins possess a number of Baculovirus Inhibitor of apoptosis Repeat (BIR) domains and a RING finger motif. Expression of either DIAP1 or DIAP2 is sufficient to suppress the ablation of adult compound eyes induced by ectopic expression of the Drosophila death genes rpr, hid, and grim. diap2 is repressed by the steroid hormone 20-hydroxyecdysone during , Drosophila metamorphosis, however, the precise function of this gene is not yet known45. diap1 mutations dominantly enhance the ablation of eyes induced by expression of Drosophila cell death genes44. DIAP1 inhibits the activity of the Drosophila caspases DCP-1, DrICE and DRONC28,46, 47. Consistent with this observation, homozygous diap 1 mutant animals are arrested during early embryonic development and exhibit a global increase in the level of apoptosis due to ectopic caspase activities46,48, 49. Taken together, these data indicate that the physiological function of diap1 is to maintain cell survival by preventing apoptosis through the inhibition of caspase activities.
Novel regulators of cell death in Drosophila
A genetic screen was conducted to identify mutations that would result in an absence of cell death in Drosophila 50. This screen was designed to distinguish mutations that directly impact the activation apoptosis from mutations that result in developmental defects leading to the promotion of apoptosis. This experiment resulted in the identification of the H99 region, which is required for almost all programmed cell death during Drosophila embryogenesis. This genetic locus is not responsible for the cell death machinery itself, as apoptosis can still be induced in H99 homozygous mutant embryos. H99 is a chromosome deletion that removes 300 Kb of the 75CD region of the fly genome, and contains the rpr, hid and grim cell death genes50, 51, 52.
The rpr, hid, and grim genes encode novel proteins that exhibit no sequence homology with known mammalian genes. However, these three genes share sequence similarity in their first 14 amino acids51,52. The expression patterns of rpr and grim mRNA correspond to the pattern of apoptosis, suggesting that these two genes may function specifically in cell death50,52. In contrast, hid is also expressed in cells that live51. Antibody staining indicates that Hid protein is associated with mitochondria53. Ectopic expression of either rpr, hid or grim is sufficient to ablate adult compound eyes by activating apoptosis, and this eye ablation phenotype can be suppressed by co-expression of the caspase inhibitor p3551,52, 54. Under physiological conditions, however, induction of apoptosis requires combinatorial functions of these cell death genes, as in embryonic central nervous system midline cells, the normal pattern of cell death requires the functions of multiple genes in the H99 region55. Furthermore, rpr mRNA is induced anticipating apoptosis that is activated by radiation and defective development56. While loss-of-function mutations have not been isolated in the rpr and grim genes, homozygous hid mutant embryos exhibit a reduced apoptosis phenotype51. H99 mutant embryos die during embryogenesis with defects in head involution, and this phenotype is due to a lack of apoptosis.
Recent studies have provided clues into the molecular mechanisms by which rpr, hid and grim activate apoptosis, and suggest that there is a complex interaction among cell death genes rpr, hid, grim, dcp-1, drice, and diap146, 47, 48, 49,57. RPR, HID and GRIM activate apoptosis by inhibiting DIAP1's ability to suppress caspases, as a gain-of-function mutation of diap1 suppresses rpr, hid, and grim-induced cell death. This result is consistent with studies indicating that DIAP1 can physically interact with RPR, HID and GRIM. In addition, rpr and grim, but not hid, have been shown to activate DCP-1 zymogens, while rpr, but not hid or grim, is able to activate pro-DrICE.
Activation of programmed cell death
Programmed cell death is an integral component of normal development in animals. Physiological signals, such as cell-cell interactions and steroid hormones, trigger programmed cell death to control differentiation and morphogenesis of tissues and to eliminate unneeded cells58, 59, 60. In addition, cellular stresses such as radiation activate apoptosis in order to minimize damage to the organism61. Therefore, it is very important to understand how programmed cell death is activated and integrated to maintain homeostasis during development.
