Abstract
Purpose
Although visual acuity is the most frequently used primary outcome measure in clinical trials of treatments for choroidal neovascularisation (CNV) due to age-related macular degeneration (AMD), contrast sensitivity may provide valuable additional information. This paper reviews the evidence for using contrast sensitivity as a measure of visual function and as an outcome measure in clinical trials in patients with subfoveal CNV due to AMD.
Methods
Medline database searches were performed to retrieve relevant articles on contrast sensitivity. In addition, articles were included from the authors' knowledge of the literature and from the reference lists of retrieved articles.
Results
The published literature demonstrates that contrast sensitivity is an important measure of visual function in patients with subfoveal CNV due to AMD. Most clinical trials of treatments for CNV due to AMD have reported visual acuity as the primary outcome. However, there is evidence that treatment (such as verteporfin therapy) may also provide additional benefits in terms of contrast sensitivity. These benefits may not be completely characterised by measurement of visual acuity alone.
Conclusions
The inclusion of contrast sensitivity as an outcome measure in studies of patients with CNV due to AMD may provide a more complete understanding of the effects of treatment on visual function and the likely benefits for patients.
Similar content being viewed by others
Introduction
Several measures are available to characterise visual function, of which visual acuity is the most widely used. Although visual acuity is undoubtedly a useful measure of visual function, other vision tests may provide valuable additional information.1, 2, 3, 4, 5 In particular, visual acuity is not a good predictor of ability to discriminate between visual targets or performance of tasks requiring distance judgement, night driving and mobility; these aspects of visual function may be better correlated with contrast sensitivity. Contrast sensitivity provides a measure of ability to see low-contrast patterns and it has been suggested that it can provide more information on visual function than visual acuity.6
Recent data from the Blue Mountains Eye Study indicated that age-related macular degeneration (AMD) is the most frequent cause of vision loss in people over the age of 50 years.7 In some cases, measurements of contrast sensitivity might detect vision loss due to AMD before any effect on visual acuity has become apparent.8
The majority of patients with severe vision loss (defined as a loss of 6 or more lines of visual acuity) due to AMD have the neovascular form of the condition, which is characterised by choroidal neovascularisation (CNV).9, 10 Vision loss is more likely to be severe in patients with lesions that lie under the foveal avascular zone (subfoveal lesions) than in those that lie outside the fovea. New treatment modalities, such as photodynamic therapy with verteporfin (Visudyne®, Novartis Pharma AG, Basel, Switzerland), have increased the range of treatments available for patients with subfoveal CNV due to AMD. The benefits of these treatments have been assessed primarily through measurements of visual acuity. Reports of clinical trials of treatments for neovascular AMD often provide little or no information on contrast sensitivity outcomes. However, contrast sensitivity findings from the Treatment of AMD with Photodynamic therapy (TAP) Investigation in patients treated with verteporfin therapy have been reported in detail.11 To set these results in context with the available data from other trials and gain a better understanding of the impact of vision loss and the benefits of treatment, this article reviews the evidence for using contrast sensitivity as a measure of visual function and as an outcome measure in clinical trials in patients with subfoveal CNV due to AMD.
Original articles included in this review were retrieved from Medline during March 2004 using two searches: (1) ‘contrast sensitivity or contrast threshold’ and ‘AMD or age-related macul*’ and ‘CNV or neovascularisation’; and (2) ‘contrast sensitivity or contrast threshold’ and ‘reading or driving or mobility or orientation or activities’. The retrieved articles (100 from search 1 and 1280 from search 2) were assessed and included if relevant information was reported. In addition, articles were included from the reference lists of retrieved articles and from the authors' knowledge of the literature.
Measurement of contrast sensitivity
Until the 1980s, the only methods available to determine contrast sensitivity involved the use of sinusoidal gratings, usually generated by a computer and displayed on a cathode ray tube.12 Although this apparatus is commercially available and can provide accurate measures of contrast sensitivity, the equipment is more suited to a research laboratory than for routine clinical use or for screening.
