Insight into risks posed by corrective gene therapy comes from an immunodeficient mouse model.
Abstract
The development of T-cell leukaemia following the otherwise successful treatment of three patients with X-linked severe combined immune deficiency (X-SCID) in gene-therapy trials using haematopoietic stem cells1 has led to a re-evaluation of this approach2. Using a mouse model for gene therapy of X-SCID, we find that the corrective therapeutic gene IL2RG itself can act as a contributor to the genesis of T-cell lymphomas, with one-third of animals being affected. Gene-therapy trials for X-SCID, which have been based on the assumption that IL2RG is minimally oncogenic3,4,5,6,7, may therefore pose some risk to patients.
Similar content being viewed by others
Main
The syndrome X-SCID is caused by faulty expression of the γ-chain of the interleukin-2 receptor (IL2RG), which results in diminished lymphoid-cell survival and proliferation. Gene therapy can restore IL2RG expression and hence adaptive immunity in X-SCID patients. The development of leukaemia after this gene therapy has been attributed to the upregulated expression of the oncogene LMO2 as a result of vector integration2.
To investigate the origin of these adverse events, we expressed IL2RG inserted into a lentiviral vector (LV) in a murine model of X-SCID, and followed the fates of mice for up to 1.5 years post-transplantation. Unexpectedly, 33% of these mice (n=15) developed T-cell lymphomas that were associated with a gross thymic mass (Fig. 1, and see supplementary information). Lymphomic tissues shared a common lymphomic stem cell, with similar vector-integration sites being evident in the DNA of the thymus, bone marrow and spleen of individual mice; however, no common integration targets were found between mice.
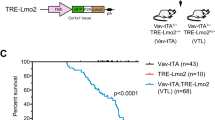
Significantly more T-cell lymphomas developed in animals transplanted with cells transduced with LV-IL2RG (triangles) compared with controls that were mock-transduced or LV-GFP vector-transduced (filled circles and open circles, respectively). LV-GFP expresses the marker gene for green fluorescence protein and is used to verify that the principal lymphomagenic event is neither insertional mutagenesis nor the presence of oncogenic elements within the vector backbone. Crosses indicate positive controls transplanted with LV-LMO2 containing the T-cell oncogene LMO2. We found 33% of LV-IL2RG mice died within 81 weeks of transplantation with either X-SCID or wild-type IL2RG (CD45.1) haematopoietic donor cells.
As expected, T-cell lymphomas were also detected in positive-control mice transduced with LMO2 in a lentiviral vector (n=12; prevalence, 50%; the cell phenotype has been described8), although none of the mock-transduced mice (n=15) or control-vector-transduced mice (LV-GFP; n=15; Fig. 1) developed thymomas. These results, generated from independent transplant experiments with 76 mice ranging from 41 to 81 weeks post-transplant, indicate that insertional mutagenesis was not the principal cause of lymphoma in our mice.
How has the oncogenicity of IL2RG been overlooked? Worldwide, some 88 mice have been treated with IL2RG in retroviral vectors3,4,5,9,10,11, but these studies were limited by their duration, which usually did not exceed 6 months post-transplant. (Our first IL2RG-induced lymphoma appeared 6 months post-transplant.) Longer-term analysis did not reveal leukaemogenesis in dogs, rhesus macaques or sheep carrying human chimaeric genes for up to one year post-transplant, but this may have been attributable to features inherent in large-animal models6,7. In the human gene-therapy trials1, leukaemias did not appear until 2–3 years after treatment.
Lymphomagenesis in our mice may result from altered signalling through one or more of the interleukin receptors that contain the IL2RG subunit (including those for interleukins 2, 4, 7, 9, 15 and 21, most of which have been associated with T-cell neoplasias). Altered cell-survival and/or tumour-suppressor functions due to abnormal receptor expression are known to occur in B-cell-receptor signalling, where overexpression of Btk blocks B-cell development but underexpression leads to B-cell neoplasia12. The long latency period before lymphomas develop in our LV-IL2RG-treated mice indicates that other complementary mutations may be required for lymphomagenesis.
Our results indicate that preclinical experimental treatments involving transgenes should include long-term follow-up before they enter clinical trials. Moreover, our findings highlight the need for continued development of vectors capable of regulated therapeutic gene expression.
References
Hacein-Bey-Abina, S. et al. N. Engl. J. Med. 346, 1185–1193 (2002).
Hacein-Bey-Abina, S. et al. Science 302, 415–419 (2003).
Soudais, C. et al. Blood 95, 3071–3077 (2000).
Otsu, M., Sugamura, K. & Candotti, F. Blood 97, 1618–1624 (2001).
Aviles Mendoza, G. J. et al. Mol. Ther. 3, 565–573 (2001).
Whitwam, T. et al. Blood 92, 1565–1575 (1998).
An, D. S. et al. J. Virol. 75, 3547–3555 (2001).
Larson, R. C., Osada, H., Larson, T. A., Lavenir, I. & Rabbitts, T. H. Oncogene 11, 853–862 (1995).
Lo, M. et al. Blood 94, 3027–3036 (1999).
Otsu, M. et al. Mol. Ther. 1, 145–153 (2000).
Otsu, M., Sugamura, K. & Candotti, F. Hum. Gene Ther. 11, 2051–2056 (2000).
Kersseboom, R. et al. J. Exp. Med. 198, 91–98 (2003).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
(DOC 33 kb)
Rights and permissions
About this article
Cite this article
Woods, NB., Bottero, V., Schmidt, M. et al. Therapeutic gene causing lymphoma. Nature 440, 1123 (2006). https://doi.org/10.1038/4401123a
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/4401123a
This article is cited by
-
Self-assembled Viral Nanoparticles as Targeted Anticancer Vehicles
Biotechnology and Bioprocess Engineering (2021)
-
Gene correction for SCID-X1 in long-term hematopoietic stem cells
Nature Communications (2019)
-
CRISPR/Cas9-modified hematopoietic stem cells—present and future perspectives for stem cell transplantation
Bone Marrow Transplantation (2019)
-
Gene correction of HBB mutations in CD34+ hematopoietic stem cells using Cas9 mRNA and ssODN donors
Molecular and Cellular Pediatrics (2018)
-
A comparison of inverted and upright laser-activated titanium nitride micropyramids for intracellular delivery
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.