Abstract
High-latitude polar deserts are among the most extreme environments on Earth. Here we describe a large and previously unappreciated habitat for photosynthetic life under opaque rocks in the Arctic and Antarctic polar deserts. This habitat is created by the periglacial movement of the rocks, which allows some light to reach their underside. The productivity of this ecosystem is at least as great as that of above-ground biomass and potentially doubles previous productivity estimates for the polar desert ecozone.
Similar content being viewed by others
Main
The underside of rocks in climatically extreme deserts acts as a refugium for photosynthetic microorganisms (defined as ‘hypoliths’) and their community (the ‘hypolithon’)1. Here, the organisms are protected from harsh ultraviolet radiation2 and wind scouring, and trapped moisture can provide them with a source of liquid water3. Colonization also usually requires the rocks to be sufficiently translucent to allow for the penetration of light — all hypoliths reported so far have been found under quartz, which is one of the most common translucent rocks4,5.
We examined 850 randomly selected opaque dolomitic rocks, without regard to the local patterns of periglacial rock sorting, on Cornwallis Island and Devon Island in the Canadian high Arctic. These are regions typical of extreme polar desert, where vegetation cover was measured to be 1.2% or less6. On Devon Island, 95% of rocks were found to be colonized, and on Cornwallis Island, 94% were colonized. The photosynthetic organisms form a well defined green band on the underside of the rocks. The mean (±s.d.) band width of these communities was 3.1 ± 1.9 cm and 3.0 ± 1.6 cm on Devon Island and Cornwallis, respectively.
The Arctic hypoliths are dominated by cyanobacteria. Species found include Gloeocapsa cf. atrata Kützing, Gloeocapsa cf. punctata Nägeli, Gloeocapsa cf. kuetzingiana Nägeli and Chroococcidiopsis-like cells; unicellular algal chlorophytes were also present7.
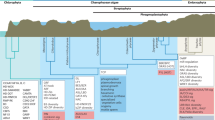
We investigated the colonization of well developed polygons, which are just one manifestation of a diversity of linear and circular features caused by the long-term action of periglacial processes8. In the Arctic, we found colonization on 68% of rocks within polygons, with a mean (±s.d.) band width of 0.7 ± 0.8 cm, where the rocks are surrounded by fine soil sorted into their centres. At the edges, where the cracks around the rocks are larger, we found 100% colonization with a mean (±s.d.) band width of 3.6 ± 1.4 cm (Fig. 1).
a, Hypolithic colonization is enhanced by rock sorting which, among other factors, increases light penetration to the underside of rocks, shown here at the edges of polygonal terrain (white lines indicate polygon edges). b, Opaque rocks are colonized on their underside by well defined bands of photosynthetic communities, seen here on an excavated rock.
We studied similar polygonal terrains at Mars Oasis on Alexander Island in the Antarctic Peninsula, a location where hypolithic colonization occurs. The percentage colonization was 5% within polygons and 100% on the edges of polygons, with mean (±s.d.) band widths of 0.7 ± 0.1 and 2.1 ± 0.3 cm, respectively. We propose that rock sorting by periglacial action, including that during freeze–thaw cycles, improves light penetration around the edges of rocks, one factor that might account for the widespread colonization of the underside of opaque rocks.
We estimated the productivity of the Arctic communities by monitoring the uptake of 14C-labelled sodium bicarbonate (for methods, see supplementary information). Assuming no carbon uptake from other sources, a conservative estimate of mean (±s.d.) productivity is about 0.8 ± 0.3 g m−2 yr−1. Given that the estimated mean (±s.d.) productivity from plants, lichens and bryo-phytes on Devon Island is 1.0 ± 0.4 g m−2 yr−1 (ref. 6), the polar hypolithon may be just as important in sequestering carbon, at least doubling previous estimates of the productivity of the rocky polar desert.
References
Golubic, S., Friedmann, I. & Schneider, J. J. Sediment. Petrol. 51, 0475–0478 (1981).
Cockell, C. S., Rettberg, P., Horneck, G., Scherer, K. & Stokes, D. M. Polar Biol. 26, 62–69 (2003).
Smith, M. C., Bowman, J. P., Scott, F. J. & Line, M. A. Antarctic Sci. 12, 177–184 (2000).
Broady, P. A. Brit. Phycol. J. 16, 231–240 (1981).
Schlesinger, W. H. et al. Ecology 84, 3222–3231 (2003).
Bliss, L. C., Svoboda, J. & Bliss, D. I. Holarctic Ecol. 7, 305–324 (1984).
Cockell, C. S. et al. Meteor. Planet. Sci. 37, 1287–1298 (2002).
Kessler, M. A. & Werner, B. T. Science 299, 380–383 (2003).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Methods
This file contains a description of the methods used to quantify the colonization of the hypolithic organisms and a description of the methods used to obtain the productivity measurements. (DOC 24 kb)
Rights and permissions
About this article
Cite this article
Cockell, C., Stokes, M. Widespread colonization by polar hypoliths. Nature 431, 414 (2004). https://doi.org/10.1038/431414a
Published:
Issue Date:
DOI: https://doi.org/10.1038/431414a
This article is cited by
-
Seasonal Variation of Hypolithic Microbiomes in the Gobi Desert
Microbial Ecology (2023)
-
Fungal diversity present on rocks from a polar desert in continental Antarctica assessed using DNA metabarcoding
Extremophiles (2021)
-
A survey of Antarctic cyanobacteria
Hydrobiologia (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.