Abstract
Purpose
To investigate the influence of subfoveal choroidal neovascularisation (CNV) on macular imaging performed using scanning laser polarimetry (SLP) of the retinal nerve fibre layer.
Methods
SLP was performed on 22 consecutive patients with angiographically verified CNV, and on 23 healthy control subjects. One eye per subject was evaluated using the GDx Nerve Fibre Analyser. Regularity of the corneal retardation on the macular SLP images was assessed according to three criteria: (1) magnitude of the ‘macular ratio’, defined as the ratio of mean retardation values along two axes (the axis with the maximum retardation and the perpendicular one, corresponding in healthy eyes to minimum retardation); (2) the values of GDx parameters which are independent of quadrant position (ellipse modulation and ellipse average); and (3) the frequency of the regular ‘bow–tie’ polarisation pattern.
Results
‘Macular ratio’ was significantly higher in the CNV group than in the control group (P≪0.001). Ellipse modulation did not differ between the groups, but ellipse average was higher in the CNV group (P=0.016). The variance for each of these two parameters was significantly higher for the CNV group (P≪0.001 for both comparisons). A ‘bow–tie’ pattern polarisation was seen in 23 of the 23 control eyes, but only in 7 of the 22 CNV eyes (P≪0.001).
Conclusion
The results show that CNV influences the macular image obtained with SLP. This suggests that measurements with SLP may be disturbed for eyes with CNV when the customised corneal compensation method, which makes use of the macular retardation image, is employed.
Similar content being viewed by others
Introduction
Scanning laser polarimetry (SLP) of the retinal nerve fibre layer is a noninvasive method, which has been widely used for glaucoma diagnosis in the last 10 years.1, 2, 3, 4, 5 SLP employs an illuminating laser beam, and measures the optical retardation, which is produced by the parallel retinal nerve fibres around the optic nerve head.6 Since the optical retardation caused by the retinal ganglion cell axons has been shown to have a linear relationship with the anatomical thickness of the retinal nerve fibre layer (RNFL), a decrease of the RNFL thickness corresponds to a loss of the retinal ganglion cells, which is the most important pathological alteration in glaucoma. In order to improve the diagnostic power of SLP, a new customised corneal compensation technique has recently been introduced.7, 8 With the conventional compensation method,1, 2, 3, 4, 5 the compensator's polarisation magnitude and slow axis orientation are fixed, and it is therefore unable in most cases to achieve complete neutralisation of the retardation caused by the cornea.9, 10, 11, 12, 13, 14, 15 In contrast, in the customised compensation technique, a polarimetric image of the macula is acquired as the first step of the measurement.7, 8, 16 Since the fovea has no retinal ganglion cell axons, the resulting polarimetric image of the macular area represents the sum of the polarimetric effects only of the cornea, the lens, and the Henle fibre layer. The second step is the imaging of the actual peripapillary RNFL. Data derived from the macular image are then automatically used for correction of the peripapillary polarimetric image. Finally, the resulting corrected image of the peripapillary RNFL is analysed by the software in the same way as for the fixed-compensation method.
Subfoveal choroidal neovascularisation (CNV) is a major cause of blindness in elderly people,17, 18, 19, 20, 21 as also is glaucoma; coincidence of the two diseases in the same patient is not infrequent.19, 20, 22, 23 However, the presence of CNV is not always obvious in a routine examination of the eye without performing angiography, especially in patients with coexisting nuclear sclerosis or glaucoma.21 It has been shown that optical retardation caused by the sclera in eyes with atrophy of the choroid and the retina leads to significant artefacts in SLP measurements.24 It is possible that CNV may also influence the measured retardation in SLP when using the new customised compensation method, where the macular area is imaged as a step in the corneal polarisation compensation process.
In order to investigate the influence of CNV on the polarimetric image obtained using SLP, we compared macular images obtained with the GDx Nerve Fibre Analyser on diseased eyes (with angiographically verified CNV) to images acquired on healthy eyes with no macula disease.
