Abstract
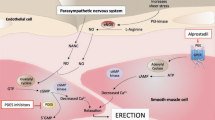
This meta-analytic study aims to estimate the likely improvements of erectile dysfunction (ED) measured by the International Index of Erectile Function (IIEF) at the highest fixed dosages of the three available PDE-5-inhibitors: sildenafil, tadalafil, and vardenafil. MEDLINE and the Cochrane Library were searched electronically for efficacy trials of PDE-5-inhibitors for treating ED. In addition drug manufacturers were contacted to provide unpublished or unrecorded congress proceedings. Randomized, double-blind, placebo-controlled, parallel-group, maximum fixed-dose, broad-spectrum efficacy trials using IIEF were included in the analysis. Data were independently extracted by two reviewers. The results were pooled using weighted mean differences. A formal indirect comparison (including Bonferroni-correction) was conducted to estimate the differences between agents. A total of 14 trials were included in the meta-analysis (three with 100 mg sildenafil, eight with 20–25 mg tadalafil, and three with 20 mg vardenafil). All trials were of good methodological quality. Overall heterogeneity was moderate: I2=33.2%, χ2=19.47, P=0.11. The funnel plot suggested moderate likelihood of publication bias. Pooled results of IIEF-improvement were for sildenafil 9.65 (95% CI: 8.50, 10.79) points, tadalafil 8.52 (7.61, 9.42) points, and vardenafil 7.50 (6.50, 8.50) points, respectively. Sildenafil proved to be significantly more effective than vardenafil (d=2.15, P=0.006), other pairwise comparisons showed no difference in efficacy. All PDE-5-inhibitors are highly effective in the treatment of ED. At maximum dosage they improve erectile function 7–10 points on the IIEF compared to placebo-treatment. There is evidence that sildenafil might be more efficacious than vardenafil, although this is to be interpreted with caution. To prove higher efficacy truly independent comparative trials are needed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Rosen RC, Fisher WA, Eardley I, Niederberger C, Nadel A, Sand M . The multinational Men's Attitudes to Life Events and Sexuality (MALES) study: I. Prevalence of erectile dysfunction and related health concerns in the general population. Curr Med Res Opin 2004; 20: 607–617.
Nicolosi A, Glasser DB, Moreira ED, Villa M, Erectile Dysfunction Epidemiology Cross National Study Group. Prevalence of erectile dysfunction and associated factors among men without concomitant diseases: a population study. Int J Impot Res 2003; 15: 253–257.
Heruti R, Shochat T, Tekes-Manova D, Ashkenazi I, Justo D . Prevalence of erectile dysfunction among young adults: results of a large-scale survey. J Sex Med 2004; 1: 284–291.
Auld RB, Brock G . Sexuality and erectile dysfunction: results of a national survey. J Sex Reprod Med 2002; 2: 50–54.
Kubin M, Wagner G, Fugl-Meyer AR . Epidemiology of erectile dysfunction. Int J Impot Res 2003; 15: 63–71.
Wyllie MG . The phosphodiesterase inhibitor ‘war’. Brit J Urol Int 2003; 91: 573.
Burls A, Gold L, Clark W . Systematic review of randomised controlled trials of sildenafil (Viagra) in the treatment of male erectile dysfunction. Br J Gen Pract 2001; 51: 1004–1012.
Fink HA, Mac Donald R, Rutks IR, Nelson DB, Wilt TJ . Sildenafil for male erectile dysfunction: a systematic review and meta-analysis. Arch Intern Med 2002; 162: 1349–1360.
Moore RA, Edwards JE, McQuay HJ . Sildenafil (Viagra) for male erectile dysfunction: a meta-analysis of clinical trial reports. BMC Urol 2002; 2: 6.
Brock GB, McMahon CG, Chen KK, Costigan T, Shen W, Watkins V et al. Efficacy and safety of tadalafil for the treatment of erectile dysfunction: results of integrated analyses. J Urol 2002; 168: 1332–1336.