The development and availability of the Pelli–Robson chart provided a means of measuring contrast sensitivity that could be applied in clinical practice as easily as the visual acuity letter chart.12 The Pelli–Robson chart has letters of equal size arranged in groups of three, with each segment decreasing in contrast by a factor of 1/√2 (a logarithmic step of 0.15). The contrast threshold corresponds to the lowest contrast at which two of the three letters in a group can be correctly read. The reliability of the test can be increased if scored by letter rather than by segment.13 Each correctly read letter reduces contrast threshold by 0.05 log units. (To score letter-by-letter, multiply [number of letters correctly read minus 3] by 0.05. Three letters are subtracted from the total letters read, because the log contrast sensitivity is equal to zero when the observer reads the first segment correctly. If fewer than three letters are read correctly, the log contrast sensitivity is set to zero.) Values recorded using the Pelli–Robson chart can be reported in terms of letters, as the log of contrast sensitivity, or as percentage contrast. On currently available Pelli–Robson charts the log contrast of the letters range from 0 (or 100%) to 2.25 (or 0.6%). The Pelli–Robson chart has been shown to be a reliable and sensitive measure of contrast sensitivity, and compares favourably with other methods.3
Greeves et al14 evaluated a set of Bailley–Lovie log minimum angle of resolution (logMAR) charts used to measure the middle and high spatial frequencies of the contrast sensitivity functions in patients with AMD. They concluded that three measurements are needed to characterise foveal visual capability in patients with AMD: (1) distance logMAR visual acuity to measure high spatial frequency resolution; (2) visual acuity with letter charts of 20 dB contrast to measure middle spatial frequency resolution; and (3) contrast sensitivity.
Contrast sensitivity and visual acuity
There have been numerous studies of the relationship between contrast sensitivity and visual acuity. For example, a study of patients enrolled in a trial of radiation therapy for neovascular AMD showed that contrast sensitivity and visual acuity measurements did not always reveal the same rates of progression of vision loss, although there was a moderate correlation between the two measures.15 Most studies have shown a moderate correlation between the two measures (correlation coefficient ⩾0.5).4, 16 Nevertheless, visual acuity and contrast sensitivity are independently associated with difficulties in performing everyday activities.4 It should also be noted that positive correlations between visual acuity and contrast sensitivity do not demonstrate that the two measures are interchangeable.17 It has, however, been reported that a 6-letter loss of contrast sensitivity has a similar impact on self-reported visual disability as a 15-letter loss of visual acuity.5
The characteristics of choroidal neovascular lesions in patients with neovascular AMD have an important impact on visual function. It has been shown that the composition and location of the lesion is related to the nature and degree of vision loss, with the strongest correlation being between overall lesion size and contrast sensitivity (r=0.52, P<0.001).18 The proportion of the lesion that was classic CNV was also strongly correlated with contrast sensitivity, especially if the study eye was the better-seeing of the two eyes.
Activities of daily living and quality of life
Vision loss can affect many activities of daily life, several of which depend on both contrast sensitivity and visual acuity. For example, in the Blue Mountains Eye Study, both visual acuity and contrast sensitivity were associated with self-reported measures of visual disability.19 People with decreased visual acuity and contrast sensitivity also have greater difficulty with high-risk driving situations than patients with good vision, even after adjusting for age, gender, mileage and cognitive impairment.20 In a study of young, middle-aged and older adult drivers, a combination of motion sensitivity, field of view, contrast sensitivity and dynamic visual acuity predicted 50% of variability in driving test scores.21
Some tasks, however, appear to be more closely related to contrast sensitivity than to visual acuity. Reduced visual acuity appears to be associated with difficulty in tasks involving good resolution and adaptation to changing light levels, whereas contrast sensitivity is more closely associated with tasks requiring distance judgement, night driving and mobility.4
A variety of everyday tasks can be affected by vision loss. For example, the time taken to read the list of ingredients on a can of food, read instructions on a medicine container and find a telephone number in a directory was significantly greater in people with poorer visual acuity, contrast sensitivity and visual field.22 In this study, visual acuity was the most important predictor for tasks requiring fine resolution (such as reading small print). Other tasks may be more closely related to other measures of visual function. For example, in another study, elderly people with good acuity had decreasing reading rates with increasing age, and the deterioration was found to be related to worsening low-contrast vision, motor ability and attentional field integrity (a measure of ability to detect peripheral flashes of light while fixating on a central target).23 Alexander et al24 showed that, when considered separately, visual acuity and contrast sensitivity were both related to performance of visual tasks. When a multivariate analysis was used, visual acuity and contrast sensitivity were independently associated with reading and telling the time. In addition, in patients with the same contrast sensitivity, visual acuity was unrelated to the ability to identify colours, products and faces. Results from another study by Leat et al suggested that contrast sensitivity may be a better predictor of reading performance with low vision aids than visual acuity,25 although this conclusion is contradicted by other investigations.26, 27