Patients and methods
The clinical investigation protocol was approved by the Ethics Committee of Semmelweis University. In all, 22 consecutive phakic Caucasian patients suffering from CNV (age 32–84 years, mean age 70.7 years) were enroled in the study, together with 23 healthy Caucasian control subjects without any ocular abnormality (age 22–44 years, mean age 28.5 years). Each enroled patient had sufficient central vision for optimal fixation with the fellow eye.
The patients with CNV had had no previous ocular surgery or photocoagulation on the study eye. In this group, 15 study eyes, had previously received photodynamic therapy. In all cases, the refractive media of the eye were sufficiently clear both for optimal SLP imaging and for fluorescein angiography. At the time of the SLP investigation, the visual acuity of the diseased eyes ranged between 0.05 and 0.50 (mean visual acuity 0.21). All the study eyes in the CNV group had subfoveal CNV. Based on the results found with fluorescein angiography, the lesions in seven eyes were of predominantly classic type; in one eye it was purely occult, and in 14 eyes they were of 100% classic type.17 The greatest linear dimension of the CNV did not exceed 7900 μm. Fluorescein angiography and CNV classification were performed by the same investigator (AP). The control subjects, recruited from the hospital staff, had undergone no previous ocular interventions. The visual acuity was 1.0 for each eye, and the visual field in each case was classified as normal using the Octopus G2 normal strategy test (Octopus 101 perimetry instrument, Interzeag AG, Schlieren, Switzerland). The retina and the macula of each eye in the control group were free from pathological alterations, as determined by examination using noncontact aspherical lenses and a slit -lamp for stereoscopic viewing.
SLP of the retinal nerve fibre layer (without pupil dilation) was performed by the same experienced investigator (AK) using the GDx Nerve Fibre Analyser instrument with software version 1.0.16 (Laser Diagnostic Technologies, Inc., San Diego, CA, USA). This noninvasive measurement technique has been described in detail elsewhere.1, 2, 3, 4, 5 In brief, a beam of 780 nm polarised laser light is projected onto the retina by the instrument. The birefringent parallel nerve fibres cause retardation in the polarisation of the light passing through them, as compared to the light passing between the fibres. This retardation of the reflected light is measured automatically by the detector unit of the instrument. The degree of the retardation corresponds to the thickness of the RNFL, and is analysed by the built-in polarisation detection unit. The RNFL thickness is automatically calculated, and is processed in a colour-coded manner in each of the 256 × 256 individual retinal positions (pixels) of a 15° retinal area. Image acquisition requires only 0.7 s. To stabilise fixation, an external fixating light is used. In the conventional compensation technique (used in the present investigation), to compensate for optical retardation caused by the central cornea a built-in fixed-angle compensator is used; this has a polarisation magnitude of 60 nm, with the slow axis set to 15° nasally downwards. In our investigations, three high-quality individual images were acquired in each case via an undilated pupil, and an averaged image was calculated using the software. The 10 pixels wide measuring ellipse was set at 1.75 disc diameters. One eye per subject was evaluated. In the control group and in case of CNV, in both eyes the selection was random; in case of no CNV in the fellow eye, the diseased eye was selected for evaluation. During image acquisition, the fovea was positioned in the centre of the screen.
Three different evaluation approaches were used. In the first method, we employ a discrimination -parameter which we call ‘macular ratio’, derived in the manner described below. The thickness value overlay software menu on the GDx imaging device was used; this displays a regular grid of boxes (each of size 16 × 16 pixels) superimposed on the retardation image, and shows the mean thickness value for each box (in μm). In our evaluation procedure, we first determined the centre of the fovea on the image. We calculated the mean retardation for the axis of maximum corneal retardation, using the retardation values of four neighbouring boxes along this axis in each direction from the fovea. We then used a similar technique to determine the mean retardation along the perpendicular axis (in eyes with a normal macular image, the minimum retardation occurs along this axis). The ratio of these two mean values (which we call ‘macular ratio’; the value for the ‘minimum axis’ divided by that for the ‘maximum axis’) was determined, and compared between the CNV and control groups. In our second approach, the GDx ‘ellipse modulation’ and ‘ellipse average’ parameter values were evaluated. These software-provided parameters are not influenced by the quadrant orientation or by the angle of the axis of corneal polarisation. The mean values and the standard deviations of these two parameters were compared between the groups. In our third method, the presence or absence on the macular images of the normal characteristic ‘bow–tie’ polarisation pattern caused by the corneal retardation was evaluated subjectively; the evaluation was carried out in a masked fashion by the same investigator (PK), without using the results of the other two investigations.
