Abstract
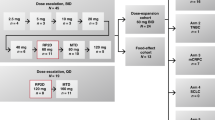
A clinical study of nolatrexed dihydrochloride (AG337, Thymitaq™) in combination with paclitaxel was performed. The aims were to optimize the schedule of administration and determine any pharmacokinetic (PK) interactions between the two drugs. In vitro combination studies were performed to assist with schedule optimization. Three patients were entered on each of three different schedules of administration of the two drugs: (1) paclitaxel 0–3 h, nolatrexed 24–144 h; (2) nolatrexed 0–120 h, paclitaxel 48–51 h; (3) nolatrexed 0–120 h, paclitaxel 126–129 h. Paclitaxel was administered at a dose of 80 mg m–2over 3 h and nolatrexed at a dose of 500 mg m–2day–1as a 120-h continuous intravenous infusion. Plasma concentrations of both drugs were determined by high performance liquid chromatography. In vitro growth inhibition studies using corresponding schedules were performed using two head and neck cancer cell lines. In both HNX14C and HNX22B cell lines, synergistic growth inhibition was observed on schedule 2, whereas schedules 1 and 3 demonstrated antagonistic effects. In the clinical study, there was no effect of schedule on the pharmacokinetics of nolatrexed. However, patients on schedules 1 and 3 had a higher clearance of paclitaxel (322–520 ml min–1m–2) than those on schedule 2 (165–238 ml min–1m–2). Peak plasma concentrations (1.66–1.93 vs 0.86–1.32 μM) and areas under the curve (392–565 vs 180–291 μM min–1) of paclitaxel were correspondingly higher on schedule 2. The pharmacokinetic interaction was confirmed by studies with human liver microsomes, nolatrexed being an inhibitor of the major routes of metabolism of paclitaxel. Toxicity was not schedule-dependent. Nolatrexed and paclitaxel may be safely given together when administered sequentially at the doses used in this study. Studies in vitro suggest some synergy, however, due to a pharmacokinetic interaction, paclitaxel doses should be reduced when administered during nolatrexed infusion. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Aisner J and Cortes-Funes H (1997) Paclitaxel in head and neck and other cancers: future prospects. Semin Oncol 24: 113–115
Belani CP, Agarwala S, Johnson J, Cohn A, Bernstein J, Langer C, Jones V, White C, Loh K, Whiter D, Chew T, Johnston A and Clendeninn N (1997a) A phase II trial with Thymitaq™ (AG337) in patients with squamous cell carcinoma of the head and neck. Proc ASCO 16: 387a
Belani CP, Lembersky B, Ramanathan R, Cohn A, Loh K, Miller W, Green M, Chew T, Johnston A and Clendeninn N (1997b) A phase II trial of Thymitaq™ (AG337) in patients with adenocarcinoma of the colon. Proc ASCO 16: 272a
Berg SL, Tolcher A, O’Shaughnessy JA, Denicoll AM, Noone M, Ognibene FP, Cowan KH and Balis FM (1995) Effect of R-verapamil on the pharmacokinetics of paclitaxel in women with breast cancer. J Clin Oncol 13: 2039–2042
Chao Y, Chan WK, Birkhofer MJ, Hu OJ, Wang SS, Huang YS, Liu M, Whang-Peng J, Chi KH, Lui WY and Lee SD (1998) Phase II and pharmacokinetic study of paclitaxel therapy for unresectable hepatocellular carcinoma patients. Br J Cancer 78: 34–39
Chou TC and Talalay P (1984) Quantitative analysis of dose–effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Reg 22: 27–55
Cresteil T, Monsarrat B, Alvinerie P, Treluyer JM, Vieira I and Wright M (1994) Taxol metabolism by human liver microsomes: identification of cytochrome P450 isozymes involved in its biotransformation. Cancer Res 54: 386–393
D’Andrea GM and Seidman AD (1997) Docetaxel and paclitaxel in breast cancer therapy: present status and future prospects. Semin Oncol 24: S1327–1344 (abstract 1962)
Geoffroy F, Patel M, Ren Q-F and Grem J (1994) Interaction of fluorouracil and paclitaxel in MCF-7 human breast cancer. Proc AACR 35: 330
