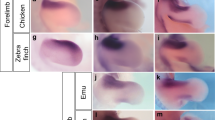
Abstract
The morphological and functional evolution of appendages has played a crucial role in the adaptive radiation of tetrapods, arthropods and winged insects. The origin and diversification of fins, wings and other structures, long a focus of palaeontology, can now be approached through developmental genetics. Modifications of appendage number and architecture in each phylum are correlated with regulatory changes in specific patterning genes. Although their respective evolutionary histories are unique, vertebrate, insect and other animal appendages are organized by a similar genetic regulatory system that may have been established in a common ancestor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Müller, G. B. & Wagner, G. P. Novelty in evolution: Restructuring the concept. Ann. Rev. Ecol. Syst. 22, 229–256 (1991).
Coates, M. I. The origin of vertebrate limbs. Development (suppl.) 169–180 (1994).
Coates, M. I. Fish fins or tetrapod limbs—a simple twist of fate? Curr. Biol. 5, 844–848 (1995).
Shubin, N. The evolution of paired fins and the origin of tetrapod limbs. Evol. Biol. 28, 39–85 (1995).
Coates, M. I. The Devonian tetrapod Acanthostega gunnari Jarvik: postcranial anatomy, basal tetrapod interrelationships and patterns of skeletal evolution. Trans. R. Soc. Edinb. 87, 363–421 (1996).
Johnson, R. & Tabin, C. The long and short of hedgehog signaling. Cell 81, 313–316 (1995).
Nelson, C. E. et al. Analysis of Hox gene expression in the chick limb bud. Development 122, 1449–1466 (1996).
Tabin, C. J. & Laufer, E. Hox genes and serial homology. Nature 361, 692–693 (1993).
Sordino, P., van der Hoeven, F. & Duboule, D. Hox gene expression in teleost fins and the origin of vertebrate digits. Nature 375, 678–681 (1995).
Kessel, M. & Gruss, P. Murine developmental control genes. Science 249, 374–379 (1990).
Mackem, S., Ranson, M. & Mahon, K. Limb-type differences in expression domains of certain chick Hox-4 genes and relationship to pattern modification for flight. Prog. Clin. Biol. Res. 383 A, 21–30 (1993).
Peterson, R. J., Papenbrock, T., Davada, M. M. & Awgulewitschh, A. The murine Hoxc cluster contains five neighboring abdB-related Hox genes that show unique spatially coordinated expression in posterior embryonic subregions. Mech. Dev. 47, 253–260 (1994).
Gibson-Brown, J. J. et al. Evidence of a role for T-box genes in the evolution of limb morphogenesis and the specification of forelimb/hindlimb identity. Mech. Dev. 56, 93–101 (1996).
Davis, A. P., Witte, D. P., Hsieh-Li, H. M., Potter, S. S. & Capecchi, M. R. Absence of radius and ulna in mice lacking hoxa-11 and hoxd-11. Nature 375, 791–795 (1995).
Vorobyeva, E. & Hinchliffe, J. R. From fins to limbs. Evol. Biol. 29, 263–311 (1996).
Hinchliffe, J. R. & Johnson, D. R. The Development of the Vertebrate Limb (Clarendon, Oxford, 1980).
Holmgren, N. On the origin of the tetrapod limb. Acta Zoologica 14, 185–295 (1933).
Holmgren, N. Contribution on the question of the origin of the tetrapod limb. Acta Zoologica 20, 89–124 (1939).
Watson, D. M. S. On the primitive tetrapod limb. Anat. Anzeiger 44, 24–27 (1913).
Gregory, W. K. & Raven, H. C. Studies on the origin and early evolution of paired fins and limbs. Ann. N. Y. Acad. Sci. 42, 273–360 (1941).
Sordino, P. & Duboule, D. Amolecular approach to the evolution of vertebrate paired appendages. Trends Ecol. Evol. 11, 114–119 (1996).
Ahlberg, P. E. & Milner, A. R. The origin and early diversification of tetrapods. Nature 368, 507–512 (1994).
Yokouchi, Y. et al. Homeobox gene expression correlated with the bifurcation process of limb cartilage development. Nature 353, 443–445 (1991).