Steroid hormones coordinate multiple cellular responses during animal development. During Drosophila metamorphosis, the steroid hormone 20-hydroxyecdysone (ecdysone) regulates diverse cellular responses including differentiation and morphogenesis, remodeling and programmed cell death to allow a complete transformation of the body plan from a crawling larva to a highly mobile adult fly58. At the end of the larval life, a pulse of ecdysone induces metamorphosis and triggers the destruction of the larval midgut and anterior muscles, morphogenesis and differentiation of adult structures, and remodeling of the central nervous system62, 63, 64, 65. Approximately 12 h following the onset of metamorphosis, another pulse of ecdysone induces the destruction of larval salivary glands63,65, 66. The destruction of larval salivary glands and midgut are accompanied by hallmarks of apoptosis including DNA fragmentation and nuclear acridine orange staining, and requires caspase activities67. Furthermore, the death of these two larval tissues is foreshadowed by transcription of the rpr and hid cell death genes. Therefore, steroid-regulated destruction of larval tissues occurs by programmed cell death responses. Among the larval tissues undergoing steroid-regulated programmed cell death, the destruction of larval salivary glands is the best characterized. Recent studies have provided insights into the molecular mechanisms by which steroid hormones regulate programmed cell death29,45. Ecdysone and ecdysone receptor complex, a heterodimer of the EcR and USP nuclear receptor proteins, directly activate transcription of a small group of early puff genes including BR-C, E74 and E9368, 69, 70. These early genes activate a large group of late genes, which are thought to direct specific biological responses. Salivary glands of homozygous BR-C, E74 or E93 mutants fail to die, indicating that these genes regulate steroid-triggered programmed cell death29,45. EcR, BR-C and E74 have been shown to regulate rpr and hid transcription45. Furthermore, homozygous E93 mutant salivary glands lack transcription of BR-C, E74, rpr, hid, dronc, ark and crq29. Together, this data indicates that the E93 gene may function near the top of the steroid signaling hierarchy to direct death responses. Similar mechanisms may be involved in other steroid induced cell deaths. In the central nervous system, for example, ecdysone regulates apoptosis, and this death is preceded by expression of EcR, and transcription of rpr and grim71,72.
Radiation triggers DNA damage and leads to activation and stabilization of p53, resulting in an increased level of p53. A high level of p53 induces stress response pathways, including cell cycle arrest and apoptosis61. Drosophila dmp53 exhibits striking similarity to its mammalian counterpart73, 74, 75. While loss-of-function mutations have not been reported, expression of a dominant negative dmp53 transgene greatly reduces radiation-activated apoptosis, indicating that Dmp53 function is required for activation of programmed cell death in the presence of DNA damage induced by radiation. Ectopic expression of Dmp53 is sufficient to induce apoptosis, but is not suppressed by co-expression of p35. This suggests that Dmp53 regulates at least two distinct programmed cell death pathways since radiation-induced cell death can be suppressed by p3573. Previous studies have shown that rpr mRNA is induced by radiation56. The upstream regulatory region of rpr is sufficient to confer radiation responsiveness, and this region contains a radiation-specific enhancer element. This enhancer element contains a sequence that is similar to the consensus DNA-binding site of human p53, and this p53 response element physically interacts with Dmp53, indicating that the rpr gene is a direct transcriptional target of p5375. Further, ark mRNA can also be induced by radiation. However, it is not clear whether ark is also a transcriptional target of p53 or is induced independent of dmp53.
