Abstract
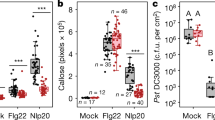
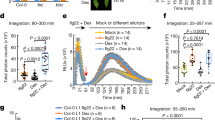
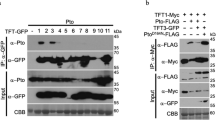
There is remarkable conservation in the recognition of pathogen-associated molecular patterns (PAMPs) by innate immune responses of plants, insects and mammals. We developed an Arabidopsis thaliana leaf cell system based on the induction of early-defence gene transcription by flagellin, a highly conserved component of bacterial flagella that functions as a PAMP in plants and mammals. Here we identify a complete plant MAP kinase cascade (MEKK1, MKK4/MKK5 and MPK3/MPK6) and WRKY22/WRKY29 transcription factors that function downstream of the flagellin receptor FLS2, a leucine-rich-repeat (LRR) receptor kinase. Activation of this MAPK cascade confers resistance to both bacterial and fungal pathogens, suggesting that signalling events initiated by diverse pathogens converge into a conserved MAPK cascade.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Boller, T. Chemoperception of microbial signals in plant cells. Annu. Rev. Plant Physiol. Plant. Mol. Biol. 46, 189–214 (1995).
Aderem, A. & Ulevitch, R. J. Toll-like receptors in the induction of the innate immune response. Nature 406, 782–787 (2000).
Meindl, T., Boller, T. & Felix, G. The bacterial elicitor flagellin activates its receptor in tomato cells according to the address-message concept. Plant Cell 12, 1783–1794 (2000).
Khush, R. S. & Lemaitre, B. Genes that fight infection. Trends Genet. 16, 442–449 (2000).
Cohn, J., Sessa, G. & Martin, G. B. Innate immunity in plants. Curr. Opin. Immunol. 13, 55–62 (2001).
Dangl, J. L. & Jones, J. D. G. Plant pathogens and integrated defence responses to infection. Nature 411, 826–833 (2001).
Nurnberger, T. & Scheel, D. Signal transmission in plant immune response. Trends Plant Sci. 6, 372–379 (2001).
Staskawicz, B. J., Mudgett, M. B., Dangl, J. L. & Galan, J. E. Common and contrasting themes of plant and animal diseases. Science 292, 2285–2289 (2001).
Samakovlis, C., Asling, B., Boman, H. G., Gateff, E. & Hultmark, D. In vitro induction of cecropin genes—an immune response in a Drosophila blood cell line. Biochem. Biophys. Res. Commun. 188, 1169–1175 (1992).
Felix, G., Duran, J., Volko, S. & Boller, T. Plants have a sensitive perception system for the most conserved domain of bacterial flagellin. Plant J. 18, 265–276 (1999).
Hayashi, F. et al. The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5. Nature 410, 1099–1103 (2001).
Gomez-Gomez, L. & Boller, T. FLS2: An LRR receptor-like kinase involved in the perception of the bacterial elicitor flagellin in Arabidopsis. Mol. Cell 5, 1003–1011 (2000).
Gomez-Gomez, L., Felix, G. & Boller, T. A single locus determines sensitivity to bacterial flagellin in Arabidopsis thaliana. Plant J. 18, 277–284 (1999).
Romeis, T. et al. Rapid Avr9- and Cf9-dependent activation of MAP kinases in tobacco cell cultures and leaves: Convergence of resistance gene, elicitor, wound, and salicylate responses. Plant Cell 11, 273–287 (1999).
Blume, B., Nurnberger, T., Nass, N. & Scheel, D. Receptor-mediated increase in cytoplasmic free calcium required for activation of pathogen defense in parsley. Plant Cell 12, 1425–1440 (2000).
Hirt, H. & Scheel, D. in Results and Problems in Cell Differentiation: MAP Kinases in Plant Signal Transduction (ed. Hirt, H.) 85–93 (Springer, Heidelberg, 2000).
Zhang, S. & Klessig, D. F. MAPK cascades in plant defense signaling. Trends Plant Sci. 6, 520–527 (2001).
Romeis, T. Protein kinases in the plant defence response. Curr. Opin. Plant Biol. 4, 407–414 (2001).
Tena, G., Asai, T., Chiu, W.-L. & Sheen, J. Plant MAP kinase signaling cascades. Curr. Opin. Plant Biol. 4, 392–400 (2001).
Ligterink, W., Kroj, T., Zurnieden, U., Hirt, H. & Scheel, D. Receptor-mediated activation of a MAP kinase in pathogen defense in plants. Science 276, 2054–2057 (1997).
Zhang, S. & Klessig, D. F. Resistance gene N-mediated de novo synthesis and activation of a tobacco mitogen-activated protein kinase by tobacco mosaic virus infection. Proc. Natl Acad. Sci. USA 95, 7433–7438 (1998).
Zhang, S., Du, H. & Klessig, D. F. Activation of the tobacco SIP kinase by both a cell-wall-derived carbohydrate elicitor and purified proteinaceous elicitins from Phytophthora spp. Plant Cell 10, 435–449 (1998).
Nuhse, T. S., Peck, S. C., Hirt, H. & Boller, T. Microbial elicitors induce activation and dual phosphorylation of the Arabidopsis thaliana MAPK6. J. Biol. Chem. 275, 7521–7526 (2000).
Cardinale, F. et al. Differential activation of four specific MAPK pathways by distinct elicitors. J. Biol. Chem. 275, 36734–36740 (2000).
Lee, J., Klessig, D. F. & Nurnberger, T. A. A harpin binding site in tobacco plasma membranes mediates activation of the pathogenesis-related gene HIN1 independent of extracellular calcium but dependent on mitogen-activated protein kinase activity. Plant Cell 13, 1079–1093 (2001).