The unpaired t-test with separate variance estimation was used to compare ‘macular ratio’, ellipse modulation, and ellipse average between the groups. The F-test was used to compare variances for ellipse modulation, and for ellipse average between the groups. The Fisher's exact test was used to compare the frequency of the regular ‘bow–tie’ polarisation pattern between the CNV group and the controls. P-values of less than 0.05 were considered statistically significant.
Results
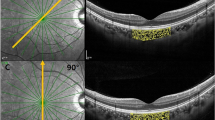
Typical examples of the appearance of corneal retardation on the macular SLP images, in this case those taken from the healthy eye and the CNV eye of the same patient, are shown in Figure 1. The angiographical image of this CNV eye is shown in Figure 2. The difference in the regularity of these retardation patterns, typical for CNV and healthy eyes, is clearly visible.
Fluorescein angiographic appearance of the subfoveal neovascular membrane. Same eye as in Figure 1(a).
In all eyes, polarisation images suitable for analysis were obtained. The results of the group comparisons are shown in Table 1. ‘Macular ratio’ was significantly higher in the CNV group than in the control group (P≪0.001). Ellipse modulation did not differ significantly between the groups, but ellipse average was higher in the CNV group (P=0.016). The variance for each of these parameters was significantly higher for the CNV group (P≪0.001 for both comparisons). When the images were subjectively evaluated for regularity, a highly significant difference was found; a ‘bow–tie’ pattern was found in 23 of the 23 control eyes but only in seven of the 22 CNV eyes (P≪0.001). The retardation images of the other 15 eyes showed a more uniform polarisation pattern around the fovea with irregular areas of intensive retardation.
Discussion
In the present cross-sectional study the influence of CNV on the macular SLP images was investigated. The background of our investigation is the recent introduction of customised corneal polarisation compensation for SLP in clinical practice.7, 8, 16 Since the orientation of the slow axis of corneal retardation varies considerably between different eyes, it cannot be completely neutralised in every case with the conventional fixed-angle compensation.9, 10, 11, 12, 13, 14, 15 With customised corneal polarisation compensation, the peripapillary polarimetric image is automatically corrected using data from the macular retardation image acquired as the first step of the measurement procedure. Since the fovea is free from retinal nerve fibres, in eyes with healthy macula this image represents the polarimetric effects only of the central cornea, the lens, and the retinal Henle layer.7, 8 In a previous study we have shown that, in contrast to fixed-angle compensation, customised compensation effectively neutralises the retardation changes of the cornea caused by laser-assisted in situ keratomileusis.16 Direct RNFL thickness values obtained with the customised compensation technique seem to be more valid than those obtained with the fixed-angle compensation.7, 8 These results suggest that customised corneal compensation may have clinical importance in glaucoma, both for diagnosis and follow-up.
Macular diseases and especially CNV, however, represent a potential problem for SLP measurements with customised compensation. CNV due to age-related macular degeneration is one of the most frequent eye diseases in the elderly, and its coexistence with glaucoma, another very frequent disease in the elderly population, is not unusual.17, 18, 19, 20, 21, 22, 23 Moreover, CNV may develop during glaucoma follow-up, and its influence on the SLP results used for monitoring the status of the glaucoma might then inappropriately influence therapeutic decisions. This is a real possibility, since the development of CNV is not always evident in a routine eye examination without pupillary dilation, if the visual acuity is not severely damaged and the eye has coexistent nuclear sclerosis.