Gerard, M., Duboule, D. & Zakany, J. C. Cooperation of regulatory elements involved in the activation of the Hoxd-11 gene. Compt. R. Acad. Sci. III 316, 985–994 (1993).
Beckers, J., Gerard, M. & Duboule, D. Transgenic analysis of a potential Hoxd-11 limb regulatory element present in tetrapods and fish. Dev. Biol. 180, 543–553 (1996).
van der Hoeven, F., Zakany, J. & Duboule, D. Gene transpositions in the HoxD complex reveal a hierarchy of regulatory controls. Cell 85, 1025–1035 (1996).
Shubin, N. & Alberch, P. Amorphogenetic approach to the origin and basic organization of the tetrapod limb. Evol. Biol. 20, 318–390 (1986).
Tabin, C. J. Why we have (only) five fingers per hand: hox genes and the evolution of paired limbs. Development 116, 289–296 (1992).
Holder, N. Developmental constraints and the evolution of vertebrate digit patterns. J. Theor. Biol. 104, 451–471 (1983).
Morse, E. On the tarsus and carpus of birds. Ann. Lyc. Nat. Hist. 10, 141–158 (1872).
Shubin, N., Crawford, A. & Wake, D. Morphological variation in the limbs of Taricha granulosa (Caudata: Salamandridae): Evolutionary and phylogenetic implications. Evolution 49, 874–884 (1995).
Greer, A. Limb reduction in the Scincid lizard genus Lerista. 2. Variation in the bone complements of the front and rear limbs and the number of postsacral vertebrae. J. Herpetol. 24, 142–150 (1980).
Lande, R. Evolutionary mechanisms of limb loss in tetrapods. Evolution 32, 73–92 (1978).
Gauthier, J. Saurischian monophyly and the origin of birds. Mem. Calif. Acad. Sci. 8, 1–55 (1986).
MacFadden, B. J. Fossil Horses (Cambridge Univ. Press, 1992).
Davis, A. P. & Capecchi, M. R. Axial homeosis and appendicular skeleton defects in mice with a targeted disruption of hoxd-11. Development 120, 2187–2198 (1994).
Davis, A. P. & Capecchi, M. R. Amutational analysis of the 5′ HoxD genes: Dissection of genetic interactions during limb development in the mouse. Development 122, 1175–1185 (1996).
Favier, B. et al. Functional cooperation between the non-paralogous genes Hoxa-10 and Hoxd0-11 in the developing forelimb and axial skeleton. Development 122, 449–460 (1996).
Dollé, P. et al. Disruption of the Hoxd-13 gene induces localized heterochrony leading to mice with neotenic limbs. Cell 75, 431–441 (1993).
Favier, B., LeMeur, M., Chambon, P. & Dollé, P. Axial skeleton homeosis and forelimb malformations in Hoxd-11 mutant mice. Proc. Natl Acad. Sci. USA 92, 310–314 (1995).
Capecchi, M. R. Function of homeobox genes in skeletal development. Ann. N. Y. Acad. Sci. 97, 34–37 (1996).
Wigglesworth, V. B. Evolution of insect wings and flight. Nature 246, 127–203 (1973).
Budd, G. The morphology of Opabinia regalis and the reconstruction of the arthropod stem-group. Lethaia 29, 1–14 (1996).
Hou, X. G. & Bergström, J. Cambrian lobopodians—ancestors of extant onychophorans? Zool. J. Linn. Soc. Lond. 114, 3–19 (1995).
Simonetta, A. M. & Delle Cave, L. in The Early Evolution of Metazoa and the Significance of Problematic Taxa (eds Simonetta, A. M. & Conway Morris, S.) 189–244 (Cambridge Univ. Press, 1991).
Budd, G. ACambrian gilled lobopod from Greenland. Nature 364, 709–711 (1993).
Chen, J. Y., Ramsköld, L. & Zhou, G. Q. Evidence for monophyly and arthropod affinity of Cambrian giant predators. Science 264, 1304–1308 (1994).
Carroll, S. B. Homeotic genes and the evolution of arthropods and chordates Nature 376, 479–485 (1995).
Struhl, G. Genes controlling segmental specification in the Drosophila thorax. Proc. Natl Acad. Sci. USA 79, 7380–7384 (1982).