Removal of apoptotic cells
Dying cells appear to be removed by multiple mechanisms based on the morphologies of apoptotic, autophagic, and non-lysosomal cell deaths2,3. During apoptosis, degradation of cellular debris is thought to occur following engulfment of apoptotic cells by macrophages. Within macrophages, fusion of apoptotic corpses and lysosomes results in the formation of phagolysosomes that degrade cellular components76. The recognition of changes in the plasma membrane of dying cells by specific macrophage surface receptors plays an important role in phagocytosis. The exposure of phosphatidylserine (PS) from the inner leaflet to the outer leaflet of the plasma membrane of apoptotic cells is the only characterized signal that is thought to trigger phagocytosis77,78. A specific receptor for PS has recently been reported, and been implicated in phagocytosis of apoptotic cells79. Other macrophage receptors such as the CD36 family of proteins are also involved in the removal of dying cells77. Studies in C. elegans led the discovery that ced-1, -2, -5, -6, -7 and -10 are important for corpse engulfment13. CED-2 encodes a relative of human CrkII, CED-5 encodes a relative of Drosophila Myoblast City and human DOCK180, CED- 6 encodes a candidate adaptor protein with relatives in flies and humans, CED-7 encodes a relative of ABC transporters, and CED-10 encodes a relative of the human GTPase Rac80, 81, 82, 83. While relatives of these genes are present in other species, the function of these genes in the removal of apoptotic cells has not been characterized outside of nematodes.
During Drosophila metamorphosis, PS exposure has been observed in tissues including eyes and the proboscis, where cells undergo developmental regulated programmed cell death84. A fly homolog of the PS receptor has also been reported based on the predicted sequence79. However, no specific mutations are available for functional assessment of the role of either PS exposure or the presumptive PS receptor in phagocytosis of apoptotic cells. The Drosophila relative of mammalian CD36, croquemort ( crq), on the other hand, has been molecularly and genetically characterized85. CRQ is specifically expressed in embryonic hemocytes/macrophages that are capable of removing dying cells. While chromosomal deletions of the crq locus do not impact their ability to engulf bacteria, macrophages in these embryos fail to phagocytose apoptotic corpses, and this defect can be restored by expression of a crq transgene86. In addition, the level of CRQ expression is sensitive to the amount of apoptosis, as embryos that are cell death defective exhibit a very low level of CRQ expression. Interestingly, crq is transcribed immediately prior to the steroid-activated programmed cell death of larval salivary glands, which exhibit morphology of autophagic cell death29,87. This observation, combined with the display of apoptotic hallmarks, the requirement of caspase activities and transcription of the apoptotic genes rpr, hid and ark, support the hypothesis that apoptotic and autophagic cell death may utilize some common components, including execution and degradation of dying cells.
Concluding remarks
Recent studies of Drosophila illustrate that they contain the conserved genetic machinery that is used during apoptosis in animals including nematodes and humans. While substantial details are known about the genes involved in cell execution, far less is known about the activation of cell death, and removal of cells in the context of developing organisms. The similarity between Drosophila and higher organisms in their signaling pathways, combined with the strength of fruit flies as a genetic system, indicates that this is an important model to understand apoptosis. Future gene prospecting and in vivo studies of gene function in flies should provide a framework for studies of cell death in higher organisms including humans.
References
Jacobson MD, Weil M, Raff MC . Programmed Cell Death in Animal Development. Cell 1997; 88:347–54.
Clarke P . Developmental cell death: morphological diversity and multiple mechanisms. Anat Embryol 1990; 181:195–213.
Schweichel J, Merker H . The morphology of various types of cell death in prenatal tissues. Teratology 1973; 7:253–66.
Kerr JF, Wyllie AH, Currie AR .Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 1972; 26:239–57.
Howie S, Harrison D, Wyllie A . Lymphocyte apoptosis-mechanisms and implications in disease. Immunol Rev 1994; 142:141–56.
Ellis RE, Yuan J, Horvitz RH . Mechanisms and functions of cell death. Annu Rev Cell Biol 1991; 7:663–98.
Ellis H, Horvitz H . Genetic control of programmed cell death in the nematode C. elegans. Cell 1986; 44:817–29.
Hengartner MO, Ellis RE, Horvitz HR . Caenorhabditis elegans gene ced-9 protects cells from programmed cell death. Nature 1992; 9:494–99.
Chinnaiyan AM, O'Rourke K, Lane BR, Dixit VM . Interaction of CED-4 with CED-3 and CED- 9: a molecular framework for cell death. Science 1997; 275:1122–6.
