Abstract
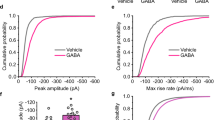
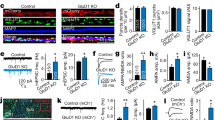
Modification of synaptic strength in the mammalian central nervous system (CNS) occurs at both pre- and postsynaptic sites1,2. However, because postsynaptic receptors are likely to be saturated by released transmitter, an increase in the number of active postsynaptic receptors may be a more efficient way of strengthening synaptic efficacy3,4,5,6,7. But there has been no evidence for a rapid recruitment of neurotransmitter receptors to the postsynaptic membrane in the CNS. Here we report that insulin causes the type A γ-aminobutyric acid (GABAA) receptor, the principal receptor that mediates synaptic inhibition in the CNS8, to translocate rapidly from the intracellular compartment to the plasma membrane in transfected HEK 293 cells, and that this relocation requires the β2 subunit of the GABAA receptor. In CNS neurons, insulin increases the expression of GABAA receptors on the postsynaptic and dendritic membranes. We found that insulin increases the number of functional postsynaptic GABAA receptors, thereby increasing the amplitude of the GABAA-receptor-mediated miniature inhibitory postsynaptic currents (mIPSCs) without altering their time course. These results provide evidence for a rapid recruitment of functional receptors to the postsynaptic plasma membrane, suggesting a fundamental mechanism for the generation of synaptic plasticity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jessell, T. M. & Kandel, E. R. Synaptic transmission: A bidirectional and self-modifiable form of cell–cell communication. Neuron 10 (suppl.), 1–10 (1993).
Bliss, T. V. P. & Collingridge, G. L. Asynaptic model of memory: Long-term potentiation in the hippocampus. Nature 361, 31–39 (1993).
Busch, C. & Sakmann, B. Synaptic transmission in hippocampal neurons: numerical reconstruction of quantal IPSCs. Cold Spring Harb. Quant. Biol. 55, 69–80 (1990).
Redman, S. Quantal analysis of synaptic potentials in neurons of the central nervous system. Physiol. Rev. 70, 165–198 (1990).
Tong, G. & Jahr, C. E. Multivesicular release from excitatory synapses of cultured hippocampal neurons. Neuron 12, 51–59 (1994).
Edwards, F. A. Anatomy and electrophysiology of fast central synapses lead to a structural model for long-term potentiation. Physiol. Rev. 75, 759–787 (1995).
Mody, I., Koninck, Y., Otis, T. S. & Soltesz, I. Bridging the cleft at GABA synapses in the brain. Trends Neurosci. 17, 517–525 (1994).
Macdonald, R. L. & Olsen, R. W. GABAAreceptor channels. Annu. Rev. Neurosci. 17, 569–602 (1994).
Hamilton, B. J. et al. Stable expression of cloned rat GABAAreceptor subunits in a human kidney cell line. Neurosci. Lett. 153, 206–209 (1993).
McKernan, R. M. & Whiting, P. J. Which GABAA-receptor subtypes really occur in the brain? Trends Neurosci. 19, 139–143 (1996).
O'Dell, T. J., Kandel, E. R. & Grant, S. G. Long-term potentiation in the hippocampus is blocked by tyrosine kinase inhibitors. Nature 353, 558–560 (1991).
Wang, Y. T. & Salter, M. W. Regulation of NMDA receptors by protein-tyrosine kinases and phosphatases. Nature 369, 233–235 (1994).
Wozniak, M., Rydzewski, B., Baker, S. P. & Raizada, M. K. The cellular and physiological actions of insulin in the central nervous system. Neurochem. Int. 22, 1–10 (1993).
Benson, D. L. & Cohen, P. A. Activity-independent segregation of excitatory and inhibitory synaptic terminals in cultured hippocampal neurons. J. Neurosci. 16, 6424–6432 (1996).
Nusser, Z., Roberts, J. D., Baude, A., Richards, J. G. & Somogyi, P. Relative densities of synaptic and extrasynaptic GABAAreceptors on cerebellar granule cells as determined by a quantitative immunogold method. J. Neurosci. 15, 2948–2960 (1995).
Thompson, S. M., Copogna, M. & Scanziani, M. Presynaptic inhibition in the hippocampus. Trends Neurosci. 16, 222–227 (1993).
Cohen, G. A., Doze, V. A. & Madison, D. V. Opioid inhibition of GABA release from presynaptic terminals of rat hippocampal interneurons. Neuron 9, 325–335 (1992).