Hellstrom WJG, Gittelman M, Karlin G, Segerson T, Thibonnier M, Taylor T et al. Vardenafil for treatment of men with erectile dysfunction: efficacy and safety in a randomized, double-blind, placebo-controlled trial. J Androl 2002; 23: 763–771.
Porst H, Rosen R, Padma-Nathan H, Goldstein I, Giuliano F, Ulbrich E et al. The efficacy and tolerability of vardenafil, a new, oral, selective phosphodiesterase type 5 inhibitor, in patients with erectile dysfunction: the first at-home clinical trial. Int J Impot Res 2001; 13: 192–199.
Brock G, Nehra A, Lipshultz LI, Karlin GS, Gleave M, Seger M et al. Safety and efficacy of vardenafil for the treatment of men with erectile dysfunction after radical retropubic prostatectomy. J Urol 2003; 170: 1278–1283.
Goldstein I, Young JM, Fischer J, Bangerter K, Segerson T, Taylor T et al. Vardenafil, a new phosphodiesterase type 5 inhibitor, in the treatment of erectile dysfunction in men with diabetes: a multicenter double-blind placebo-controlled fixed-dose study. Diabetes Care 2003; 26: 777–783.
Hatzichristou D, Montorsi F, Buvat J, Laferriere N, Bandel TJ, Porst H et al. The efficacy and safety of flexible-dose vardenafil (levitra) in a broad population of European men. Eur Urol 2004; 45: 634–641.
Burris AS, Banks SM, Sherins RJ . Quantitative assessment of nocturnal penile tumescence and rigidity in normal men using a home monitor. J Androl 1989; 10: 492–497.
Rosen RC, Riley A, Wagner G . The International Index of Erectile Function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology 1997; 49: 822–830.
Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 1996; 17: 1–12.
Higgins JP, Thompson SG, Deeks JJ, Altman DG . Measuring inconsistency in meta-analyses. BMJ 2003; 327: 557–560.
Bucher HC, Guyatt GH, Griffith LE, Walter SD . The results of direct and indirect treatment comparisons in meta-analysis of randomized controlled trials. J Clin Epidemiol 1997; 50: 683–691.
Deeks JJ, Altman DG, Bradburn MJ . Statistical methods for examining heterogeneity and combining results from several studies in meta-analysis. In: Egger M, Smith GD, Altman DG, (eds.), Systematic Reviews in Health Care: Meta-Analysis in Context. London: BMJ Publishing Group, 2003 pp. 248–282.
Song F, Altman DG, Glenny AM, Deeks JJ . Validity of indirect comparison for estimating efficacy of competing interventions: empirical evidence from published meta-analyses. BMJ 2003; 326: 472.
Montorsi F, McDermott TED, Morgan R, Olsson A, Schultz A, Kirkeby HJ et al. Efficacy and safety of fixed-dose oral sildenafil in the treatment of erectile dysfunction of various etiologies. Urology 1999; 53: 1011–1018.
Padma-Nathan H, Stecher VJ, Sweeney M, Orazem J, Tseng L-J, deRiesthal H . Minimal time to successful intercourse after sildenafil citrate: results of a randomized, double-blind, placebo-controlled trial. Urology 2003; 62: 400–403.
Shirai M . Randomized, placebo-controlled, double-blind study of oral sildenafil in Japanese men with erectile dysfunction. Nishinihon J Urol 2000; 62: 373–382.
Goldstein I, Lue TF, Padma-Nathan H, Rosen RC, Steers WD, Wicker PA . Oral sildenafil in the treatment of erectile dysfunction. New Engl J Med 1998; 338: 1397–1404.
Lue TF . A study of sildenafil (viagra), a new oral agent for the treatment of male erectile dysfunction. J Urol 1997; 157: 181.
Brock G, Iglesias J, Toulouse K, Ferguson KM, Pullman WE, Anglin G . Efficacy and safety of IC351 treatment for ED. Eur Urol 2001; 39: 106.
