Abstract
Lolium perenne/Lolium perenne/Festuca pratensis triploid hybrids are a potentially important source of material for determining the genetic control of agronomically and scientifically important characters and determining how genetic maps relate to physical maps. The evolutionary relationship between L. perenne (Lp) and F. pratensis (Fp) is, however, unclear. In order to determine the genomic relationships between Lp and Fp, genomic in situ hybridization (GISH) and the Chapman and Kimber mathematical model (1992) were used to analyse metaphase I in meiocytes of an LpLpFp triploid hybrid. Both analyses clearly demonstrated that recombination occurs preferentially between the homologous Lp genomes in the LpLpFp triploid, indicating that the genomes of the two species have diverged. Direct analysis of homologous vs. homoeologous chromosome association, as measured by GISH, was in broad agreement with the mathematical analysis. It is therefore concluded that the Chapman and Kimber model (1992) is a valid means of assessing chromosome pairing in these triploid hybrids. The significance of the data for elucidating the closeness of the relationship of L. perenne and F. pratensis is discussed.
Similar content being viewed by others
Introduction
A knowledge of the similarity of the genomes in different species provides a means by which evolutionary relationships can be assessed (Kihara, 1937). In addition, it also provides an important starting point in alien introgression programmes (Gale & Miller, 1987). Many attempts have been made to introgress genes into crops from related, frequently uncultivated, relatives. Such gene transfer is facilitated by homologous or homoeologous recombination at meiosis. A knowledge of the similarity of the genome(s) of the crop to those of related species is important, as this permits an estimation of the frequency of recombination that will occur in interspecific hybrids. Thus, this information allows predictions to be made of the likelihood of transferring specific target genes from one species to another.
Genome relationships can be studied by meiotic analysis of polyploid hybrids, and the development of mathematical models has led to a more objective, quantitative approach (Alonso & Kimber, 1981; Crane & Sleper, 1989; Chapman & Kimber, 1992; Sybenga, 1996). Although mathematical models have proved to be invaluable for determining genome relationships, they become redundant if it is possible to distinguish individual genomes and chromosomes in a hybrid either morphologically or using cytological techniques such as C-banding, fluorescent in situ hybridization (FISH) or genomic in situ hybridization (GISH) (Phillips & Strickland, 1966; Naranjo et al., 1987; King et al., 1993, 1994; Miller et al., 1994).
Lolium perenne/Lolium perenne/Festuca pratensis (LpLpFp) triploid hybrids are a potentially important source of material for determining the genetic control of agronomically and scientifically important characters and in assessing the relationship of physical and genetic maps (King et al., 1997, 1998). The phylogenetic relationship between L. perenne (Lp) and F. pratensis (Fp) is, however, unclear. Diploid hybrids, although showing a high frequency of homoeologous recombination at meiosis, are nearly completely sterile (Lewis, 1966; Jauhar, 1975). However, LpLpFp triploids, derived by hybridizing synthetic tetraploid L. perenne with diploid F. pratensis, frequently show both male and female fertility (Jauhar, 1975). The ease of hybridization, the high frequency of homoeologous chromosome pairing observed at meiosis in both diploid and triploid hybrids and a preliminary molecular genetic study on the phylogeny of these species (Stammers et al., 1995) indicate that they are closely related. In contrast, the sterility of the diploid hybrid, the ease with which Lp and Fp chromosomes can be distinguished using genomic in situ hybridization (GISH) (King et al., 1997, 1998) and the position and number of rDNA sites in the two species (Thomas et al., 1996, 1997a) indicates that they are distantly related. Thus, the relationship between Lp and Fp remains a paradox at present.
In order to determine the genomic relationships between Lp and Fp, GISH and the Chapman and Kimber model (1992) have been used to analyse metaphase I in meiocytes of the LpLpFp triploid hybrid described by King et al. (1997, 1998). The observations and implications of this work are discussed.