Struhl, G. Ahomoeotic mutation transforming leg to antenna in Drosophila. Nature 292, 635–638 (1981).
Gibson, G. & Gehring, W. J. Head and thoracic transformations caused by ectopic expression of Antennapedia during Drosophila development. Development 102, 657–675 (1988).
Stuart, J., Brown, S., Beeman, R. & Denell, R. Adeficiency of the homeotic complex of the beetle Tribolium. Nature 350, 72–47 (1991).
Averof, M. & Akam, M. Hox genes and the diversification of insect–crustacean body plans. Nature 376, 420–423 (1995).
Vachon, G. et al. Homeotic genes of the Bithorax complex repress limb development in the abdomen of the Drosophila embryo through the target gene. Cell 71, 437–450 (1992).
Panganiban, G. et al. The development of crustacean limbs and the evolution of arthropods. Science 270, 1363–1366 (1995).
Manton, S. M. Mandibular Mechanisms and the Evolution of Arthropods Vol. 247 (British Museum and Queen Mary College, London, 1964).
Wheeler, W. C., Cartwright, P. & Hayashi, C. Y. Arthropod phylogeny: a combined approach. Cladistics 9, 1–39 (1993).
Boore, J. L., Collins, T. M., Stanton, D., Daehler, L. L. & Brown, W. M. Deducing the pattern of arthropod phylogeny from mitochondrial DNA rearrangements. Nature 376, 163–165 (1995).
Cohen, S. M. & Jürgens, G. Proximal–distal pattern formation in Drosophila: cell autonomous requirement for Distal-less gene activity in limb development. EMBO J. 8, 2045–2055 (1989).
Cohen, S. et al. Distal-less encodes a homeodomain protein required for limb development in Drosophila. Nature 338, 432–434 (1989).
Panganiban, G., Nagy, L. & Carroll, S. B. The development and evolution of insect limb types. Curr. Biol. 4, 671–675 (1994).
Popadic, A., Rusch, D., Peterson, M., Rogers, B. T. & Kaufman, T. C. Origin of the arthropod mandible. Nature 380, 395 (1996).
Jeram, A. J., Selden, P. A. & Edwards, D. Land animals in the Silurian: Arachinids and myriapods from Shropshire, England. Science 250, 658–661 (1990).
Kukalová-Peck, J. The Insects of Australia 2nd edn (Cornell University Press, Ithaca, NY, 1991).
Snodgrass, R. Principles of Insect Morphology (McGraw-Hill, New York, 1935).
. Kukalova-Peck, J. Origin and evolution of insect wings and their relation to metamorphosis, as documented from the fossil record. J. Morphol. 156, 53–126 (1978).
Marden, J. H. & Kramer, M. G. Surface-skimming stoneflies: A possible intermediate stage in insect flight evolution. Science 266, 427–430 (1994).
Cohen, B. et al. Allocation of the thoracic imaginal primordia in the Drosophila embryo. Development 117, 597–608 (1993).
Diaz-Benjumea, F. & Cohen, S. M. Interaction between dorsal and ventral cells in the imaginal disc directs wing development in Drosophila. Cell 75, 741–752 (1993).
Carroll, S. B. et al. Pattern formation and eyespot determination in butterfly wings. Science 265, 109–114 (1994).
Averof, M. & Cohen, S. M. Evolutionary origin of insect wings from ancestral gills. Nature 385, 627–630 (1997).
Lee, J. J. et al. Secretion and localized transcription suggest a role in positional signaling for products of the segmentation gene hedgehog. Cell 71, 33–50 (1992).
Tabata, T. et al. The Drosophila hedgehog gene is expressed specifically in posterior compartment cells and is a target of engrailed regulation. Genes Dev. 6, 2635–2645 (1992).
Basler, D. & Struhl, G. Compartment boundaries and the control of Drosophila limb pattern by hedgehog protein. Nature 368, 208–214 (1994).
Posakony, L., Raftery, L. & Gelbart, W. Wing formation in Drosophila melanogaster requires decapentaplegic gene function along the anterior–posterior compartment boundary. Mech. Dev. 33, 69–82 (1991).