Gianni L, Kearns C, Giani A, Capri G, Vigano L, Locatelli A, Bonadonna G and Egorin M (1995) Non-linear pharmacokinetic and metabolism of paclitaxel and its pharmacokinetic/pharmacodynamic relationships in humans. J Clin Oncol 13: 180–190
Gianni L, Vigano L, Locatelli A, Capri G, Giani A, Tarenzi E and Bonadonna G (1997) Human pharmacokinetic characterization and in vitro study of the interaction between doxorubicin and paclitaxel in patients with breast cancer. J Clin Oncol 15: 1906–1915
Goldspiel BR (1997) Clinical overview of the taxanes. Pharmacotherapy 17: 110–125
Harris JW, Rahman A, Kim B-R, Guengerich FP and Collins JM (1994) Metabolism of Taxol by human hepatic microsomes and liver slices: Participation of cytochrome P450 3A4 and an unknown P450 enzyme. Cancer Res 54: 4026–4035
Huizing MT, Keung ACF, Rosing H, van der Kuij V, ten Bokkel-Huinink WW, Mandjes IM, Dubbelman ARC, Pinedo HM and Beijnen JH (1993) Pharmacokinetics of paclitaxel and metabolism in a randomised comparative study in platinum-pretreated ovarian cancer patients. J Clin Oncol 11: 2127–2135
Jamis-Dow CA, Klecker RW, Katki AG and Collins JM (1995) Metabolism of taxol by human and rat liver in vivo: A screen for drug interactions and interspecies differences. Cancer Chemother Pharmacol 36: 107–114
Jamis-Dow CA, Pearl ML, Watkins PB, Blake DS, Klecker RW and Collins JM (1997) Predicting drug interactions in vitro from experiments in vitro – Human studies with paclitaxel and ketoconazole. Am J Clin Oncol – Cancer Clin Trials 20: 592–599
Kano Y, Akutsu M, Tsunoda S, Mori K, Suzuki K and Adachi KI (1998) In vitro schedule-dependent interaction between paclitaxel and SN-38 (the active metabolite of irinotecan) in human carcinoma cell lines. Cancer Chemother Pharmacol 42: 91–98
Kearns CM (1997) Pharmacokinetics of the taxanes. Pharmacotherapy 17: 105S–109S
Kennedy MJ, Zahurak ML, Donehower RC, Noe D, Grochow LB, Sartorius S, Chen T-L, Bowling K, Duerr M and Rowinsky EK (1998) Sequence-dependent hematological toxicity associated with the 3-hour paclitaxel/cyclophosphamide doublet. Clin Cancer Res 4: 349–356
Keung ACF, Kaul S, Pinedo HM, ten Bokkel Huinink WW and Beijnen JH (1993) Pharmacokinetics of taxol given by 3-h or 24-h infusion to patients with ovarian carcinoma. Proc ASCO 12: 130 (abstract 321)
Kuhn J, Rizzo J, Chang S, Schold C, Spence A, Berger M, Robins H, Mehta M, Bozik M, Fulton D, Rector D and Prados M (1997) Effects of anticonvulsants on the pharmacokinetics and metabolic profile of paclitaxel. Proc ASCO 16: 224 (abstract 787)
Lillibridge JH, Liang BH, Kerr BM, Webber S, Quart B, Shetty BV and Lee CA (1998) Characterization of the selectivity and mechanism of human cytochrome P450 inhibition by the human immunodeficiency virus-protease inhibitor nelfinavir mesylate. Drug Metab Disp 26: 609–161
Loh K, Stuart K, Cohn A, White C, Hines J, Miller W, Green MR, Chew T, Johnston A and Clendeninn N (1997) A Phase II trial of Thymitaq™ (AG337) in patients with adenocarcinoma of the pancreas. Proc ASCO 16: 265a
Panday VR, Huizing MT, Willemse PH, De Graeff A, ten Bokkol Huinink WW, Vermorken JB and Beijnen JH (1997) Hepatic metabolism of paclitaxel and its impact in patients with altered hepatic function. Semin Oncol 24: S11–S38
Rafi I, Taylor GA, Calvete JA, Boddy AV, Balmanno K, Bailey N, Lind M, Calvert AH, Webber S, Jackson RC, Johnston AJ, Clendeninn N and Newell DR (1995) Clinical pharmacokinetic and pharmacodynamic studies with the non-classical antifolate thymidylate synthase inhibitor 3,4-dihydro-2-amino-6-methyl-4-oxo-5-(4-pyridylthio)-quinazolone dihydrochloride (AG337) given by 24-hour continuous intravenous infusion. Clin Cancer Res 1: 1275–1284