Liao, D., Hessler, N. A. & Malinow, R. Activation of postsynaptically silent synapses during pairing-induced LTP in CA1 region of hippocampal slice. Nature 375, 400–404 (1995).
Kamatchi, G. L., Bhakthavatsalam, P., Chandra, D. & Bapna, J. S. Inhibition of insulin hyperphagia by gamma aminobutyric acid antagonists in rats. Life Sci. 34, 2297–2301 (1984).
Garry, D. J., Sorenson, R. L., Elde, R. P., Maley, B. E. & Madsen, A. Immunohistochemical colocalization of GABA and insulin in beta-cells of rat islet. Diabetes 35, 1090–1095 (1986).
Rorsman, P. et al. Glucose-inhibition of glucagon secretion involves activation of GABAA-receptor chloride channels. Nature 341, 233–236 (1989).
Otis, T. S., De Koninck, Y. & Mody, I. Lasting potentiation of inhibition is associated with an increased number of gamma-aminobutyric acid type A receptors activated during miniature inhibitory postsynaptic currents. Proc. Natl Acad. Sci. USA 91, 7698–7702 (1994).
Kullmann, D. M. Amplitude fluctuations of dual-component EPSCs in hippocampal pyramidal cells: Implications for long-term potentiation. Neuron 12, 1111–1120 (1994).
Edwards, F. LTP is a long term problem. Nature 350, 271–272 (1991).
Isaac, J. T., Nicoll, R. A. & Malenka, R. C. Evidence for silent synapses: implications for the expression of LTP. Neuron 15, 427–434 (1995).
Wang, Y. T., Neuman, R. S. & Bieger, D. Nicotinic cholinoceptor-mediated excitation in ambigual motoneurones of the rat. Neuroscience 40, 759–767 (1991).
Ewert, M., Shivers, B. D., Luddens, H., Mohler, H. & Seeburg, P. H. Subunit selectivity and epitope characterization of mAbs directed against the GABAA/benzodiazepine receptor. J. Cell Biol. 110, 2043–2048 (1990).
Ueno, S., Zorumski, C., Bracamontes, J. & Steinbach, J. H. Endogenous subunits can cause ambiguities in the pharmacology of exogenous gamma-aminobutyric acid A receptors expressed in human embryonic kidney 293 cells. Mol. Pharmacol. 50, 931–938 (1996).
MacDonald, J. F., Mody, I. & Salter, M. W. Regulation of N-methyl-D-aspartate receptors revealed by intracellular dialysis of murine neurones in culture. J. Physiol. (Lond.) 414, 17–34 (1989).
Bekkers, J. M., Richerson, G. B. & Stevens, C. F. Origin of variability in quantal size in cultured hippocampal neurons and hippocampal slices. Proc. Natl Acad. Sci. USA 87, 5359–5362 (1990).
Acknowledgements
We thank M. Wojtowicz and F. Dorri for assistance with slice experiments; C. Kaufman and D. Gunnersen for GABAA receptor β2 and γ2 subunit cDNAs; J. H. Steinbach for the α1FLAGcDNA; D. Carter for the 4D4 cells; C. C. Yip for insulin; and M. W. Salter for helpful comments on the manuscript. This work was supported by the Medical Research Council of Canada and Neuroscience Network of Centers of Excellence of Canada. Y.T.W. is a research scholar of the Heart and Stroke Foundation of Canada/Ontario; Z.G.X. is a MRC-NCE fellow and W.Y.L. is a NCE fellow. H.Y.M. is supported by a studentship from the Canadian Foundation for the Study of Infant Death.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wan, Q., Xiong, Z., Man, H. et al. Recruitment of functional GABAA receptors to postsynaptic domains by insulin. Nature 388, 686–690 (1997). https://doi.org/10.1038/41792
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/41792
This article is cited by
-
Insulin effects on core neurotransmitter pathways involved in schizophrenia neurobiology: a meta-analysis of preclinical studies. Implications for the treatment
Molecular Psychiatry (2023)
-
Function of the GABAergic System in Diabetic Encephalopathy
Cellular and Molecular Neurobiology (2023)
-
Effect of eIF2α in Neuronal Injury Induced by High Glucose and the Protective Mechanism of Resveratrol
Molecular Neurobiology (2023)
-
ALG13 participates in epileptogenesis via regulation of GABAA receptors in mouse models
Cell Death Discovery (2020)
-
Mechanism underlying starvation-dependent modulation of olfactory behavior in Drosophila larva
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.