Materials and methods
Triploid LpLpFp hybrids were generated by intercrossing the synthetic autotetraploid Lp (variety Meltra, 2n=4x=28) as the female parent with diploid Fp (Bf 1183, 2n=2x=14) as the pollen parent. The pollen mother cells of a single LpLpFp triploid hybrid (2n=3x=21) resulting from this cross (which has been used to generate a BC1 population for introgression mapping; King et al., 1997; 1998) was analysed. Inflorescences were fixed in 3:1 ethanol-acetic acid solution.
In this work, the term paired arm refers to any chiasmate association between two arms of a pair of chromosomes. The Chapman and Kimber model (1992) analyses the frequency of paired arms rather than the chiasma frequency in triploid hybrids. Thus, in order that the GISH analysis could be compared directly with the Chapman and Kimber model, the frequency of paired arms rather than the total number of chiasmata was recorded.
Total genomic DNA was extracted from young actively growing leaves of Fp and Lp as described by Dellaporta et al. (1983). Fp DNA, which was used exclusively as the probe, was sheared to approximately 5 kb using a sonicating water bath and then labelled using either digoxigenin-11-dUTP (Boehringer Mannheim), rhodamine-4-dUTP (Amersham) or Texas red-5-dCTP (Dupont). Blocking DNA was prepared by autoclaving the DNA of Lp for 2 min, which produced fragments of approximately 200 bp in length. The in situ hybridization protocol used followed that described by Thomas et al. (1994).
Slides were analysed using a Leica DM/RB epifluorescence microscope with filter blocks for DAPI, fluorescein and rhodamine. Images were captured with a monochrome CCD camera and false colours applied as described by Thomas et al. (1996).
Results and discussion
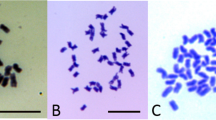
Metaphase I was analysed in 72 pollen mother cells of the LpLpFp hybrid. Using the GISH technique, it was possible to distinguish the Lp and Fp chromosomes in each pollen mother cell observed (Figure 1). Thus, it was possible to record the presence or absence and position of the Lp and Fp chromosomes in each meiotic configuration (Table 1).
Metaphase I of the LpLpFp triploid with the Fp chromosomes in green after GISH using Fp DNA as a probe. (a) One ‘frying pan’ trivalent with the Fp chromosome as the handle (arrow), six Lp/Lp bivalents and six Fp univalents; (b) one frying pan trivalent with the Fp chromosome as the handle (arrows), one Lp/Fp bivalent (arrowhead), five Lp/Lp bivalents, one Lp univalent and five Fp univalents; (c) four frying pan trivalents (arrows) three with the Fp chromosome in the ring and one with the Fp chromosome in the handle, plus one V-shaped chain trivalent (arrowhead), two Lp/Lp bivalents and two Fp univalents; (d) five frying pan trivalents (arrows), four with the Fp chromosome in the ring and one with the Fp chromosome as handle, one Lp bivalent, two Lp univalents and two Fp univalents. Bar represents 10 μm.
Each of the 14 Lp and seven Fp chromosomes can be placed in one of seven homologous/homoeologous groups, such that each group contains two homologous Lp chromosomes and one homoeologous Fp chromosome. In the absence of preferential pairing, the three chromosomes in each group will pair with each other at random. Thus, each Lp chromosome can pair homologously with the other Lp chromosome and homoeologously with the Fp chromosome in the same group at equal frequency, giving a 1:1 ratio of Lp/Fp to Lp/Lp pairing. The Fp chromosome can only pair homoeologously. Consequently, in the absence of preferential pairing, the ratio of homoeologous recombination to homologous recombination is 2:1. Therefore, in the absence of trivalent formation, Lp/Fp bivalents should outnumber Lp/Lp bivalents by 2:1. However, in the 72 pollen mother cells analysed, only 28 Lp/Fp bivalents were observed compared with 235 Lp/Lp bivalents (Table 1). Clearly, this does not fit a 2 Lp/Fp:1Lp/Lp ratio (χ21=368.93, P<0.001). Again, with random pairing, there should be twice as many Lp univalents as Fp univalents; but significantly more Fp univalents were observed than Lp univalents (χ21=142.46, P<0.001) (Table 1).