Contrast sensitivity is closely correlated with ability to detect and discriminate between visual targets. For example, one study showed that elderly people require greater contrast to perceive faces than younger people even if both groups have good acuity, and the authors concluded that their measures of face perception represented different visual abilities from those involved in visual acuity.28 Although the reasons for this difference are unclear, further investigation has supported the finding: the best predictors of thresholds for recognition of real-world targets (faces, road signs and objects) were age and contrast sensitivity.29 Including visual acuity as a predictor did not improve the model, and the authors concluded that contrast sensitivity might be more useful for identifying difficulties with everyday visual activities that involve identification and discrimination of objects. Consistent with these findings, a study of patients with AMD showed that contrast sensitivity was the best predictor of computer task accuracy.30
Several studies have found that mobility and orientation are correlated with contrast sensitivity. In a study of patients with AMD, low-contrast sensitivity and visual field extent were the most important predictors of mobility performance on a laboratory obstacle course and two real-world courses (one indoor and one outdoor).31 This finding is supported by a study of walking speed on an obstacle course, which showed that visual field and contrast sensitivity predicted 29–35% of variation in mobility in people with AMD.32 Similarly, a study of partially sighted patients (ambulatory patients with at least light perception recruited from a vision rehabilitation centre) found that contrast sensitivity and visual field had the greatest effect on orientation-mobility, but that visual acuity had a negligible effect.33
Loss of spatial awareness and poor mobility or orientation can increase the risk of accidents such as falls. In the Beaver Dam Eye Study, people over 60 years of age were more likely to have had a fall within the past year if they had contrast sensitivity of 1.50 or worse than if they had a value of 1.55 or better.34 Similarly, people with best-corrected visual acuity of 20/20 or better were less likely to have had a fall than those with a visual acuity of 20/25 or worse. The increased likelihood of falls translated into higher incidences of hip fractures in people with worse visual acuity or contrast sensitivity. Similar findings were reported in the Blue Mountains Eye Study: poor visual acuity, contrast sensitivity and visual field were significant risk factors for reporting two or more falls in the last 12 months, and the risk of hip fractures was significantly related to all measures of visual function.35
The relative importance of visual acuity and contrast sensitivity as predictors of hip-fracture risk is unclear. In one study, poor contrast sensitivity and depth perception were associated with a greater risk of hip fracture, but visual acuity was not.36 Conversely, the Epidemiology of Osteoporosis (EPIDOS) study found that visual acuity was associated with hip fracture risk, but depth perception and contrast sensitivity were not.37
Several studies have shown that vision loss results in lower quality of life scores on a variety of scales. For example, measurements of utility values have shown that patients with a visual acuity of 20/40 or worse in the better-seeing eye would trade between 19 and 60% of their remaining lifetime in return for perfect vision in each eye.38, 39, 40 Similarly, Carta et al41 showed that near visual acuity and contrast sensitivity were strongly associated with quality of life outcomes on a quality of life/visual function questionnaire in 120 patients with cataract, glaucoma, AMD, branch retinal vein occlusion, presbyopia or minor refractive defects. The relationship between contrast sensitivity loss and such measures of quality of life is unclear. Nevertheless, the demonstrable impact of a loss of contrast sensitivity on activities of daily living indicates that it would be expected to have a marked effect on quality of life. Further research is needed to explore the relationship between different measures of visual function and quality of life scores.
Patients with poor visual function are also more likely to need to be placed in a nursing home. In the Beaver Dam Eye Study, analyses using a multivariate model (adjusted for confounding factors) showed that people in the lowest category of each measure of visual function had an odds ratio for nursing home placement of 4.23 for best-corrected visual acuity and 2.40 for contrast sensitivity, compared with those in highest category.42
Impact of treatment on contrast sensitivity: evidence from clinical trials
The strong associations between contrast sensitivity and functional abilities provide a rationale for including contrast sensitivity measurements in clinical trials. Although visual acuity is the most frequently cited primary outcome measure in clinical trials of treatments for CNV secondary to AMD, several studies have included contrast sensitivity as a secondary outcome. Considering both visual acuity and contrast sensitivity when assessing the outcomes of clinical trials may provide a more complete picture of the effects of treatment on vision than either measure alone.
At present, two treatments have been proven effective in CNV due to AMD in randomised, controlled clinical trials and are recommended as the standard of care:43, 44 laser photocoagulation and photodynamic therapy with verteporfin, also known as verteporfin therapy.
Laser photocoagulation
In the Macular Photocoagulation Study (MPS), 206 eyes with recurrent subfoveal CNV were assigned to laser photocoagulation or no treatment.45 Patients were required to have at least some classic CNV: 85% of patients had classic CNV with no other areas of presumed CNV (with or without blood or elevated blocked fluorescence). Patients treated with laser photocoagulation lost a mean of 2.7 lines of visual acuity at 24 months, compared with 3.4 lines in the untreated eyes (P=0.46). For contrast threshold, however, there was a significant difference between the two groups: laser-treated eyes had a median contrast threshold of 14% at month 24, compared with 20% for untreated eyes (P=0.01). The MPS group also investigated vision outcomes in 373 eyes with new subfoveal CNV due to AMD, 78% of which had classic CNV with no occult CNV (with or without blood or elevated blocked fluorescence).46 In these eyes, visual acuity deteriorated through month 24 in both the laser-treated and untreated group (but to a lesser extent in the treated eyes) and treated eyes maintained better contrast thresholds than untreated eyes. Laser-treated eyes lost 3.0 lines of visual acuity and had a median contrast threshold of 14% (equivalent to 20 letters on currently available charts) at month 24, compared with a loss of 4.4 lines of visual acuity (P<0.001) and a median contrast threshold of 28% (14 letters) (P<0.001) for untreated eyes.