Rafi I, Boddy AV, Calvete JA, Taylor GA, Newell DR, Bailey NP, Lind MJ, Green M, Hines J, Johnstone A, Clendeninn N and Calvert AH (1998) Preclinical and phase I clinical studies with the nonclassical antifolate thymidylate synthase inhibitor nolatrexed dihydrochloride given by prolonged administration in patients with solid tumors. J Clin Oncol 16: 1131–1141
Rowinsky EK (1997) Paclitaxel: dosing and schedule issues. Oncology 11: 7–19
Rowinsky EK, Gilbert MR, McGuire WP, Noe DA, Grochow LB, Forastiere AA, Ettinger DS, Lubejko BG, Clark B, Sartorius SE, Cornblath DR, Hendricks CB and Donehower RC (1991) Sequences of taxol and cisplatin: A phase I and pharmacologic study. J Clin Oncol 9: 1692–1703
Schlichenmyer WJ, Donehower RC, Chen TL, Bowling MK, McQuire WP and Rowinsky EK (1995) Pre-treatment H2 receptor antagonists that differ in P450 modulation activity: comparative effects on paclitaxel clearance rates and neutropenia. Cancer Chemother Pharmacol 36: 227–232
Siddiqui N, Boddy AV, Thomas HD, Bailey NP, Robson L, Lind MJ and Calvert AH (1997) A clinical and pharmacokinetic study of the combination of carboplatin and paclitaxel for epithelial ovarian cancer. Br J Cancer 75: 287–294
Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Vistica D, Warren JY, Bokesch H, Kenney S and Boyd MR (1990) New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst 82: 1107–1112
Sonnichsen D and Relling M (1994) Clinical pharmacokinetics of paclitaxel. Clin Pharmacokin 27: 256–269
Sonnichsen DS, Liu Q, Schuetz EG, Schuetz JD, Pappo A and Relling MV (1995) Variability in human cytochrome P450 paclitaxel metabolism. J Pharmacol Exp Ther 275: 566–575
Stuart KE, Hajdenberg J, Cohn A, Loh KK, Miller W, White C and Clendeninn NJ (1996) A Phase II trial of Thymitaq™ (AG337) in patients with hepatocellular carcinoma. Proc. ASCO 15: 202
Vanhoefer U, Harstrick A, Wilke H, Schleucher N, Walles H, Schroder J and Seeber S (1995) Schedule-dependent antagonism of paclitaxel and cisplatin in human gastric and ovarian carcinoma cell lines in vitro. Eur J Cancer 31A: 92–97
Venook AP, Egorin MJ, Rosner GL, Brown TD, Jahan TM, Batist G, Hohl R, Budman D, Ratain MJ, Kearns CM and Schilsky RL (1998) Phase I and pharmacokinetics trial of paclitaxel in patients with hepatic dysfunction. J Clin Oncol 16: 1811–1819
Vigano L, Lacatelli A, Bonadonna G and Egorin MJ (1995) Nonlinear pharmacokinetics and metabolism of paclitaxel and its pharmacokinetic/ pharmacodynamic relationships in humans. J Clin Oncol 13: 180–190
Vokes EE, Haraf DJ, Stenson K, Stupp R, Malone D, Levin J and Weichelsbaum RR (1995) The role of paclitaxel in the treatment of head and neck cancer. Semin Oncol 22 S12: 8–12
Webber SE, Bleckman TM, Attard J, Deal JG, Kathardekar V, Welsh KM, Webber S, Janson CA, Matthews DA, Smith WW, Freer ST, Jordan SR, Bacquet RJ, Howland EF, Booth CLJ, Ward RW, Hermann SM, White J, Morse CA, Hilliard JA and Bartlett CA (1993) Design of thymidylate synthase inhibitors using protein crystal-structures – the synthesis and biological evaluation of a novel class of 5-substituted quinazolinones. J Med Chem 36: 733–746
Wilson WH, Berg SL, Bryant G, Wittes RE, Bates S, Fojo A, Steinberg SM, Goldspiel BR, Herdt J and O’Shaughnessy J (1994) Paclitaxel in doxorubicin-refractory or mitoxantrone-refractory breast cancer: a phase I/II trial of 96-hour infusion. J Clin Oncol 12: 1621–1629
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Hughes, A., Griffin, M., Newell, D. et al. Clinical pharmacokinetic and in vitro combination studies of nolatrexed dihydrochloride (AG337, Thymitaq™) and paclitaxel. Br J Cancer 82, 1519–1527 (2000). https://doi.org/10.1054/bjoc.2000.1172
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1172