Faria GE, Torres LO, Koff WJ, Claro JA, Cairoli CD, Fregonesi A et al. Efficacy and safety of tadalafil in Brazilian men with ED. Int J Impot Res 2003; 15: 10.
Padma-Nathan H, McMurray JG, Pullman WE, Whitaker JS, Saoud JB, Ferguson KM et al. On-demand IC351 (Cialis) enhances erectile function in patients with erectile dysfunction. Int J Impot Res 2001; 13: 2–9.
U.S. Food and Drug Administration/Center for Drug Evaluation and Research. NDA-21368 [review]. http://www.fda.gov/cder/approval/index.htm.2004.
Hellstrom WJ, Gittelman M, Karlin G, Segerson T, Thibonnier M, Taylor T et al. Sustained efficacy and tolerability of vardenafil, a highly potent selective phosphodiesterase type 5 inhibitor, in men with erectile dysfunction: results of a randomized, double-blind, 26-week placebo-controlled pivotal trial. Urology 2003; 61: 8–14.
U.S. Food and Drug Administration/Center for Drug Evaluation and Research. NDA-21400 [review]. http://www.fda.gov/cder/approval/index.htm.2003.
Berner M, Kriston L, Harms A . Covariates of treatment effect in trial with oral sildenafil in the treatment of erectile dysfunction – a meta-analytic regression analysis. J Sex Med 2005; 2: 56.
Montorsi F, Verheyden B, Meuleman E, Junemann KP, Moncada I, Valiquette L et al. Long-term safety and tolerability of tadalafil in the treatment of erectile dysfunction. Eur Urol 2004; 45: 339–345.
Potempa AJ, Ulbrich E, Bernard I, Beneke M, Vardenafil Study Group. Efficacy of vardenafil in men with erectile dysfunction: a flexible-dose community practice study. Eur Urol 2004; 46: 73–79.
Benchekroun A, Faik M, Benjelloun S, Bennani S, El Mrini M, Smires A . A baseline-controlled, open-label, flexible dose-escalation study to assess the safety and efficacy of sildenafil citrate (Viagra) in patients with erectile dysfunction. Int J Impot Res 2003; 15: 19–24.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of interest
There was no specific funding for this project. MB currently holds research grants from the German Ministry for Education and Research, Boehringer Ingelheim Inc., Pfizer Inc. and Willmar Schwabe Inc. He did receive tuition fees from Glaxo Smith Kline, Hormosan Kwizda, Lilly, Pfizer and Willmar Schwabe. He also received travel expenses from the European Sexual Dysfunction Alliance affiliate (ISG e.V.) that receives sponsoring from the pharmaceutical industry. LK did receive travel expenses reimbursement from Pfizer and Wilmar Schwabe. AH holds a paid part time position with the European Sexual Dysfunction Alliance affiliate (ISG e.V.).
Rights and permissions
About this article
Cite this article
Berner, M., Kriston, L. & Harms, A. Efficacy of PDE-5-inhibitors for erectile dysfunction. A comparative meta-analysis of fixed-dose regimen randomized controlled trials administering the International Index of Erectile Function in broad-spectrum populations. Int J Impot Res 18, 229–235 (2006). https://doi.org/10.1038/sj.ijir.3901395
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijir.3901395
Keywords
This article is cited by
-
Contemporary cost-analysis comparison of direct-to-consumer vs. traditional prescriptions of phosphodiesterase-5 inhibitors
International Journal of Impotence Research (2023)
-
Direct comparison of tadalafil with sildenafil for the treatment of erectile dysfunction: a systematic review and meta-analysis
International Urology and Nephrology (2017)
-
The impact of formulary replacement of sildenafil by vardenafil at a local VA hospital
International Journal of Impotence Research (2008)
-
The application of quantitative methods for identifying and exploring the presence of bias in systematic reviews: PDE-5 inhibitors for erectile dysfunction
International Journal of Impotence Research (2008)
-
A typology of men's sexual attitudes, erectile dysfunction treatment expectations and barriers
International Journal of Impotence Research (2007)