For eyes with extrafoveal or juxtafoveal CNV secondary to AMD, treated eyes were found to lose less visual acuity than untreated eyes.47, 48 Contrast sensitivity results were not reported for these eyes.
Verteporfin therapy
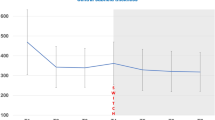
Verteporfin therapy is recommended for patients with subfoveal predominantly classic CNV (lesions composed of ⩾50% classic CNV) due to AMD based on the 1- and 2-year visual acuity results from the TAP Investigation.49, 50 Verteporfin-treated patients with predominantly classic CNV were found to be less likely to lose at least 15 letters of visual acuity (the primary outcome measure) at the month 12 examination than patients who received placebo (33 vs 61%, respectively, P<0.001). This difference was maintained through 24 months (Figure 1a).50 Although verteporfin-treated patients with predominantly classic CNV had significant visual acuity benefits in the TAP Investigation, there were no statistically significant differences in visual acuity outcomes between the treated and untreated patients with minimally classic CNV (lesions composed of <50% classic CNV but >0%) (Figure 1b).50 However, a retrospective exploratory analysis suggested that smaller minimally classic lesions (no larger than 4 MPS DA) may have treatment benefit.51
Percentage of patients with a loss of at least 15 letters of visual acuity (0.3 logMAR) or at least 6 letters (0.3 log units) of contrast sensitivity at the month 24 examination in the TAP Investigation for patients with (a) predominantly classic CNV and (b) minimally classic CNV at baseline. P-values are for Pearson χ2 tests.
Contrast sensitivity was a secondary outcome measure in the TAP Investigation. In the total study population (which included 242 patients with subfoveal predominantly classic CNV, 306 with minimally classic CNV, and 61 with no classic CNV), mean contrast sensitivity remained stable at the month 24 examination in the verteporfin-treated patients (mean loss of 1.3 letters), whereas placebo recipients lost a mean of 5.2 letters.50 Additional analyses were conducted to explore the contrast sensitivity outcomes in the total study population and the subgroups with predominantly classic or minimally classic CNV.11 These analyses showed that the contrast sensitivity results in patients with predominantly classic CNV were consistent with the visual acuity outcomes (Figure 1a). In patients with minimally classic CNV, however, verteporfin-treated patients were less likely to lose 6 or more letters of contrast sensitivity than placebo recipients, despite there being no significant difference in terms of visual acuity (Figure 1b). The reasons for the difference between the visual acuity and contrast sensitivity findings in the eyes with minimally classic CNV are unclear. It is possible that contrast sensitivity provides an earlier indication of vision loss than visual acuity and that longer-term follow-up might reveal visual acuity benefits, or that treatment has different effects on these two measures in this subgroup. Nevertheless, reducing the risk of a loss of contrast sensitivity might represent a treatment benefit for these patients.
Contrast sensitivity was also measured as a secondary outcome in the Verteporfin In Photodynamic therapy (VIP) Trial.52 In the VIP Trial, 20% of verteporfin-treated patients with occult with no classic CNV lost at least 9 letters of contrast sensitivity at the month 24 examination, compared with 34% of placebo recipients (P=0.01). As in the patients with predominantly classic CNV included in the TAP Investigation,50 the contrast sensitivity findings in patients with occult with no classic CNV were consistent with the visual acuity findings. At the month 24 examination, 55% of verteporfin-treated eyes with occult with no classic CNV had lost at least 15 letters of visual acuity, compared with 68% of the placebo recipients (P=0.032).52
Comparison of the visual acuity and contrast sensitivity outcomes between verteporfin-treated eyes in the TAP Investigation and VIP Trial and eyes that received laser photocoagulation in the MPS is difficult because of differences in the characteristics of the eyes enrolled in these studies. Nevertheless, it is worth noting that the results from verteporfin therapy in the TAP Investigation and laser photocoagulation in the MPS both showed stabilisation of contrast sensitivity. At the month 24 examinations, verteporfin-treated eyes lost a mean of 1.3 letters from baseline50 and laser-treated eyes with classic CNV with no occult CNV had no change in median contrast threshold from baseline (14% at both visits, which is equivalent to 20 letters on the currently available chart).46 Untreated eyes in the TAP Investigation lost a mean of 5.2 letters of contrast sensitivity by the month 24 examination,50 and the contrast threshold of untreated eyes in the MPS deteriorated from 14% (20 letters) at baseline to 28% (14 letters) at month 24.46
Current guidelines for the treatment of CNV due to AMD recommend verteporfin therapy for subfoveal lesions that are composed of predominantly classic CNV or occult with no classic CNV, based on the visual acuity outcomes.44 The contrast sensitivity outcomes described above strengthen the evidence for recommending verteporfin therapy in these patients. Laser photocoagulation is generally recommended only for lesions that are extrafoveal or juxtafoveal, although before verteporfin therapy became available it was also considered for small subfoveal lesions.53
Mobility and orientation are both associated with visual field and contrast sensitivity.31, 32, 33 Treatments that reduce the loss of both contrast sensitivity and central visual field may therefore help to maintain mobility and orientation. Although no measurements of central visual field have been reported in the randomised, controlled clinical trials of verteporfin therapy, treatment has been shown to reduce lesion growth, compared with placebo. At the month 24 examination in the TAP Investigation, verteporfin-treated eyes had smaller lesions than eyes that were given placebo: lesions larger than 9 MPS disc areas occurred in 15% of verteporfin-treated eyes and 38% of eyes given placebo.50 Similarly, in AMD patients with occult with no classic CNV enrolled in the VIP Trial 16% of verteporfin-treated eyes had lesions larger than 9 MPS disc areas at the month 24 examination, compared with 38% of eyes given placebo.52 The strong correlation between lesion size and contrast sensitivity,18 suggests that reducing lesion size may also help to preserve contrast sensitivity.