James C, Gschmeissner S, Fraser A, Evan GI . CED-4 induces chromatin condensation in Schizosaccharomyces pombe and is inhibited by direct physical association with CED-9. Curr Biol 1997; 7:246–52.
Spector MS, Desnoyers S, Hoeppner DJ, Hengartner MO . Interaction between the C. elegans cell-death regulators CED-9 and CED-4. Nature 1997; 385:653–6.
Wu D, Wallen HD, Inohara N, Nunez G . Interaction and regulation of the Caenorhabditis elegans death protease CED-3 by CED-4 and CED-9. J Biol Chem 1997; 272:21449–54.
Metzstein M, Stanfield G, Horvitz H . Genetics of programmed cell death in C. elegans: past, present and future. Trends Genet 1998; 14:410–6.
Alnemri ES, Livingston DJ, Nicholson DW, Salvesen G, Thornberry NA, Wong WW, et al. Human ICE/CED- 3 protease nomenclature. Cell 1996; 87:171.
Li P, Nijhawan D, Budihardjo I, Srinivasula S, Ahmad M, Alnemri E, et al. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 1997; 91:479–89.
Yuan J, Horvitz H . The Caenorhabditis elegans cell death gene ced-4 encodes a novel protein and is expressed during the period of extensive programmed cell death. Development 1992; 116:309–20.
Zou H, Henzel WJ, Liu X, Lutschg A, Wang X . Apaf-1, ahumanprotein homologous to C. elegans CED-4, participates in cytochrome c-dependent activation of caspase-3. Cell 1997; 90:405–13.
Hengartner MO, Horvitz HR . C. elegans cellsurvival gene ced-9 encodes a functional homolog of the mammalian proto-oncogene bcl-2. Cell 1994; 76:665–76.
Rubin G, et al. Comparative genomics of the eukaryotes. Science 2000; 287:2204–15.
Adams MD, et al. The genome sequence of Drsophila melanogaster. Science 2000; 287:2185–95.
Song Z, Steller H . Death by design: mechanismand control of apoptosis. Trends Cell Biol 1999; 9:M49–52.
Chen P, Rodriguez A, Erskine R, Thach T, Abrams J . Dredd, a novel effector of the apoptosis activators reaper, grim, and hid in Drosophila. Dev Biol 1998; 201:202–16.
Dorstyn L, Read S, Quinn L, Richardson H, Kumar S . DECAY, a novel Drosophila caspase related to mammalian caspase-3 and caspase-7. J Biol Chem 1999; 274:30778–83.
Dorstyn L, Colussi P, Quinn L, Richardson H, Kumar S . DRONC, an ecdysone-inducible Drosophila caspase. Proc Natl Acad Sci USA 1999; 96:4307–12.
Fraser A, Evan G . Identification of a Drosophila melanogaster ICE/CED-3-related protease, drICE. EMBO J 1997; 16:2805–13.
Inohara N, Koseki T, Hu Y, Chen S, Nunez G . CLARP, a death effector domain-containing protein interacts with caspase-8 and regulates apoptosis. Proc Natl Acad Sci USA 1997; 94:10717–22.
Song Z, McCall K, Steller H . DCP-1, a Drosophila cell death protease essential for development. Science 1997; 275:536–40.
Meier P, Silke J, Leevers S, Evan G . The Drosophila caspase DRONC is regulated by DIAP1. EMBO J 2000; 19:598–611.
Lee C-Y, Wendel D, Reid P, Lam G, Thummel C, Baehrecke E . E93 directs steroid-regulated programmed cell death in Drosophila .Molecular Cell 2000; In Press
Green D, Kroemer G . The central executioners of apoptosis: caspases or mitochondria? Trends Cell Biol 1998; 8:267–71.
McCall K, Steller H . Requirement for DCP-1 caspase During Drosophila Oogenesis. Science 1998; 279:230–4.