Capdevila, J. & Guerrero, I. The Drosophila segment polarity gene patched interacts with decapentaplegic in wing development. EMBO J. 6, 715–729 (1994).
Sanicola, M., Sekelsky, J., Elson, S. & Gelbart, W. M. Drawing a stripe in Drosophila imaginal discs: negative regulation of decapentaplegic and patched expression. Genetics 139, 745–756 (1995).
Nellen, D., Burke, R., Struhl, G. & Basler, K. Direct and long-range actions of a Dpp morphogen gradient. Cell 85, 357–368 (1996).
Lecuit, T. et al. Two distinct mechanisms for long-range patterning by Decapentaplegic in the Drosophila wing. Nature 381, 387–393 (1996).
Echelard, Y. et al. Sonic hedgehog, a member of a family of putative signaling molecules, is implicated in the regulation of CNS polarity. Cell 75, 1417–1430 (1993).
Krauss, S., Concordet, J. P. & Ingham, P. W. Afunctionally conserved homolog of the Drosophila segment polarity gene hh is expressed in tissues with polarizing activity in zebrafish embryos. Cell 75, 1431–1444 (1993).
Chang, D. T. et al. Products, genetic linkage and limb patterning activity of a murine hedgehog gene. Development 120, 3339–3353 (1994).
Riddle, R. D. et al. Sonic hedgehog mediates the polarizing activity of the ZPA. Cell 75, 1401–1416 (1995).
Tickle, C. Genetics and limb development. Dev. Genet. 19, 1–8 (1996).
Irvine, K. & Weischaus, E. fringe, a boundary-specific signaling molecule, mediates interactions between dorsal and ventral cells during Drosophila wing development. Cell 79, 595–606 (1994).
Spreicher, S., Thomas, U., Hinz, U. & Knust, E. The Serrate locus of Drosophila and its role in morphogenesis or imaginal discs: control of cell proliferation. Development 120, 535–544 (1994).
Kim, J., Irvine, K. & Carroll, S. Cell recognition, signal induction, and symmetrical gene activation at the dorsal-ventral boundary of the developing Drosophila wing. Cell 82, 795–802 (1995).
Couso, J. P., Knust, E. & Martinez Ariias, A. Serrate and wingless cooperate to induce vestigial gene expression and wing formation in Drosophila. Curr. Biol. 5, 1437–1448 (1995).
Diaz-Benjumea, F. J. & Cohen, S. Serrate signals through Notch to establish a Wingless-dependent organizer at the dorsal/ventral compartment boundary of the Drosophila wing. Development 121, 4215–4225 (1995).
Kim, J. et al. Integration of positional signals and regulation of wing formation and identity by Drosophila vestigial gene. Nature 382, 133–138 (1996).
Todt, W. L. & Fallon, J. F. Development of the apical ectodermal ridge in the chick wing bud. J. Embryol. Exp. Morphol. 80, 21–41 (1984).
Rodriguez-Estaban, C. et al. Radical fringe positions the apical ectodermal ridge at the dorsoventral boundary of the vertebrate limb. Nature 386, 360–361 (1997).
Laufer, E. et al. Expression of Radical fringe in limb-bud ectoderm regulates apical ectodermal ridge formation. Nature 386, 366–373 (1997).
Williams, J. A., Paddock, S. W. & Carroll, S. B. Pattern formation in a secondary field: A hierarchy of regulatory genes subdivides the developing Drosophila wing disc into discrete sub-regions. Development 117, 571–584 (1993).
Couso, J. P., Bate, M. & Martinez-Ariias, A. Awingless-dependent polar coordinate system in Drosophila imaginal discs. Science 259, 484–489 (1993).
Parr, B. A. & McMahon, A. P. Dorsalizing signal Wnt-7a required for normal polarity of D-V and A-P axes of mouse limb. Nature 374, 350–353 (1995).
Riddle, R. D. et al. Induction of the LIM homeobox gene Lmx-1 by Wnt-7a establishes dorsoventral pattern in the vertebrate limb. Cell 83, 631–640 (1995).
Vogel, A. et al. Dorsal cell fate specified by chick Lmx1 during vertebrate limb development. Nature 378, 716–720 (1995).