There have been anecdotal reports of patients treated with verteporfin therapy claiming to have improved vision and a reduced perception of scotoma (increased transparency) after treatment despite having no increase (or even a decrease) in visual acuity. It is possible that improvements in contrast sensitivity could explain these cases. It should be noted that this hypothesis has not been directly tested in clinical studies and should be treated with caution.
Radiation therapy
Radiation therapy has been investigated in several small, uncontrolled studies, some of which have indicated that treatment could stabilise visual acuity.54, 55 A study of 203 patients with subfoveal CNV due to AMD found no significant benefits of external beam radiotherapy (compared with observation alone) in terms of the primary outcome measure, the proportion of patients with a visual acuity loss of at least 3 lines.56 However, there was a significant benefit in terms of the percentage of patients with a loss of at least 0.3 log units of contrast sensitivity (representing a halving of the contrast threshold from the baseline value) at 24 months. Positive contrast sensitivity findings have also been reported in small-scale studies of radiation therapy,57 although other studies, including the largest controlled trial of radiation therapy to date, have reported no significant benefits.58, 59
Other treatment modalities
A variety of surgical techniques have been investigated for patients with neovascular AMD. Submacular surgery frequently has poor visual outcomes,60, 61, 62 possibly because the CNV is often irreversibly attached to the RPE. To date, no large-scale randomised clinical trials have reported the effects of surgical techniques on visual acuity and contrast sensitivity. More information will become available on completion of an ongoing National Institutes of Health (NIH)-sponsored investigation, the Submacular Surgery Trials.63, 64
Other investigational approaches to the treatment of neovascular AMD include transpupillary thermotherapy and antiangiogenic and angiostatic agents,65 but there have been no reports of contrast sensitivity outcomes with these approaches.
Summary
A survey of ophthalmologists attending a UK ophthalmology congress suggested that contrast sensitivity is generally considered to be of low importance compared with visual acuity.66 Our review of the literature suggests that this perception is not supported by the available evidence.
Contrast sensitivity is an important, additional measure of visual function in patients with subfoveal CNV secondary to AMD. In particular, contrast sensitivity may be a better predictor of activities of daily living, mobility and orientation than visual acuity. Including contrast sensitivity as an outcome measure in studies of patients with CNV due to AMD may provide a more complete understanding of the effects of treatment on visual function and the likely benefits for patients.
Laser photocoagulation (rarely used for subfoveal lesions) and verteporfin therapy (currently recommended for subfoveal lesions that are composed of predominantly classic CNV or occult with no classic CNV with evidence of recent disease progression) have been found to reduce the risk of losing both contrast sensitivity and visual acuity in patients with neovascular AMD. Verteporfin therapy was also found to improve contrast sensitivity outcomes in patients with minimally classic CNV even in the absence of a visual acuity benefit, which may represent a potential treatment benefit for these patients. Further investigation is needed to explore the impact of treatment on contrast sensitivity and visual acuity in patients with subfoveal CNV due to AMD.
References
Arden GB . The importance of measuring contrast sensitivity in cases of visual disturbance. Br J Ophthalmol 1978; 62: 198–209.
Elliott DB, Hurst MA, Weatherill J . Comparing clinical tests of visual function in cataract with the patient's perceived visual disability. Eye 1990; 4: 712–717.
Rubin GS . Reliability and sensitivity of clinical contrast sensitivity tests. Clin Vision Sci 1988; 2: 169–177.