In our study, the regularity of the retardation pattern on the macular SLP images was investigated, and compared between a group of eyes with CNV and a control group of healthy eyes. Retardation on the macular image is caused by the polarisation from the anterior segment of the eye, mainly from the cornea. In CNV, however, this may be superimposed on the polarisation effect of the subfoveal fibrovascular tissue, blood and exudate. This possibility is strongly supported by published data on the considerable polarimetric effect of the sclera, which is clearly visible and measurable in eyes with peripapillary atrophy.24 Although in principle, the axis and magnitude of the measured macular retardation may also be influenced by the CNV condition, such an influence was not investigated in the present study. The slow axis of the corneal polarisation is variable in the population, and therefore a comparison would not provide useful information in this pilot study with a relatively small number of participants. The magnitude as well as the angle of the anterior segment retardation is variable among eyes; the instrument we used for SLP measurements had a fixed-magnitude compensator, and this probably influenced the measured values. However, the regularity of the retardation, that is, the typical ‘bow–tie’ appearance of retardation contours on the macular image, is independent of both the magnitude and the angle of the anterior segment polarisation, and therefore is appropriate for comparison purposes.
As evaluated by one of the investigators, a regular ‘bow–tie’ retardation pattern was found only in one-third of the CNV eyes, but in all of the control eyes. This difference is significant both in statistical and clinical terms, and confirms the recent data obtained with SLP on eyes with different macular pathologies.25 The ‘macular ratio’, the ratio of the retardation for the axis with minimum retardation over that for the perpendicular axis with maximum retardation, was significantly higher in the CNV group, showing objectively that even the axis with the minimum corneal retardation does have a considerable retardation in CNV eyes, compared to that found in healthy eyes. This effect can be attributed to the polarising property of the subretinal fibrovascular tissue, blood, and exudate, which of course does not have the ‘bow–tie’ spatial pattern typical for the corneal effect. This irregular polarisation pattern of the CNV group was verified in a different way when ellipse modulation and ellipse average, parameters not influenced by quadrant positions or the position of the axis of corneal polarisation, were compared. The variance (and standard deviation) for each of the two parameters was significantly higher in the CNV group, which suggests that the retardation values in the CNV group did not show a regular polarisation pattern.
In conclusion, our results show that subfoveal CNV does influence the macular image with SLP, and therefore may influence the results of SLP performed with the customised compensation method. Further studies are necessary to clarify the magnitude of this influence and its significance in clinical decisions. Our results also suggest that besides the newly described ‘screen’ compensation method for SLP with variable corneal compensation,25 fixed-angle polarisation compensation method, which does not use macular images, may still have an appropriate future role when using SLP on eyes with CNV.
References
Holló G, Süveges I, Nagymihály A, Vargha P . Scanning laser polarimetry of the retinal nerve fibre layer in primary open angle and capsular glaucoma. Br J Ophthalmol 1997; 81: 857–861.
Weinreb RN, Zangwill L, Berry CC, Bathija R, Sample PA . Detection of glaucoma with scanning laser polarimetry. Arch Ophthalmol 1998; 116: 1583–1589.
Yamada N, Chen PP, Mills RP, Leen MM, Stamper RL, Lieberman MF et al. Glaucoma screening using the scanning laser polarimeter. J Glaucoma 2000; 9: 254–261.
Choplin NT, Lundy DC . The sensitivity and specificity of scanning laser polarimetry in the detection of glaucoma in a clinical setting. Ophthalmology 2001; 108: 899–904.
Poinoosawmy D, Tan JCH, Bunce C, Hichings RA . The ability of the GDx Nerve Fibre Analyser neural network to diagnose glaucoma. Graefe's Arch Clin Exp Ophthalmol 2001; 239: 122–127.
Weinreb RN, Dreher AW, Coleman A . Histopathologic validation of Fourier-ellipsometry measurements of retinal nerve fiber thickness. Arch Ophthalmol 1990; 108: 557–560.
Weinreb RN, Bowd C, Zangwill LM . Glaucoma detection using scanning laser polarimetry with variable corneal polarization compensation. Arch Ophthalmol 2003; 121: 218–224.
Bagga H, Greenfield DS, Feuer W, Knighton RW . Scanning laser polarimetry with variable corneal compensation and optical coherence tomography in normal and glaucomatous eyes. Am J Ophthalmol 2003; 135: 521–529.
Greenfield DS, Knighton RW, Huang X-R . Effect of corneal polarization axis on assessment of retinal nerve fiber layer thickness by scanning laser polarimetry. Am J Ophthalmol 2000; 129: 715–722.
