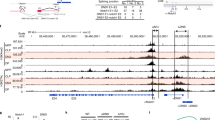
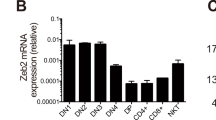
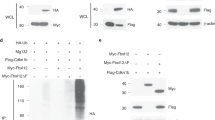
Abstract
Proper development of the thymus and differentiation of T-lymphocytes requires cell–cell interactions between the developing T-lymphocytes and the thymic epithelia. The Delta/Serrate/Lag-2 (DSL)/Notch signal-transduction pathway is known to govern cell fate decisions required for proper development through direct cell–cell interactions. The functional consequences of specific DSL/Notch interactions during the development of a complex organ, such as the thymus, have not been thoroughly elucidated, however. In order to examine the role of DSL proteins during thymus development and T-lymphocyte differentiation, we targeted expression of JAGGED1 in T-lymphocyte progenitors via the control of the proximal lck promoter. Here, we report that expression of JAGGED1 in T cells causes premature involution of the thymus by directing thymic epithelial cells to undergo an apoptotic program. Adoptive transfer of JAGGED1 transgenic bone marrow into non-transgenic mice revealed that JAGGED1 expression on T cells does not alter T-cell differentiation, but is directly responsible for involution of the thymus. We propose that the phenotype of the lck-JAGGED1 transgenic mice is a direct result of specific DSL/Notch interactions and improper cell-to-cell signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Brou C, Logeat F, Gupta N, Bessia C, LeBail O, Doedens JR et al. A novel proteolytic cleavage involved in Notch signaling: the role of the disintegrin-metalloprotease TACE. Mol Cell 2000; 5: 207–216.
Mumm JS, Kopan R . Notch signaling: from the outside in. Dev Biol 2000; 228: 151–165.
Schroeter EH, Kisslinger JA, Kopan R . Notch-1 signalling requires ligand-induced proteolytic release of intracellular domain [see comments]. Nature 1998; 393: 382–386.
Shimizu K, Chiba S, Hosoya N, Kumano K, Saito T, Kurokawa M et al. Binding of Delta1, Jagged1, and Jagged2 to Notch2 rapidly induces cleavage, nuclear translocation, and hyperphosphorylation of Notch2. Mol Cell Biol 2000; 20: 6913–6922.
Allman D, Punt JA, Izon DJ, Aster JC, Pear WS . An invitation to T and more: notch signaling in lymphopoiesis. Cell 2002; 109 (Suppl): S1–S11.
Artavanis-Tsakonas S, Rand MD, Lake RJ . Notch signaling: cell fate control and signal integration in development. Science 1999; 284: 770–776.
Miele L, Osborne B . Arbiter of differentiation and death: Notch signaling meets apoptosis. J Cell Physiol 1999; 181: 393–409.
Milner LA, Bigas A . Notch as a mediator of cell fate determination in hematopoiesis: evidence and speculation. Blood 1999; 93: 2431–2448.
Ohishi K, Katayama N, Shiku H, Varnum-Finney B, Bernstein ID . Notch signalling in hematopoiesis. Semin Cell Dev Biol 2003; 14: 143–150.
Osborne B, Miele L . Notch and the immune system. Immunity 1999; 11: 653–663.
Felli MP, Maroder M, Mitsiadis TA, Campese AF, Bellavia D, Vacca A et al. Expression pattern of notch1, 2 and 3 and Jagged1 and 2 in lymphoid and stromal thymus components: distinct ligand–receptor interactions in intrathymic T cell development. Int Immunol 1999; 11: 1017–1025.
Hoyne GF, Le Roux I, Corsin-Jimenez M, Tan K, Dunne J, Forsyth LM et al. Serrate1-induced notch signalling regulates the decision between immunity and tolerance made by peripheral CD4(+) T cells. Int Immunol 2000; 12: 177–185.
Jimenez E, Vicente A, Sacedon R, Munoz JJ, Weinmaster G, Zapata AG et al. Distinct mechanisms contribute to generate and change the CD4:CD8 cell ratio during thymus development: a role for the Notch ligand, Jagged1. J Immunol 2001; 166: 5898–5908.
Kaneta M, Osawa M, Sudo K, Nakauchi H, Farr AG, Takahama Y . A role for pref-1 and HES-1 in thymocyte development. J Immunol 2000; 164: 256–264.
Shawber C, Boulter J, Lindsell CE, Weinmaster G . Jagged2: a serrate-like gene expressed during rat embryogenesis. Dev Biol 1996; 180: 370–376.
Anderson G, Jenkinson EJ . Lymphostromal interactions in thymic development and function. Nat Rev Immunol 2001; 1: 31–40.
van Ewijk W, Hollander G, Terhorst C, Wang B . Stepwise development of thymic microenvironments in vivo is regulated by thymocyte subsets. Development 2000; 127: 1583–1591.
Germain RN . T-cell development and the CD4–CD8 lineage decision. Nat Rev Immunol 2002; 2: 309–322.
Bellavia D, Campese AF, Alesse E, Vacca A, Felli MP, Balestri A et al. Constitutive activation of NF-kappaB and T-cell leukemia/lymphoma in Notch3 transgenic mice. EMBO J 2000; 19: 3337–3348.
Robey E, Chang D, Itano A, Cado D, Alexander H, Lans D et al. An activated form of Notch influences the choice between CD4 and CD8T cell lineages. Cell 1996; 87: 483–492.
Deftos ML, Huang E, Ojala EW, Forbush KA, Bevan MJ . Notch1 signaling promotes the maturation of CD4 and CD8 SP thymocytes. Immunity 2000; 13: 73–84.