Rubin GS, Roche KB, Prasada-Rao P, Fried LP . Visual impairment and disability in older adults. Optom Vis Sci 1994; 71: 750–760.
Rubin GS, Bandeen-Roche K, Huang GH, Munoz B, Schein OD, Fried LP et al. The association of multiple visual impairments with self-reported visual disability: SEE project. Invest Ophthalmol Vis Sci 2001; 42: 64–72.
Jin CJ, Wu DZ, Wu L . The contrast sensitivity function in low vision. Yen Ko Hsueh Pao 1992; 8: 45–48.
Foran S, Wang JJ, Mitchell P . Causes of visual impairment in two older population cross-sections: the Blue Mountains Eye Study. Ophthalmic Epidemiol 2003; 10: 215–225.
Hyvarinen L, Laurinen P, Rovamo J . Contrast sensitivity in evaluation of visual impairment due to macular degeneration and optic nerve lesions. Acta Ophthalmol (Copenh) 1983; 61: 161–170.
Bressler NM, Bressler SB, Fine SL . Age-related macular degeneration. Surv Ophthalmol 1988; 32: 375–413.
Hyman L In: Hampton GR, Nelsen PT (eds) Age-related Macular Degeneration: Principles and Practice. Raven Press: New York, 1992, pp. 1–35.
Rubin GS, Bressler NM . Effects of verteporfin therapy on contrast sensitivity: results from the Treatment of Age-Related Macular Degeneration With Photodynamic Therapy (TAP) Investigation—TAP Report No. 4. Retina 2002; 22: 536–544.
Pelli DG, Robson JG, Wilkins AJ . The design of a new letter chart for measuring contrast sensitivity. Clin Vision Sci 1988; 2: 187–199.
Elliott DB, Bullimore MA, Bailey IL . Improving the reliability of the Pelli–Robson contrast sensitivity test. Clin Vision Sci 1991; 6: 471–475.
Greeves AL, Cole BL, Jacobs RJ . Assessment of contrast sensitivity of patients with macular disease using reduced contrast near visual acuity charts. Ophthalmic Physiol Opt 1988; 8: 371–377.
Bellmann C, Unnebrink K, Rubin GS, Miller D, Holz FG . Visual acuity and contrast sensitivity in patients with neovascular age-related macular degeneration. Results from the Radiation Therapy for Age-Related Macular Degeneration (RAD-) Study. Graefes Arch Clin Exp Ophthalmol 2003; 241: 968–974.
Rubin GS, West SK, Munoz B, Bandeen-Roche K, Zeger S, Schein O et al. A comprehensive assessment of visual impairment in a population of older Americans. The SEE Study. Salisbury Eye Evaluation Project. Invest Ophthalmol Vis Sci 1997; 38: 557–568.
Haegerstrom-Portnoy G, Schneck ME, Lott LA, Brabyn JA . The relation between visual acuity and other spatial vision measures. Optom Vis Sci 2000; 77: 653–662.
Doris N, Hart PM, Chakravarthy U, McCleland J, Stevenson M, Hudson C et al. Relation between macular morphology and visual function in patients with choroidal neovascularisation of age related macular degeneration. Br J Ophthalmol 2001; 85: 184–188.
Ivers RQ, Mitchell P, Cumming RG . Visual function tests, eye disease and symptoms of visual disability: a population-based assessment. Clin Experiment Ophthalmol 2000; 28: 41–47.
McGwin Jr G, Chapman V, Owsley C . Visual risk factors for driving difficulty among older drivers. Accid Anal Prev 2000; 32: 735–744.
Wood JM . Age and visual impairment decrease driving performance as measured on a closed-road circuit. Hum Factors 2002; 44: 482–494.
Owsley C, McGwin Jr G, Sloane ME, Stalvey BT, Wells J . Timed instrumental activities of daily living tasks: relationship to visual function in older adults. Optom Vis Sci 2001; 78: 350–359.
Lott LA, Schneck ME, Haegerstrom-Portnoy G, Brabyn JA, Gildengorin GL, West CG . Reading performance in older adults with good acuity. Optom Vis Sci 2001; 78: 316–324.
Alexander MF, Maguire MG, Lietman TM, Snyder JR, Elman MJ, Fine SL . Assessment of visual function in patients with age-related macular degeneration and low visual acuity. Arch Ophthalmol 1988; 106: 1543–1547.
Leat SJ, Woodhouse JM . Reading performance with low vision aids: relationship with contrast sensitivity. Ophthalmic Physiol Opt 1993; 13: 9–16.
Legge GE, Ross JA, Isenberg LM, LaMay JM . Psychophysics of reading. Clinical predictors of low-vision reading speed. Invest Ophthalmol Vis Sci 1992; 33: 677–687.
Tejeria L, Harper RA, Artes PH, Dickinson CM . Face recognition in age related macular degeneration: perceived disability, measured disability, and performance with a bioptic device. Br J Ophthalmol 2002; 86: 1019–1026.