Nagata S . Apoptotic DNA fragmentation. Exp Cell Res 2000; 256:12–8.
Yokoyama H, Mukae N, Sakahira H, Okawa K, Iwamatsu A, Nagata S . A novel activation mechanism of caspase-activated DNase from Drosophila melanogaster. J Biol Chem 2000; 275:12978–86.
Mukae N, Yokoyama H, Yokokura T, Sakoyama Y, Sakahira H, Nagata S . Identification and developmental expression of inhibitor of caspase-activated DNase (CAD) in Drosophila melanogaster. J Biol Chem 2000; In Press.
Zhou L, Song Z, Tittel J, Steller H . HAC-1, a Drosophila homolog of APAF-1 and CED-4, functions in developmental and radiation-induced apoptosis. MolCell 1999; 4:745–55.
Rodriguez A, Oliver H, Zou H, Chen P, Wang X, Abrams JM . Dark is a Drosophila homologue of Apaf-1/CED-4 and functions in an evolutionarily conserved death pathway. Nat Cell Biol 1999; 1:272–9.
Kanuka H, Sawamoto K, Inohara N, Matsuno K, Okano H, Miura M . Control of the cell death pathway by Dapaf-1, a Drosophila Apaf-1/CED-4-related caspase activator. Mol Cell 1999; 4:757–69.
Li P, Nijhawan D, Budihardjo I, Srinivasula S, Ahmad M, Alnemri E, et al. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 1997; 91:479–89.
Igaki T, Kanuka H, Inohara N, Sawamoto K, Nunez G, Okano H, et al. Drob-1, a Drosophila member ofthe Bcl-2/CED-9 family that promotes cell death. Proc Natl Acad Sci USA 2000; 97:662–7.
Colussi PA, Quinn LM, Huang DCS, Coombe M, Read SH, Richardson H, et al. Debcl, a proapoptotic Bcl-2 homologue, is a component of the Drosophila melanogaster cell death machinery. J Cell Biol 2000; 148:703–14.
Zhang H, Huang Q, Ke N, Matsuyama S, Hammock B, Godzik A, et al. Drosophila pro-apoptotic Bcl-2/Bax homologue reveals evolutionary conservation of cell death mechanisms. J Biol Chem 2000; In Press.
Brachmann C, Jassim O, Wachsmuth B, Cagan R . The drosophila bcl-1 family member dBorg-1 functions in the apoptotic response to UV-irradiation. Curr Biol 2000; 10:547–50.
Jones G, Jones D, Zhou L, Steller H, Chu Y . Deterin, a New inhibitor of apoptosis from Drosophila Melanogaster. J Biol Chem 2000; In Press.
Hay BA, Wassarman DA, Rubin GM . Drosophila homologs of baculovirus inhibitor of apoptosis proteins function to block cell death. Cell 1995; 83:1253–62.
Jiang C, Lamblin A-FJ, Steller H, Thummel CT . A steroid-triggered transcriptional hierarchy controls salivary gland cell death during Drosophila metamorphosis. Mol Cell 2000; 5:445–55.
Wang SL, Hawkins CJ, Yoo SJ, Moller H-AJ, Hay BA . The Drosophila caspase inhibitor DIAP1 is essential for cell survival and is negatively regulated by HID. Cell 1999; 98:453–63.
Hawkins C, Yoo S, Peterson E, Wang S, Vernooy S, Hay B . The Drosophila caspase DRONC is a glutamate/aspartate protease whose activity is regulated by DIAP1, HID and GRIM. J Biol Chem 2000; In Press.
Lisi S, Mazzon I, White K . Diverse domains of THREAD/DIAP1 are required to inhibit apoptosis induced by REAPER and HID in Drosophila. Genetics 2000; 154:669–78.
Goyal L, McCall K, Agapite J, Hartwieg E, Steller H . Induction of apoptosis by Drosophila reaper, hid and grim through inhibition of IAP function. EMBO J 2000; 19:589–97.