Raff, R. The Shape of Life (Univ. Chicago Press, 1996).
Panganiban, G. et al. The origin and evolution of animal appendages. Proc. Natl Acad. Sci. USA 94, 5162–5166 (1997).
Wall, N. A. & Hogan, B. L. M. Expression of bone morphogenetic protein-4 (BMP-4), bone morphogenetic protein-7 (BMP-7), fibroblast growth factor-8 (FGF-8) and Sonic hedgehog (SHH) during branchial arch development in the chick. Mech. Dev. 53, 383–392 (1995).
Marigo, V., Scott, M. P., Johnson, R. L., Goodrich, L. V. & Tabin, C. J. Conservation in hedgehog signaling: induction of a chicken patched homolog by Sonic hedgehog in the developing limb. Development 122, 1225–1233 (1996).
Roth, V. L. Homology and hierarchies: Problems solved and unresolved. J. Evol. Biol. 4, 167–194 (1991).
Wagner, G. P. The origin of morphological characters and the biological basis of homology. Evolution 43, 1157–1171 (1989).
Bolker, J. A. & Raff, R. A. Developmental genetics and traditional homology. BioEssays 18, 489–494 (1996).
Carroll, R. L. Vertebrate Paleontology (Freeman, San Francisco, 1988).
Jarvik, E. The Structure and Evolution of the Vertebrates Vol. 1 (Academic, New York, 1980).
Jarvik, E. The Devonian tetrapod Ichthyostega. Fossils and Strata 40, 1–213 (1996).
Fromental-Ramain, C. et al. Specific and redundant functions of the paralogous Hoxa-9 and Hoxd-9 genes in forelimb and axial skeleton patterning. Development 122, 461–472 (1996).
Mortlock, D. P., Post, L. C. & Innis, J. W. The molecular basis of hypodactyly (Hd): a deletion in Hoxa13 leads to arrest of digital arch formation. Nature Genet. 13, 284–289 (1996).
Mortlock, D. P. & Innis, J. W. Mutation of HOXA13 in hand–foot–genital syndrome. Nature Genet. 15, 179–181 (1997).
Saunders, J. The proximo-distal sequence of origin of the parts of the chick wing and the role of the ectoderm. J. Exp. Zool. 108, 363–403 (1948).
Summerbell, D., Lewis, J. H. & Wolpert, L. Postional information in chick limb morphogenesis. Nature 244, 492–496 (1973).
Lebedev, O. A. & Coates, M. I. The postcranial skeleton of the Devonian tetrapod Tulerpeton curtum Lebedev. Zool. J. Linn. Soc. 113, 302–348 (1995).
Hou, X. G., Bergström, J. & Ahlberg, P. Anomalocaris and other large animals in the Lower Cambrian Chenjiang fauna of southwest China. Geol Forening. Forhandling. 117, 163–183 (1995).
Acknowledgements
We thank P. Ahlberg, G. Budd, A. C. Burke, M. Coates, A. Meyer, G. Panganiban, P. Sniegowski, D. Wake, R. S. Winters, L. Wolpert and members of our laboratories for their critiques of drafts of this manuscript. S.B.C. is an investigator of the HHMI. C.T. is supported by grants from the NIH and the American Cancer Society. N.S. is supported by grants from the NSF, from the National Geographic Society and from the Research Foundation of the University of Pennsylvania.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shubin, N., Tabin, C. & Carroll, S. Fossils, genes and the evolution of animal limbs. Nature 388, 639–648 (1997). https://doi.org/10.1038/41710
Issue Date:
DOI: https://doi.org/10.1038/41710
This article is cited by
-
The pelvic girdle in extant gonorynchiformes (Teleostei: Otomorpha)
Zoomorphology (2024)
-
Fossil evidence for a pharyngeal origin of the vertebrate pectoral girdle
Nature (2023)
-
Speciation and historical migration pattern interaction: examples from P. nigra and P. sylvestris phylogeography
European Journal of Forest Research (2023)
-
Co-option of the limb patterning program in cephalopod eye development
BMC Biology (2022)
-
Galeaspid anatomy and the origin of vertebrate paired appendages
Nature (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.