Ninety-nine chain trivalents were observed (Table 1). There are two opportunities for the Fp chromosome to be at the end of a linear trivalent and only one at the middle position. Thus, if there is no preferential pairing, the ratio at which the Fp chromosome should occur at the end of a trivalent compared with the middle of a trivalent is 2:1. Thus, in the 99 chain trivalents observed, the Fp chromosome should be at the end of the configuration on approximately 66 occasions and in the middle on 33 occasions. However, the Fp chromosome was at the middle position in only 14 trivalents and was at the end position in 85, which was significantly different from the expected 2:1 ratio (χ21=15.56, P<0.001). The two chromosomes at the ends of the configuration each only need to pair once to the chromosome in the centre of the configuration. In contrast, the chromosome at the centre of the configuration has to pair twice. The Fp chromosome therefore occupied a position that required the least homoeologous pairing.
Analysis of the 137 ‘frying pan’ trivalents revealed a similar pattern (Table 1). In the absence of preferential pairing, the ratio at which the Fp chromosome should appear in the ring relative to the handle is 2:1. However, the Fp chromosome was located within the ring significantly less frequently than expected (χ21=21.93, P<0.001). The Fp chromosome is not, therefore, randomly located within the configuration. Chromosomes within the ring have to form chiasmata either twice with one chromosome or three times with two chromosomes, whereas the chromosome forming the handle only needs to form one chiasma with one chromosome. As in the chain trivalents, the Fp chromosome is preferentially located to form the least number of chiasmata with Lp chromosomes.
In the absence of preferential pairing in the LpLpFp hybrid, it is expected that the ratio of homoeologous to homologous paired arms would be 2:1. Thus, out of the 1098 paired arms observed, approximately 732 would be expected to be between homoeologous Lp/Fp chromosomes and 366 between homologous Lp chromosomes. However, only 364 of the paired arms were between homoeologous chromosomes. Unsurprisingly, a chi-squared analysis rejected a 2Lp/Fp:1Lp/Lp ratio (χ21=553.5, P<0.001).
In summary, the GISH analysis overwhelmingly demonstrates that preferential pairing occurs between the Lp homologous chromosomes in the LpLpFp triploid. To ascertain if each of the individual Fp chromosomes was pairing at equal frequencies with its Lp homoeologues, a binomial analysis, as described by Hall (1955), was performed. If each group of three chromosomes has the same propensity to form trivalents, then the number of trivalents observed from cell to cell should fit a binomial distribution. This was found to be the case (χ24=1.50), demonstrating that each of the Fp chromosomes paired homoeologously with its Lp partners at equal frequency.
Only two triradial trivalents were observed in this study. However, the low frequency of this type of trivalent was not unexpected, because this type of configuration is very rare in Lolium (Thomas et al., 1997b). In addition, two Fp/Fp associations were observed. Such associations, although rare, have been documented in other hybrids including wheat ×rye (Hutchinson et al., 1983; King et al., 1994), wheat×Thinopyrum bessarabicum (King et al., 1994) and L. multiflorum×F. drymeja (Thomas & Morgan 1990).
Using GISH, it is possible to observe the frequency of homologous and homoeologous recombination and, hence, the level of preferential pairing in the LpLpFp hybrid. In other hybrids, in which it has not previously been possible to identify the chromosomes from different genomes, the extent to which homoeologous and homologous pairing occurs has been estimated using mathematical models. One such model (Alonso & Kimber, 1981; Chapman & Kimber, 1992) was applied to the present data in order to determine the accuracy of its predictions. This model was chosen as it has been found to show results similar to other mathematical models.
