Abstract
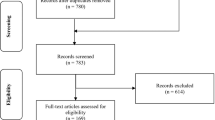
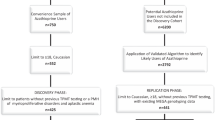
One of the major goals of pharmacogenetics is to elucidate mechanisms and identify patients at increased risk of adverse events (AEs). To date, however, there have been only a few successful examples of this type of approach. In this paper, we describe a retrospective case–control pharmacogenetic study of an AE of unknown mechanism, characterized by elevated levels of serum alanine aminotransferase (ALAT) during long-term treatment with the oral direct thrombin inhibitor ximelagatran. The study was based on 74 cases and 130 treated controls and included both a genome-wide tag single nucleotide polymorphism and large-scale candidate gene analysis. A strong genetic association between elevated ALAT and the MHC alleles DRB1*07 and DQA1*02 was discovered and replicated, suggesting a possible immune pathogenesis. Consistent with this hypothesis, immunological studies suggest that ximelagatran may have the ability to act as a contact sensitizer, and hence be able to stimulate an adaptive immune response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Abbreviations
- AE:
-
adverse event
- ALAT:
-
alanine aminotransferase
- DIP:
-
deletion/insertion polymorphism
- EXGEN:
-
An explorative pharmacogenetic case–control study, seeking explanation of ximelagatran's pharmacodynamic effects on transient hepatic enzyme elevations during long-term treatment
- DRB1:
-
the HLA class II DR molecule is a heterodimer consisting of an α (DRA) and a β chain (DRB), of which the latter contains all the polymorphisms specifying the peptide binding specificities. The so-called low-resolution type or two-number code (DRB1*01, and so on) corresponds to all the alleles that encode the DR antigen. The four-number codes (HLA-DRB1*0101, 0102, 0103, and so on) refer to specific DRB1*01 alleles
- FDR:
-
false discovery rate
- GPMT:
-
guinea pig maximization test
- GWS:
-
genome-wide scan based on haplotype tagging SNPs
- HLA:
-
human leucocyte antigen. The human MHC
- LD:
-
linkage disequilibrium
- LTT:
-
lymphocyte transformation test
- NCBI:
-
National Center for Biotechnology Information
- MHC:
-
major histocompatibility complex: set of genes coding for proteins involved in antigen presentation
- PCR:
-
polymerase chain reaction
- qPCR:
-
quantitative PCR
- SNP:
-
single nucleotide polymorphism
- ULN:
-
upper limit of normal
References
Lee WM, Larrey D, Olsson R, Lewis JH, Keisu M, Auclert L et al. Hepatic findings in long-term clinical trials of ximelagatran. Drug Safety 2005; 28: 351–370.
Harenberg J, Jörg I, Weiss C . Observations of alanine aminotransferase and aspartate aminotransferase in THRIVE studies treated orally with ximelagatran. Int J Toxicol 2006; 25: 165–169.
AstraZeneca Decides to Withdraw Exanta™ http://www.astrazeneca.com/pressrelease/5217.aspx.
Hinds DA, Stuve LL, Nilsen GB, Halperin E, Eskin E, Ballinger DG et al. Whole-genome patterns of common DNA variation in three human populations. Science 2005; 307: 1072–1079.
Storey JD . A direct approach to false discovery rates. J R Stat Soc B 2001; 64: 479–498.
Yunis EJ, Larsen CE, Fernandez-Vina M, Awdeh ZL, Romero T, Hansen JA et al. Inheritable variable sizes of DNA stretches in the human MHC: conserved extended haplotypes and their fragments or blocks. Tissue Antigens 2003; 62: 1–20.
Miretti MM, Walsh EC, Ke X, Delgado M, Griffiths M, Hunt S et al. A high-resolution linkage-disequilibrium map of the human major histocompatibility complex and first generation of tag single-nucleotide polymorphisms. Am J Hum Genet 2005; 76: 634–646.
Maria VAJ, Victorino RMM . Diagnostic value of specific T-cell reactivity to drugs in 95 cases of drug induced liver injury. Gut 1997; 41: 534–540.
De Groot AS . Immunomics: discovering new targets for vaccines and therapeutics. Drug Discov Today 2006; 11: 203–209.
Gibert M, Sanchez-Mazas A . Geographic patterns of functional categories of HLA-DRB1 alleles: a new approach to analyse associations between HLA-DRB1 and disease. Eur J Immunogenet 2003; 30: 361–374.
Berlin M, Fogdell-Hahn A, Olerup O, Eklund A, Grunewald J . HLA-DR predicts the prognosis in Scandinavian patients with pulmonary sarcoidosis. Am J Respir Crit Care Med 1997; 156: 1601–1605.
Vergani D, Mieli-Vergani G . Autoimmune hepatitis. Minerva Gastroenterol Dietol 2004; 50: 113–123.
Sharma SK, Balamurugan A, Saha PK, Pandey RM, Mehra NK . Evaluation of clinical and immunogenetic risk factors for the development of hepatotoxicity during antituberculosis treatment. Am J Respir Crit Care Med 2002; 166: 916–919.
Holt MP, Ju C . Mechanisms of drug-induced liver injury. AAPS J 2006; 8: E48–E54.