Kogure S, Chiba T, Kinoshita T, Kowa H, Tsukahara S . Effects of artefacts on scanning laser polarimetry of retinal nerve fibre layer thickness measurement. Br J Ophthalmol 2000; 84: 1013–1017.
Zhou Q, Weinreb RN . Individualized compensation of anterior segment birefringence during scanning laser polarimetry. Invest Ophthalmol Vis Sci. 2002; 43: 2221–2228.
Weinreb RN, Bowd C, Greenfield DS, Zangwill LM . Measurement of the magnitude and axis of corneal polarization with scanning laser polarimetry. Arch Ophthalmol 2002; 120: 901–906.
Knighton RW, Huang X-R, Greenfield DS . Analytical model of scanning laser polarimetry for retinal nerve fibre layer assessment. Invest Ophthalmol Vis Sci. 2002; 43: 383–392.
Greenfield DS, Knighton RW, Feuer WJ, Schiffman JC, Zangwill L, Weinreb RN . Correction for corneal polarization axis improves the discriminating power of scanning laser polarimetry. Am J Ophthalmol 2002; 134: 27–33.
Garway-Heath D, Greaney MJ, Caprioli J . Correction for erroneous compensation of anterior segment birefringence with the scanning laser polarimeter for glaucoma diagnosis. Invest Ophthalmol Vis Sci 2002; 43: 1465–1474.
Holló G, Katsanos A, Kóthy P, Kerek A, Süveges I . Influence of LASIK on scanning laser polarimetric measurement of the retinal nerve fibre layer with fixed angle and customised corneal polarisation compensation. Br J Ophthalmol 2003; 87: 1241–1246.
Macular Photocoagulation Study Group. Subfoveal neovascular lesions in age-related macular degeneration. Arch Ophthalmol 1991; 109: 1242–1257.
Vingerling JR, Dielemans I, Hofman A, Grobee DE, Hijmering M, Kramer CFL et al. The prevalence of age-related maculopathy in the Rotterdam Study. Ophthalmology 1995; 102: 205–210.
Buch H, Vinding T, Nielsen NV . Prevalence and causes of visual impairement according to World Health Organisation and United States criteria in an aged, urban, Scandinavian population. Ophthalmology 2001; 108: 2347–2357.
Trautner C, Haastert B, Richter B, Berger M, Giani G . Incidence of blindness in southern Germany due to glaucoma and degenerative conditions. Invest Ophthalmol Vis Sci 2003; 44: 1031–1034.
Sickenberg M . Early detection, diagnosis and management of choroidal neovascularisation in age-related macular degeneration: the role of ophthalmologists. Ophthalmologica 2001; 215: 247–253.
Perasalo R . The prevalence of macular degeneration in a cohort of institutionalized geriatric glaucoma patients. Acta Ophthalmol 1994; 72: 175–177.
Kozobolis VP, Detorakis ET, Tsilimbaris M, Vlachonicolis IG, Tsambarlakis IC, Pallikaris IG . Correlation between age-related macular degeneration and pseudoexfoliation syndrome in the population of Crete (Greece). Arch Ophthalmol 1999; 117: 664–669.
Hoh ST, Greenfield DS, Liebmann JM, Maw R, Ishikawa H, Chew SJ et al. Factors affecting image acquisition during scanning laser polarimetry. Ophthalmic Surg Lasers 1998; 29: 545–551.
Bagga H, Greenfield DS, Knighton RW . Scanning laser polarimetry with variable corneal compensation: identification and correction for corneal birefringence in eyes with macular disease. Invest Ophthalmol Vis Sci 2003; 44: 1969–1976.
Acknowledgements
This work was supported by Hungarian national grant for medical research No. ETT 011/2003 (Dr Holló). We have no financial interest in any instrument or technique used in the study. We thank Peter Vargha for performing the statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Katsanos, A., Kóthy, P., Papp, A. et al. Influence of subfoveal choroidal neovascularisation on macular imaging with scanning laser polarimetry of the retinal nerve fibre layer. Eye 19, 117–122 (2005). https://doi.org/10.1038/sj.eye.6701431
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.eye.6701431