Owsley C, Sekuler R, Boldt C . Aging and low-contrast vision: face perception. Invest Ophthalmol Vis Sci 1981; 21: 362–365.
Owsley C, Sloane ME . Contrast sensitivity, acuity, and the perception of ‘real-world’ targets. Br J Ophthalmol 1987; 71: 791–796.
Scott IU, Feuer WJ, Jacko JA . Impact of visual function on computer task accuracy and reaction time in a cohort of patients with age-related macular degeneration. Am J Ophthalmol 2002; 133: 350–357.
Kuyk T, Elliott JL . Visual factors and mobility in persons with age-related macular degeneration. J Rehabil Res Dev 1999; 36: 303–312.
Hassan SE, Lovie-Kitchin JE, Woods RL . Vision and mobility performance of subjects with age-related macular degeneration. Optom Vis Sci 2002; 79: 697–707.
Marron JA, Bailey IL . Visual factors and orientation-mobility performance. Am J Optom Physiol Opt 1982; 59: 413–426.
Klein BE, Klein R, Lee KE, Cruickshanks KJ . Performance-based and self-assessed measures of visual function as related to history of falls, hip fractures, and measured gait time. The Beaver Dam Eye Study. Ophthalmology 1998; 105: 160–164.
Ivers RQ, Cumming RG, Mitchell P, Attebo K . Visual impairment and falls in older adults: the Blue Mountains Eye Study. J Am Geriatr Soc 1998; 46: 58–64.
Cummings SR, Nevitt MC, Browner WS, Stone K, Fox KM, Ensrud KE et al. Risk factors for hip fracture in white women. Study of Osteoporotic Fractures Research Group. N Engl J Med 1995; 332: 767–773.
Dargent-Molina P, Favier F, Grandjean H, Baudoin C, Schott AM, Hausherr E et al. Fall-related factors and risk of hip fracture: the EPIDOS prospective study. Lancet 1996; 348: 145–149.
Brown GC . Vision and quality-of-life. Trans Am Ophthalmol Soc 1999; 97: 473–511.
Brown GC, Sharma S, Brown MM, Kistler J . Utility values and age-related macular degeneration. Arch Ophthalmol 2000; 118: 47–51.
Brown GC, Brown MM, Sharma S . Difference between ophthalmologists' and patients' perceptions of quality of life associated with age-related macular degeneration. Can J Ophthalmol 2000; 35: 127–133.
Carta A, Braccio L, Belpoliti M, Soliani L, Sartore F, Gandolfi SA et al. Self-assessment of the quality of vision: association of questionnaire score with objective clinical tests. Curr Eye Res 1998; 17: 506–511.
Klein BE, Moss SE, Klein R, Lee KE, Cruickshanks KJ . Associations of visual function with physical outcomes and limitations 5 years later in an older population: the Beaver Dam Eye Study. Ophthalmology 2003; 110: 644–650.
American Academy of Ophthalmology. Preferred Practice Pattern™. Age-related Macular Degeneration. http://www.aao.org, 2001, Accessed June 2004.
Verteporfin Roundtable 2000 Participants, Treatment of Age-Related Macular Degeneration with Photodynamic Therapy (TAP) Study Group Principal Investigators, Verteporfin In Photodynamic Therapy (VIP) Study Group Principal Investigators. Guidelines for using verteporfin (Visudyne®) in photodynamic therapy to treat choroidal neovascularization due to age-related macular degeneration and other causes. Retina 2002; 22: 6–18.
Macular Photocoagulation Study Group. Laser photocoagulation of subfoveal recurrent neovascular lesions in age-related macular degeneration. Results of a randomized clinical trial. Arch Ophthalmol 1991; 109: 1232–1241.
Macular Photocoagulation Study Group. Laser photocoagulation of subfoveal neovascular lesions in age-related macular degeneration. Results of a randomized clinical trial. Arch Ophthalmol 1991; 109: 1220–1231.
Macular Photocoagulation Study Group. Argon laser photocoagulation for neovascular maculopathy. Five-year results from randomized clinical trials. Arch Ophthalmol 1991; 109: 1109–1114.
Macular Photocoagulation Study Group. Laser photocoagulation for juxtafoveal choroidal neovascularization. Five-year results from randomized clinical trials. Arch Ophthalmol 1994; 112: 500–509.
Treatment of Age-Related Macular Degeneration with Photodynamic Therapy (TAP) Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin: one-year results of 2 randomized clinical trials—TAP Report 1. Arch Ophthalmol 1999; 117: 1329–1345.
Treatment of Age-Related Macular Degeneration with Photodynamic Therapy (TAP) Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin: two-year results of 2 randomized clinical trials—TAP Report 2. Arch Ophthalmol 2001; 119: 198–207.