White K, Grether ME, Abrams JM, Young L, Farrell K, Steller H . Genetic control of programmed cell death in Drosophila. Science 1994; 264:677–83.
Grether ME, Abrams JM, Agapite J, White K, Steller H . The head involution defective gene of Drosophila melanogaster functions in programmed cell death. Genes and Dev 1995; 9:1694–708.
Chen P, Nordstrom W, Gish B, Abrams JM . grim, a novel cell death gene in Drosophila. Genes and Dev 1996; 10:1773–82.
Haining W, Carboy-Newcomb C, Wei C, Steller H . The proapoptotic function of Drosophila Hid is conserved in mammalian cells. Proc Natl Acad Sci USA 1999; 96:4936–41.
White K, Tahaoglu E, Steller H . Cell killing by the Drosophila gene reaper. Science 1996; 271:805–7.
Zhou L, Schnitzler A, J. A, Schwartz LM, Steller H, Nambu JR . Cooperative functions of the reaper and head involution defective genes in the programmed cell death of Drosophila central nervous system midline cells. Proc Natl Acad Sci USA 1997; 94:5131–6.
Nordstrom W, Chen P, Steller H, Abrams J . Activation of the reaper gene during ectopic cell killing in Drosophila. Dev Biol 1996; 180:213–26.
Song Z, Guan B, Bergman A, Nicholson D, Thornberry N, Peterson E, et al. Biochemical and genetic interactions between Drosophila caspases and the proapoptotic genes rpr, hid, and grim. Mol Cell Biol 2000; 20:2907–14.
Baehrecke EH . Steroid regulation of programmed cell death during Drosophila development. Cell Death and Diff 2000; In Press.
Kurada P, White K . Ras promotes cell survival in Drosophila by downregulating hid expression. Cell 1998; 95:319–29.
Bergmann A, Agapite J, McCall K, Steller H . The Drosophila gene hid is a direct molecular targetof Ras-dependent survival signaling. Cell 1998; 95:331–41.
Steller H . Drosophila p53: meeting the grim reaper. Nat Cell Biol 2000; 2:E100–2.
Riddiford LM . Hormones and Drosophila development. In: Bate M, Martinez Arias A eds. The development of Drosophila melanogaster. Cold Spring Harbor Laboratory Press: Cold Spring Harbor 1993:899–940.
Handler AM . Ecdysteroid titres during pupal and adult development in Drosophila melanogaster. Dev Biol 1982; 93:73–82.
Pak MD, Gilbert LI . Adevelopmental analysis of ecdysteroids during the metamorphosis of Drosophila melanogaster. J Liq Chrom 1987; 10:2591–611.
Robertson CW . The metamorphosis of Drosophila melanogaster, including an accurately timed account of the principal morphological changes. Journal of Morphology 1936; 59:351–99.
Sliter TJ, Gilbert LI . Developmental arrest and ecdysteroid deficiency resulting from mutations at the dre4 locus of Drosophila. Genetics 1992; 130:555–68.
Jiang C, Baehrecke EH, Thummel CT . Steroid regulated programmed cell death during Drosophila metamorphosis. Development 1997; 124:4673–83.
Thomas HE, Stunnenberg HG, Stewart AF . Heterodimerization of the Drosophila ecdysonereceptor with retinoid X receptor and ultraspiracle. Nature 1993; 362:471–5.
Thummel C . Files onsteroids- Drosophila metamorphosis and the mechanisms of steroid hormone action. Trends Genet 1996; 12:306–10.
Yao T-P, Segraves WA, Oro AE, Mc Keown M, Evans RM . Drosophila ultraspiracle modulates ecdysone receptor function via heterodimer formation. Cell 1992; 71:63–72.
Robinow S, Talbot WS, Hogness DS, Truman JW . Programmed cell death in the Drosophila CNS is ecdysone-regulated and coupled with a specific ecdysone receptor isoform. Development 1993; 119:1251–9.