Fowlkes BJ, Robey EA . A reassessment of the effect of activated Notch1 on CD4 and CD8T cell development. J Immunol 2002; 169: 1817–1821.
Izon DJ, Punt JA, Xu L, Karnell FG, Allman D, Myung PS et al. Notch1 regulates maturation of CD4+ and CD8+ thymocytes by modulating TCR signal strength. Immunity 2001; 14: 253–264.
Valdez P, Robey E . Notch and the CD4 versus CD8 lineage decision. Cold Spring Harb Symp Quant Biol 1999; 64: 27–31.
Wolfer A, Bakker T, Wilson A, Nicolas M, Ioannidis V, Littman DR et al. Inactivation of Notch 1 in immature thymocytes does not perturb CD4 or CD8T cell development. Nat Immunol 2001; 2: 235–241.
Radtke F, Wilson A, Stark G, Bauer M, van Meerwijk J, MacDonald HR et al. Deficient T cell fate specification in mice with an induced inactivation of Notch1. Immunity 1999; 10: 547–558.
Wilson A, MacDonald HR, Radtke F . Notch 1-deficient common lymphoid precursors adopt a B cell fate in the thymus. J Exp Med 2001; 194: 1003–1012.
Koch U, Lacombe TA, Holland D, Bowman JL, Cohen BL, Egan SE et al. Subversion of the T/B lineage decision in the thymus by lunatic fringe-mediated inhibition of Notch-1. Immunity 2001; 15: 225–236.
Jaleco AC, Neves H, Hooijberg E, Gameiro P, Clode N, Haury M et al. Differential effects of Notch ligands Delta-1 and Jagged-1 in human lymphoid differentiation. J Exp Med 2001; 194: 991–1002.
Karanu FN, Murdoch B, Gallacher L, Wu DM, Koremoto M, Sakano S et al. The notch ligand jagged-1 represents a novel growth factor of human hematopoietic stem cells. J Exp Med 2000; 192: 1365–1372.
Ohishi K, Varnum-Finney B, Bernstein ID . Delta-1 enhances marrow and thymus repopulating ability of human CD34(+)CD38(−) cord blood cells. J Clin Invest 2002; 110: 1165–1174.
Gray GE, Mann RS, Mitsiadis E, Henrique D, Carcangiu ML, Banks A et al. Human ligands of the Notch receptor. Am J Pathol 1999; 154: 785–794.
Chaffin KE, Beals CR, Wilkie TM, Forbush KA, Simon MI, Perlmutter RM . Dissection of thymocyte signaling pathways by in vivo expression of pertussis toxin ADP-ribosyltransferase. EMBO J 1990; 9: 3821–3829.
He Y, Pear WS . Notch signalling in B cells. Semin Cell Dev Biol 2003; 14: 135–142.
Hernandez-Hoyos G, Alberola-Ila J . A Notch so simple influence on T cell development. Semin Cell Dev Biol 2003; 14: 121–125.
Mackall CL, Gress RE . Thymic aging and T-cell regeneration. Immunol Rev 1997; 160: 91–102.
Izon DJ, Aster JC, He Y, Weng A, Karnell FG, Patriub V et al. Deltex1 redirects lymphoid progenitors to the B cell lineage by antagonizing Notch1. Immunity 2002; 16: 231–243.
Beverly LJ, Capobianco AJ . Perturbation of Ikaros isoform selection by MLV integration is a cooperative event in Notch(IC)-induced T cell leukemogenesis. Cancer Cell 2003; 3: 551–564.
Dorsch M, Zheng G, Yowe D, Rao P, Wang Y, Shen Q et al. Ectopic expression of Delta4 impairs hematopoietic development and leads to lymphoproliferative disease. Blood 2002; 100: 2046–2055.
Yan XQ, Sarmiento U, Sun Y, Huang G, Guo J, Juan T et al. A novel Notch ligand, Dll4, induces T-cell leukemia/lymphoma when overexpressed in mice by retroviral-mediated gene transfer. Blood 2001; 98: 3793–3799.
Ascano JM, Beverly LJ, Capobianco AJ . The C-terminal PDZ-ligand of JAGGED1 is essential for cellular transformation. J Biol Chem 2003; 278: 8771–8779.
Wildin RS, Garvin AM, Pawar S, Lewis DB, Abraham KM, Forbush KA et al. Developmental regulation of lck gene expression in T lymphocytes. J Exp Med 1991; 173: 383–393.
Acknowledgements
We thank members of the Capobianco laboratory for support and technical assistance. We are grateful to S Artavanis-Tsakonas for kindly providing reagents used in this study. We thank the members of the Wistar Institute histology and flow cytometry cores. This work was funded in part by grants from the American Cancer Society (RPG LBC-99465 to A J C), the National Cancer Institute (ROI CA 83736 to A J C), the Leukemia and Lymphoma Society (LLS 1298 to A J C) and the NCI National Institutes of Health Training Grant 5T32 CA59268 (to J M A). L J B is a graduate student at the University of Cincinnati in the Department of Molecular Genetics, Biochemisty and Microbiology.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Genes and Immunity website (http://www.nature.com/gene).
Supplementary information
Rights and permissions
About this article
Cite this article
Beverly, L., Ascano, J. & Capobianco, A. Expression of JAGGED1 in T-lymphocytes results in thymic involution by inducing apoptosis of thymic stromal epithelial cells. Genes Immun 7, 476–486 (2006). https://doi.org/10.1038/sj.gene.6364318
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gene.6364318