Treatment of Age-Related Macular Degeneration with Photodynamic Therapy (TAP) and Verteporfin In Photodynamic Therapy (VIP) Study Groups. Effect of lesion size, visual acuity, and lesion composition on visual acuity change with and without verteporfin therapy for choroidal neovascularization secondary to age-related macular degeneration—TAP and VIP Report No. 1. Am J Ophthalmol 2003; 136: 407–418.
Verteporfin In Photodynamic Therapy (VIP) Study Group. Verteporfin therapy of subfoveal choroidal neovascularization in age-related macular degeneration: two-year results of a randomized clinical trial including lesions with occult with no classic choroidal neovascularization—Verteporfin In Photodynamic Therapy Report 2. Am J Ophthalmol 2001; 131: 541–560.
Macular Photocoagulation Study Group. Subfoveal neovascular lesions in age-related macular degeneration: guidelines for evaluation and treatment in the Macular Photocoagulation Study. Arch Ophthalmol 1991; 109: 1242–1257.
Bergink GJ, Hoyng CB, van der Maazen RW, Vingerling JR, van Daal WA, Deutman AF . A randomized controlled clinical trial on the efficacy of radiation therapy in the control of subfoveal choroidal neovascularization in age-related macular degeneration: radiation versus observation. Graefes Arch Clin Exp Ophthalmol 1998; 236: 321–325.
Chakravarthy U, Houston RF, Archer DB . Treatment of age-related subfoveal neovascular membranes by teletherapy: a pilot study. Br J Ophthalmol 1993; 77: 265–273.
Hart PM, Chakravarthy U, Mackenzie G, Chisholm IH, Bird AC, Stevenson MR et al. Visual outcomes in the subfoveal radiotherapy study: a randomized controlled trial of teletherapy for age-related macular degeneration. Arch Ophthalmol 2002; 120: 1029–1038.
Jaakkola A, Heikkonen J, Tarkkanen A, Immonen I . Visual function after strontium-90 plaque irradiation in patients with age-related subfoveal choroidal neovascularization. Acta Ophthalmol Scand 1999; 77: 57–61.
Marcus DM, Sheils WC, Johnson MH, McIntosh SB, Leibach DB, Maguire A et al. External beam irradiation of subfoveal choroidal neovascularization complicating age-related macular degeneration: one-year results of a prospective, double-masked, randomized clinical trial. Arch Ophthalmol 2001; 119: 171–180.
Marcus DM, Sheils WC, Young JO, McIntosh SB, Johnson MH, Alexander J et al. Radiotherapy for recurrent choroidal neovascularisation complicating age related macular degeneration. Br J Ophthalmol 2004; 88: 114–119.
Lewis H . Subfoveal choroidal neovascularization: is there a role for submacular surgery? Am J Ophthalmol 1998; 126: 127–129.
Olsen TW . Submacular surgery for age-related macular degeneration. Semin Ophthalmol 1997; 12: 34–44.
Roth DB, Downie AA, Charles ST . Visual results after submacular surgery for neovascularization in age-related macular degeneration. Ophthalmic Surg Lasers 1997; 28: 920–925.
Bressler NM . Submacular surgery. New information, more questions. Arch Ophthalmol 1997; 115: 1071–1072.
Bressler NM, Hawkins BS, Steinberg P, McDonald HR . Are the submacular surgery trials still relevant in an era of photodynamic therapy? Ophthalmology 2001; 108: 435–436.
Hooper CY, Guymer RH . New treatments in age-related macular degeneration. Clin Experiment Ophthalmol 2003; 31: 376–391.
Hart PM, Chakravarthy U, Stevenson MR . Questionnaire-based survey on the importance of quality of life measures in ophthalmic practice. Eye 1998; 12: 124–126.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Monés, J., Rubin, G. Contrast sensitivity as an outcome measure in patients with subfoveal choroidal neovascularisation due to age-related macular degeneration. Eye 19, 1142–1150 (2005). https://doi.org/10.1038/sj.eye.6701717
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.eye.6701717
Keywords
This article is cited by
-
Impact of intravitreal injection therapy on contrast sensitivity in patients with nAMD and DME
Graefe's Archive for Clinical and Experimental Ophthalmology (2023)
-
Association of Vision-related Quality of Life with Visual Function in Age-Related Macular Degeneration
Scientific Reports (2019)
-
Cost-Effectiveness Models in Age-Related Macular Degeneration: Issues and Challenges
PharmacoEconomics (2016)
-
Baseline clinical measures and early response predict success in verteporfin photodynamic therapy for neovascular age-related macular degeneration
Eye (2010)
-
Contrast sensitivity after intravitreal antivascular endothelial growth factor therapy for myopic choroidal neovascularization
Graefe's Archive for Clinical and Experimental Ophthalmology (2010)