Robinow S, Draizen TA, Truman JW . Genes that induce apoptosis: transcriptional regulation in identified, doomed neurons of the Drosophila CNS. Dev Biol 1997; 190:206–13.
Ollmann M, Young L, Di Como C, Karim F, Belvin M, Robertson S, et al. Drosophila p53 is a structural and functional homolog of the tumor suppressor p53. Cell 2000; 101:91–101.
Jin S, Martinek S, Joo W, Wortman J, Mirkovic N, Sali A, et al. Identification and characterization of a p53 homologue in Drosophila melanogaster. Proc Natl Acad Sci USA 2000; 97:7301–6.
Brodsky M, Nordstrom W, Tsang G, Kwan E, Rubin G, Abrams J . Drosophila p53 binds a damage response element at the reaper locus. Cell 2000; 101:103–13.
Aderem A, Underhill D . Mechanisms of phagocytosis in macrophages. Annu Rev Immunol 1999; 17:593–623.
Savill J . Recognition and phagocytosis of cells undergoing apoptosis. Br Med Bull 1997; 53:491–508.
Franc N, White K, Ezekowitz R . Phagocytosis and development: back to the future. Curr Opin Immunol 1999; 11:47–52.
Fadok V, Bratton D, Rose D, Pearson A, Ezekewitz R, Henson P . Areceptor for phosphatidylserine-specific clearance of apoptotic cells. Nature 2000; 405:85–90.
Reddien PW, Horvitz HR CED-2/CrkII and CED-10/Rac control phagocytosis and cell migration in Caenorhabditis elegans. Nature Cell Biol 2000; 2:131–6.
Liu QA, Hengartner MO . Candidate adaptor protein CED-6 promotes the engulfment of apoptotic cells in C. elegans. Cell 1998; 93:961–72.
Wu YC, Horvitz HR . The C.elegans cell corpse engulfment gene ced-7 encodes a protein similar to ABC transporters. Cell 1998; 93:951–60.
Wu YC, Horvitz HR . C. elegans phagocytosis and cell-migration protein CED-5 is similar to human DOCK180. Nature 1998; 392:501–4.
van den Eijnde SM, Boshart L, Baehrecke EH, DeZeeuw. CI, Reutelingsperger CPM, Vermeij-Keers C Cell surface exposure of phosphatidylserine during apoptosis is phylogenetically conserved. Apoptosis 1998; 3:9–16.
Franc NC, Dimarcq J-L, Lagueux M, Hoffmann J, Ezekowitz AB . Croquemort, a novel DrosophilaM hemocyte/macrophage receptor that recognizes apoptotic cells. Immunity 1996; 4:431–43.
Franc NC, Heitzler P, Ezekowitz AB, White K . Requirement for Croquemort in phagocytosis of apoptotic cells in Drosophila. Science 1999; 284:1991–4.
von Gaudecker B, Schmale E-M . Substrate-histochemical investigations and ultrahistochemical demonstrations of acid phosphatase in larval and prepupal salivary glands of Drosophila melanogasater. Cell Tiss Res 1974; 155:75–89.
Acknowledgements
We thank members of the Baehrecke laboratory for helpful discussions and comments on this manuscript. Work on this subject has been supported by NRICGP/USDA grant 9501913 and NIH grant GM59136.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
LEE, CY., BAEHRECKE, E. Genetic regulation of programmed cell death in Drosophila. Cell Res 10, 193–204 (2000). https://doi.org/10.1038/sj.cr.7290048
Issue Date:
DOI: https://doi.org/10.1038/sj.cr.7290048
Keywords
This article is cited by
-
The whole genome sequence of the Mediterranean fruit fly, Ceratitis capitata (Wiedemann), reveals insights into the biology and adaptive evolution of a highly invasive pest species
Genome Biology (2016)
-
Caspase-independent cell death?
Oncogene (2004)
-
Thyroid hormone regulation of apoptotic tissue remodeling during anuran metamorphosis
Cell Research (2001)