The model states that, in a triploid hybrid, genomes 1 and 2 are designated the more closely related pair (not necessarily homologous) with a relative affinity of x. Both are equally and more distantly related to genome 3 with a relative affinity of y so that x≲y. The relative affinity is expressed as the proportions of the three different arm pairings that may be formed so that the ratio among (1–2) (1–3) and (2–3) arm pairs is x : y : y (Chapman & Kimber, 1992). The values of x and y will therefore be related by the equation x+y=1. This definition sets maximum and minimum values on x. If the relative affinity of the two most closely related genomes is so strong that no chiasmata occur between the two most closely related genomes (1 and 2) and the most distantly related genome (3), then x will be 1 and y will be 0. If the relative affinity of genomes 1 and 2 equals that between (1 and 3) and (2 and 3) then x will equal y and have a numerical value of 0.5. Generally, a hybrid with a value of x above 0.8 is considered to be a 2:1 type, in which two genomes have an equal affinity with each other and an equal and lower affinity with a third genome. A hybrid with a value of x less than 0.8 is considered to be a 3:0 type, in which all the genomes have an equal affinity (Alonso & Kimber, 1981).
Chapman & Kimber (1992) developed the equation shown below to determine the relationship (the relative affinity) between the three genomes in a triploid

where x=the relative affinity and

where t is the number of trivalents per cell/b, g is the number of ring bivalents per cell/b and u is the number of univalents per cell/b. In all cases, b is the basic chromosome number, and c is the mean arm-pairing frequency.
The value of x, the relative affinity, calculated from meiotic data from the LpLpFp triploid is equal to 0.8405, indicating that the hybrid is a 2:1 type, although only marginally so. This observation is in agreement with the direct analysis of chromosome pairing made using GISH, but the degree of preferential pairing is probably greater than the value of x indicates. Nevertheless, the model provides a valid method of assessing chromosome pairing in triploid hybrids in which it is not possible to distinguish the chromosomes of individual genomes.
The relative affinity can also be calculated by observation of the number of ring bivalents relative to trivalents. Alonso & Kimber (1981) observed that curves showing the relationship of x to the number of ring bivalents and x to the number of trivalents always intersect at an x-value of 0.8. Thus, if the number of trivalents is greater than the number of ring bivalents (x is less than 0.8), then the hybrid can be considered to be a 3:0 type. More trivalents (236) were observed than ring bivalents (226) even though the GISH analysis overwhelmingly demonstrated the hybrid to be a 2:1 type. Thus, it was not possible to predict whether the LpLpFp hybrid was a 3:0 or 2:1 type by comparing the number of ring bivalents and trivalents. We therefore conclude that a comparison of the number of ring bivalents and trivalents is not a robust method of testing the affinity of the genomes.
The Chapman and Kimber model assumes that pairing of one chromosome arm with any other precludes any involvement of a third. Thus, frying pan trivalents, in which one arm has exchanged pairing partners somewhere along its length and therefore paired with two other chromosomes, must be rare. However, in the LpLpFp hybrid, there were significantly more frying pan trivalents (137) than linear or V-shaped trivalents (99) (χ21=5.8, P<0.02). Chapman & Kimber (1992) warn that, should a data set include a high frequency of complex figures (frying pan, ‘bird cage’ and triradial trivalents), it is generally unanalysable using their model. The high frequency of frying pan trivalents in our data shows that there is more than one pairing initiation site per chromosome arm. The opportunity for three chromosomes to form trivalents is therefore greater than if there is only one pairing initiation site per arm. The present data demonstrate that, with multiple pairing initiation sites, it is possible to have a high trivalent frequency (exceeding the number of ring bivalents) even with preferential pairing.
The results of the meiotic analysis described (GISH and the Chapman and Kimber model) clearly demonstrate that the Fp genome is distinct from the Lp genome. Analyses of chromosome configurations at metaphase I have long been considered valid in studying the phylogenetic relationships of the parental chromosomes in interspecific hybrids. The fundamental assumption of such analyses is that homologous or closely related chromosomes will be held together by chiasmata at metaphase I. The frequency of such associations is taken as a measure of the relatedness of the species.