Castell JV . Allergic hepatitis: a drug-mediated organ-specific immune reaction. Clin Exp Allergy 1998; 28 (Suppl 4): 13–19.
Pichler WJ . Pharmacological interaction of drugs with antigen-specific immune receptors: the p-i concept. Curr Opin Allergy Clin Immunol 2002; 2: 301–305.
Gerber BO, Pichler WJ . Noncovalent interactions of drugs with immune receptors may mediate drug-induced hypersensitivity reactions. AAPS J 2006; 8: E160–E165.
Britschgi M, von Greyerz S, Burkhart C, Pichler WJ . Molecular aspects of drug recognition by specific T cells. Curr Drug Targets 2003; 4: 1–11.
Innocenti F, Undevia SD, Iyer L, Chen PX, Das S, Kocherginsky M et al. Genetic variants in the UDP-glucuronosyltransferase 1A1 gene predict the risk of severe neutropenia of irinotecan. J Clin Oncol 2004; 22: 1382–1388.
Sconce EA, Khan TI, Wynne HA, Avery P, Monkhouse L, King BP et al. The impact of CYP2C9 and VKORC1 genetic polymorphism and patient characteristics upon warfarin dose requirements: proposal for a new dosing regimen. Blood 2005; 106: 2329–2333.
Rauch A, Nolan D, Martin A, McKinnon E, Almeida C, Mallal S . Prospective genetic screening decreases the incidence of abacavir hypersensitivity reactions in the Western Australian HIV cohort study. Clin Infect Dis 2006; 43: 99–102.
Pirmohamed M, Park BK . Genetic susceptibility to adverse drug reactions. Trends Pharmacol Sci 2001; 22: 298–305.
March R . Challenges and opportunities of pharmacogenetics in drug development. Pers Med 2006; 3: 195–206.
Carlson CS, Eberle MA, Rieder MJ, Yi Q, Kruglyak L, Nickerson DA . Selecting a maximally informative set of single-nucleotide polymorphisms for association analyses using linkage disequilibrium. Am J Hum Genet 2004; 74: 106–120.
Gabriel SB, Schaffner SF, Nguyen H, Moore JM, Roy J, Blumenstiel B et al. The structure of haplotype blocks in the human genome. Science 2002; 296: 2225–2229.
Traherne JA, Horton R, Roberts AN, Miretti MM, Hurles ME, Stewart CA et al. Genetic analysis of completely sequenced disease-associated MHC haplotypes identifies shuffling of segments in recent human history. PLoS Genet 2006; J2: e9.
Gual L, Martinez A, Fernandez-Arquero M, Garcia-Rodriguez MC, Ferreira A, Fontan G et al. Major histocompatibility complex haplotypes in Spanish immunoglobulin A deficiency patients: a comparative fine mapping microsatellite study. Tissue Antigens 2004; 64: 671–677.
Sham PC, Curtis D . Monte Carlo tests for associations between disease and alleles at highly polymorphic loci. Ann Hum Genet 1995; 59: 97–105.
Devlin B, Bacanu S-A, Roeder K . Genomic control to the extreme. Nat Genet 2004; 36: 1129–1130.
Pritchard JK, Stephens M, Donnely P . Inference of population structure using multilocus genotype data. Genetics 2000; 155: 945–959.
Nyfeler B, Pichler WJ . The lymphocyte transformation test for the diagnosis of drug allergy: sensitivity and specificity. Clin Exp Allergy 1997; 27: 175–181.
Acknowledgements
We thank all the subjects who donated genetic samples as part of EXGEN and the LTT study. We also thank everyone from CVGI Genetics in Mölndal, Safety Assessment in Södertälje and R&D Genetics and Statistical Science in Alderley Park who contributed to this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Duality of Interest
Professor Lon Cardon acts as a consultant to AstraZeneca.
Supplementary Information accompanies the paper on the The Pharmacogenomics Journal website (http://www.nature.com/tpj)
Supplementary information
Rights and permissions
About this article
Cite this article
Kindmark, A., Jawaid, A., Harbron, C. et al. Genome-wide pharmacogenetic investigation of a hepatic adverse event without clinical signs of immunopathology suggests an underlying immune pathogenesis. Pharmacogenomics J 8, 186–195 (2008). https://doi.org/10.1038/sj.tpj.6500458
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.tpj.6500458
Keywords
This article is cited by
-
Being precise with anticoagulation to reduce adverse drug reactions: are we there yet?
The Pharmacogenomics Journal (2024)
-
Genome-wide association study of abnormal elevation of ALT in patients exposed to atabecestat
BMC Genomics (2023)
-
Managing the challenge of drug-induced liver injury: a roadmap for the development and deployment of preclinical predictive models
Nature Reviews Drug Discovery (2020)
-
High-throughput confocal imaging of differentiated 3D liver-like spheroid cellular stress response reporters for identification of drug-induced liver injury liability
Archives of Toxicology (2019)
-
Characterisation of the HLA-DRB1*07:01 biomarker for lapatinib-induced liver toxicity during treatment of early-stage breast cancer patients with lapatinib in combination with trastuzumab and/or taxanes
The Pharmacogenomics Journal (2018)