Sheen, J. Signal transduction in maize and Arabidopsis mesophyll protoplasts. Plant Physiol. (in the press).
Durrant, W. E., Rowland, O., Piedras, P., Hammond-Kosack, K. E. & Jones, J. D. G. cDNA-AFLP reveals a striking overlap in race-specific resistance and wound response gene expression profiles. Plant Cell 12, 963–977 (2000).
Maleck, K. et al. The transcriptome of Arabidopsis thaliana during systemic acquired resistance. Nature Genet. 26, 403–410 (2000).
Schenk, P. M. et al. Coordinated plant defense responses in Arabidopsis revealed by microarray analysis. Proc. Natl Acad. Sci. USA 97, 11655–11660 (2000).
Asai, T. et al. Fumonisin B1-induced cell death in Arabidopsis protoplasts requires jasmonate-, ethylene-, and salicylate-dependent signaling pathways. Plant Cell 12, 1823–1835 (2000).
Glazebrook, J. Genes controlling expression of defense responses in Arabidopsis—2001 status. Curr. Opin. Plant Biol. 4, 301–308 (2001).
Eulgem, T., Rushton, P. J., Robatzek, S. & Somssich, I. E. The WRKY superfamily of plant transcription factors. Trends Plant Sci. 5, 199–206 (2000).
Eulgem, T., Rushton, P. J., Schmeizer, E., Hahlbrock, K. & Somssich, I. E. Early nuclear events in plant defence signalling: rapid gene activation by WRKY transcription factors. EMBO J. 18, 4689–4699 (1999).
Du, L. & Chen, Z. Identification of genes encoding receptor-like protein kinases as possible targets of pathogen- and salicylic acid-induced WRKY DNA-binding proteins in Arabidopsis. Plant J. 24, 837–847 (2000).
Kovtun, Y., Chiu, W.-L., Tena, G. & Sheen, J. Function analysis of oxidative stress-activated mitogen-activated protein kinase cascade in plants. Proc. Natl Acad. Sci. USA 97, 2940–2945 (2000).
Kovtun, Y., Chiu, W.-L., Zeng, W. & Sheen, J. Suppression of auxin signal transduction by a MAPK cascade in higher plants. Nature 395, 716–720 (1998).
Mizoguchi, T., Ichimura, K., Yoshida, R. & Shinozaki, K. in Results and Problems in Cell Differentiation: MAP Kinases in Plant Signal Transduction (ed. Hirt, H.) 29–38 (Springer, Heidelberg, 2000).
Jouannic, S. et al. Plant MAP kinase kinase kinase structure, classification and evolution. Gene 233, 1–11 (1999).
Xiang, C., Han, P., Lutziger, I., Wang, K. & Oliver, D. J. A mini binary vector series for plant transformation. Plant Mol. Biol. 40, 711–717 (1999).
Peck, S. C. et al. Directed proteomics identifies a plant-specific protein rapidly phosphorylated in response to bacterial and fungal elicitors. Plant Cell 13, 1467–1475 (2001).
Yang, K.-Y., Liu, Y. & Zhang, S. Activation of a mitogen-activated protein kinase pathway is involved in disease resistance in tobacco. Proc. Natl Acad. Sci. USA 98, 741–746 (2001).
Petersen, M. et al. Arabidopsis MAP kinase 4 negatively regulates systemic acquired resistance. Cell 103, 1111–1120 (2000).
Frye, C. A., Tang, D. & Innes, R. W. Negative regulation of defense responses in plants by a conserved MAPKK kinase. Proc. Natl Acad. Sci. USA 98, 373–378 (2001).
Swiderski, M. R. & Innes, R. W. The Arabidopsis PBS1 resistance gene encodes a member of a novel protein kinase subfamily. Plant J. 26, 101–112 (2001).
Madhani, H. D. & Fink, G. R. The riddle of MAP kinase signalling specificity. Trends Genet. 14, 151–155 (1998).
Patharkar, O. R. & Cushman, J. C. A stress-induced calcium-dependent protein kinase from Mesembryanthemum crystallinum phosphorylates a two-component pseudo-response regulator. Plant J. 24, 679–691 (2000).
Acknowledgements
We thank C. Xiang for the pCB302 minibinary vector, O. R. Patharkar and J. Cushman for the DsRed-CSP1 plasmid; S. Volko and J. Stone for sharing unpublished information; S.-H. Cheng, B. Moore and I. Hwang for technical advice; W.-C. Chen for help in bioinformatics; and S. Ramu and F. Rolland for critical reading of the manuscript. This work was supported by a NSF plant genome project grant to J.S. and F.M.A., a USDA grant to J.S., and a NIH grant to F.M.A. T.A. was supported in part by fellowships from the Toyobo Biotechnology Foundation and the Uehara Memorial Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Asai, T., Tena, G., Plotnikova, J. et al. MAP kinase signalling cascade in Arabidopsis innate immunity. Nature 415, 977–983 (2002). https://doi.org/10.1038/415977a
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/415977a
This article is cited by
-
Transcriptomics of temperature-sensitive R gene-mediated resistance identifies a WAKL10 protein interaction network
Scientific Reports (2024)
-
Plant U-box E3 ligases PUB20 and PUB21 negatively regulate pattern-triggered immunity in Arabidopsis
Plant Molecular Biology (2024)
-
Cadmium toxicity: its’ uptake and retaliation by plant defence system and ja signaling
BioMetals (2024)
-
Genome-wide identification and immune response analysis of mitogen-activated protein kinase cascades in tea geometrid, Ectropis grisescens Warren (Geometridae, Lepidoptera)
BMC Genomics (2023)
-
Transcriptome analysis of sugarcane reveals rapid defense response of SES208 to Xanthomonas albilineans in early infection
BMC Plant Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.