Abstract
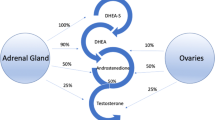
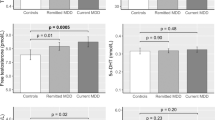
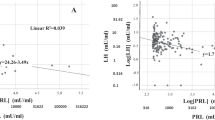
The aim of the present study was to investigate a possible correlation between decreased androgen levels and female sexual function index (FSFI) in women with low libido and compare these findings with normal age-matched subjects. In total, 20 premenopausal women with low libido (mean age 36.7; range 24–51 y) and 20 postmenopausal women with low libido (mean age 54; 45–70 y), and 20 premenopausal healthy women (mean age 32.2; range 21–51 y) and 20 postmenopausal healthy women (mean age 53.5; range 48–60 y) as controls were enrolled in the current study. Women with low libido had symptoms for at least 6 months and were in stable relationships. All premenopausal patients had regular menstrual cycles and all postmenopausal patients and controls were on estrogen replacement therapy. None of the patients were taking birth control pills, corticosteroids or had a history of chronic medical illnesses. All completed the FSFI and Beck's Depression Inventory (BDI) questionnaires. Hormones measured included: cortisol; T3, T4 and TSH; estradiol; total and free testosterone; dehydroepiandrosterone sulfate (DHEA-S); sex hormone binding globulin (SHBG). We performed statistical analysis by parametric and nonparametric comparisons and correlations, as appropriate. We found significant differences between the women with low libido and the controls in total testosterone, free testosterone and DHEA-S levels and full-scale FSFI score for both pre- and postmenopausal women (P<0.05). In addition, decreased total testosterone, free testosterone and DHEA-S levels positively correlated with full-scale FSFI score and FSFI-desire, FSFI-arousal, FSFI-lubrication and FSFI-orgasm scores (P<0.05). Our data suggest that women with low libido have lower androgen levels compared to age-matched normal control groups and their decreased androgen levels correlate positively with female sexual function index domains.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hartmann U, Heiser K, Rüffer-Hesse C, Kloth G . Female sexual desire disorders: subtypes, classification, personality factors and new directions for treatment. World J Urol 2002; 20: 79–88.
Davis S, McCloud P, Strauss B, Burger H . Testosterone enhances estradiol's effects on postmenopausal bone density and sexuality. Maturitas 1996; 21: 227–236.
Davis S . Androgen replacement in women: a commentary. J Clin Endocr Metab 1999; 84: 1886–1891.
Davis S . Testosterone and sexual desire in women. J Sex Educ Ther 2000; 25: 25–32.
Sarrel P, Dobay B, Wiita B . Estrogen and estrogen–androgen replacement in postmenopausal women dissatisfied with estrogen–only therapy: sexual behavior and neuroendocrine responses. J Reprod Med 1998; 43: 847–856.
Sarrel P . Effect of hormone replacement therapy on sexual psychophysiology and behavior in postmenopause. J Women Health Gender Based Med 2000; 9(Suppl 1): 25–32.
Guay AT . Decreased testosterone in regularly menstruating women with decreased libido: a clinical observation. J Sex Marital Ther 2001; 27: 513–519.
Riley A, Riley E . Controlled studies on women presenting with sexual disorders: I. Endocrine status. J Sex Marital Ther 2000; 26: 269–283.
Rosen R et al. The female sexual function index (FSFI): a multidimensional self-report instrument for the assessment of female sexual function. J Sex Marital Ther 2000; 26: 191–208.
Tazuke S, Khaw K, Barrett-Connor E . Exogenous estrogen and endogenous sex hormones. Medicine 1992; 71: 44–51.
Guay AT, Jacobson J . Decreased free testosterone and dehydroepiandrosterone-sulfate (DHEA-S) levels in women with decreased libido. J Sex Marital Ther 2002; 28: 129–142.
Bachmann G et al. Female androgen insufficiency: The Princeton consensus statement on definition, classification, and assessment. Fertil Steril 2002; 4: 660–665.
Laumann EO, Paik A, Rosen RC . Sexual dysfunction in the United States: prevalence and predictors. JAMA 1999; 281: 537–544.
Munarriz R et al. Androgen replacement therapy with dehydroepiandrosterone for androgen insufficiency and female sexual dysfunction: androgen and questionnaire results. J Sex Marital Ther 2002; 28: 165–173.
Massafra C et al. Androgens and osteocalcin during the menstrual cycle. J Clin Endoc Met 1999; 84: 971–974.
Barnhart KT et al. The effect of dehydroepiandrosterone supplementation to symptomatic perimenopausal women on serum endocrine profiles, lipid parameters, and health-related quality of life. J Clin Endocr Metab 1999; 84: 3896–3902.
Parker Jr CR et al. Effects of aging on adrenal function in the human: responsiveness and sensitivity of adrenal androgens and cortisol to adrenocorticotropin in premenopausal and postmenopausal women. J Clin Endocr Metab 2000; 85: 48–54.
Longcope C . Adrenal and gonadal androgen secretion in normal females. Clin Endocr Metab 1986; 15: 213–228.
Nappi RE et al. Serum allopregnanolone relate to FSFI score during the menstrual cycle. J Sex Marital Ther 2003; 29: 95–102.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Turna, B., Apaydin, E., Semerci, B. et al. Women with low libido: correlation of decreased androgen levels with female sexual function index. Int J Impot Res 17, 148–153 (2005). https://doi.org/10.1038/sj.ijir.3901294
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijir.3901294
Keywords
This article is cited by
-
Neuroendocrinology of sexual behavior
International Journal of Impotence Research (2022)
-
Not All Progestins are Created Equally: Considering Unique Progestins Individually in Psychobehavioral Research
Adaptive Human Behavior and Physiology (2020)
-
Effect of a multi-ingredient based food supplement on sexual function in women with low sexual desire
BMC Women's Health (2019)
-
Do Psychosocial Factors Moderate the Relation between Testosterone and Female Sexual Desire? The Role of Interoception, Alexithymia, Defense Mechanisms, and Relationship Status
Adaptive Human Behavior and Physiology (2019)
-
Androgen receptor gene polymorphism and sexual function in midlife women
Archives of Gynecology and Obstetrics (2019)