Kimber & Yen (1990) have sought to quantify the relatedness of diploid species based on chromosome associations at metaphase I in autoallotriploids. However, differences in other factors such as DNA amounts can have profound effects on chromosome pairing at metaphase I (Morgan et al., 1986). Lolium temulentum has 50% more DNA than L. multiflorum (Hutchinson et al., 1983). Thomas (1995) studied the inverse triploid hybrids of L. temulentum (T)×L. multiflorum (M). The combinations studied were: one large genome+two small genomes (TMM) and two large genomes+one small genome (TTM). When these data sets were analysed by the Alonso & Kimber model (1981), the model identified TTM as 3:0 but classified TMM as 2:1. The discrepancies between these inverse types of autoallotriploids have serious implications for the use of pairing data in determining phylogenetic relationships. However, in the present situation, the fact that the Lp genome can be so easily distinguished from the Fp genome by GISH demonstrates that the repetitive fraction of DNA of the two species has undergone considerable divergence.
References
Alonso, L. C. and Kimber, G. (1981). The analysis of meiosis in hybrids. II. Triploid hybrids. Can J Genet Cytol, 23: 221–234.
Chapman, C. G. D. and Kimber, G. (1992). Developments in the meiotic analysis of hybrids. I. Review of theory and optimization in triploids. Heredity, 68: 97–103.
Crane, C. F. and Sleper, D. A. (1989). A model of meiotic chromosome association in triploids. Genome, 32: 82–98.
Dellaporta, S. L., Wood, J. and Hicks, J. B. (1983). A plant DNA minipreparation: version 11. Plant Mol Biol Rep, 1: 19–21.
Gale, M. D. and Miller, T. E. (1987). The introduction of alien genetic variation into wheat. In: Lupton, F. G. H. (ed.) Wheat Breeding. Its Scientific Basis, pp. 173–210. Chapman & Hall, London.
Hall, M. H. (1955). Binomial analysis of chromosome behaviour in hybrid plants. Pennsylvania Acad Sci, 29: 121–126.
Hutchinson, J., Miller, T. E. and Reader, S. M. (1983). C-banding at meiosis as a means of assessing chromosome affinities in the Tritiaceae. Can J Genet Cytol, 25: 319–323.
Jauhar, P. P. (1975). Chromosome relationships between Lolium and Festuca (Gramineae). Chromosoma, 52: 103–121.
Kihara, H. (1937). Genomanalyse bei Triticum und Aegilops VII. Kurz åbersicht ûber die Ergenbnisse des Jahre 1934–36. Mem Coll Agric Kyoto Univ, 41.
Kimber, G. and Yen, Y. (1990). Genomic analysis of diploid plants. Proc Natl Acad Sci USA, 87: 3205–3209.
King, I. P., Purdie, K. A., Orford, S. E., Reader, S. M. and Miller, T. E. (1993). Detection of homoeologous chiasma formation in Triticum durum×Thinopyrum bessarabicum hybrids using genomic in situ hybridization. Heredity, 71: 369–372.
King, I. P., Reader, S. M., Purdie, K. A., Orford, S. E. and Miller, T. E. (1994). A study of the effect of a homoeologous promoter on chromosome pairing in wheat/rye hybrids using genomic in situ hybridization. Heredity, 72: 318–321.
King, I. P., Morgan, W. G., Harper, J. A., Meredith, M. R., Jones, R. N., Armstead, I. P. et al. (1997). Introgression mapping: a tool to integrate physical and genetic maps. Plant and Animal Genome V, San Diego, CA.
King, I. P., Morgan, W. G., Armstead, I. P., Harper, J. A., Hayward, M. D., Bollard, A. et al. (1998). Introgression mapping in the grasses. I. Introgression of Festuca pratensis chromosomes and chromosome segments into Lolium perenne. Heredity, 81: 462–467.
Lewis, E. J. (1966). The production and manipulation of new breeding material in Lolium-Festuca. In: Hill, A. G. G. (ed.) Proc. X Intern. Grassland Congr. Helsinki, pp. 688–693. Valtioneuvoston Kirjapaino, Helsinki.
Miller, T. E., Reader, S. M., Purdie, K. A. and King, I. P. (1994). Determination of the frequency of wheat-rye chromosome pairing in wheat×rye hybrids with and without chromosome 5B. Theor Appl Genet, 89: 255–258.
Morgan, W. G., Thomas, H., Evans, M. and Borrill, M. (1986). Cytogenetic studies of interspecific hybrids between diploid species of Festuca. Can J Genet Cytol, 28: 921–925.
Naranjo, T., Roca, A., Goicoecha, P. G. and Giraldez, R. (1987). Arm homoeology of wheat and rye chromosomes. Genome, 29: 873–882.
Phillips, L. L. and Strickland, M. A. (1966). The cytology of a hybrid between Gossypium hirsutum and G. longicalyx. Can J Genet Cytol, 8: 91–95.
Stammers, M., Harris, J., Evans, G. M., Hayward, M. D. and Forster, J. W. (1995). Use of random PCR (RAPD) technology to analyse phylogenetic relationships in the Lolium/Festuca complex. Heredity, 74: 19–27.
Sybenga, J. (1996). Chromosome pairing affinity and quadrivalent formation in polyploids: do segmental allopolyploids exist. Genome, 39: 1176–1184.
Thomas, H. M. (1995). Meiosis of triploid Lolium II. Discrepancies between the analyses of chromosome configurations at metaphase I in inverse autoallotriploid combinations. Heredity, 75: 446–452.
Thomas, H. M. and Morgan, W. G. (1990). Analysis of synaptonemal complexes and chromosome pairing at metaphase I in the diploid intergeneric hybrid Lolium multiflorum×Festuca drymeja. Genome, 33: 465–471.
Thomas, H. M., Morgan, W. G., Meredith, M. R., Humphreys, M. W., Thomas, H. and Leggett, J. M. (1994). Identification of parental and recombined chromosomes in hybrid derivatives of Lolium multiflorum×Festuca pratensis by genomic in situ hybridization. Theor Appl Genet, 88: 909–913.
Thomas, H. M., Harper, J. A., Meredith, M. R., Morgan, W. G., Thomas, I. D., Timms, E. and King, I. P. (1996). Comparison of ribosomal DNA sites in Lolium species by fluorescence in situ hybridization. Chrom Res, 4: 486–490.
Thomas, H. M., Harper, J. A., Meredith, M. R., Morgan, W. G. and King, I. P. (1997a). Physical mapping of ribosomal DNA sites in Festuca arundinacea and related species by in situ hybridization. Genome, 40: 406–410.
Thomas, H. M., Morgan, W. G., Harper, J. A., Meredith, M. R. and King, I. P. (1997b). Meiosis in triploid Lolium IV. Distribution of chiasmata in autotriploids. Cytologia, 62: 383–387.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
King, I., Morgan, W., Harper, J. et al. Introgression mapping in the grasses. II. Meiotic analysis of the Lolium perenne/Festuca pratensis triploid hybrid. Heredity 82, 107–112 (1999). https://doi.org/10.1038/sj.hdy.6884680
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.hdy.6884680
Keywords
This article is cited by
-
Production of pollenless triploid lily hybrids from Lilium pumilum DC. × ‘Brunello’
Euphytica (2018)
-
Exploitation of interspecific diversity for monocot crop improvement
Heredity (2013)
-
Variability of ribosomal DNA sites inFestuca pratensis, Lolium perenne, and their intergeneric hybrids, revealed by FISH and GISH
Journal of Applied Genetics (2010)
-
Chromosome pairing in allotetraploid hybrids of Festuca pratensis × Lolium perenne revealed by genomic in situ hybridization (GISH)
Chromosome Research (2008)
-
Introgression mapping in the grasses
Chromosome Research (2007)