Abstract
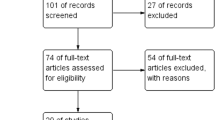
The present paper aims to analyze and discuss the available evidence supporting the relationship between male sexual function and elevated prolactin (PRL) levels (HPRL). Two different sources of data were analyzed. Clinical data were derived from a series of patients seeking medical care for sexual dysfunction at our Unit. Out of 418 studies, 25 papers were used with a meta-analytic approach to evaluate the overall prevalence of HPRL in patients with erectile dysfunction (ED) and to study the influence of HPRL and its treatment on male sexual function. Among 4215 patients (mean age 51.6 ± 13.1 years) consulting for sexual dysfunction at our Unit, 176 (4.2%) showed PRL levels above the normal range. Meta-analytic data showed that HPRL is a rare condition among patients with ED (2 [1;3]%). Either clinical and meta-analytic data confirm a stepwise negative influence of PRL on male sexual desire (S = 0.00004 [0.00003; 0.00006]; I = −0.58915 [−0.78438; −0.39392]; both p < 0.0001 from meta-regression analysis). Normalization of PRL levels is able to improve libido. The role of HPRL in ED remains inconclusive. Data from a meta-analytic approach showed that either HPRL or reduced T levels were independently associated with ED rates. The normalization of PRL levels only partially restored ED. HPRL did not significantly contribute to ED severity, in our clinical setting. In conclusion, treating HPRL can restore normal sexual desire, whereas its effect on erection is limited.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Lotti F, Corona G, Maseroli E, Rossi M, Silverii A, Degl’innocenti S, et al. Clinical implications of measuring prolactin levels in males of infertile couples. Andrology. 2013;1:764–71.
Bachelot A, Binart N. Reproductive role of prolactin. Reproduction. 2007;133:361–9.
Corona G, Rastrelli G, Comeglio P, Guaraldi F, Mazzatenta D, Sforza A, et al. The metabolic role of prolactin: systematic review, meta-analysis and preclinical considerations. Expert Rev Endocrinol Metab. 2022;17:533–45.
Wennbo H, Kindblom J, Isaksson OG, Törnell J. Transgenic mice overexpressing the prolactin gene develop dramatic enlargement of the prostate gland. Endocrinology. 1997;138:4410–5.
Corona G, Rastrelli G, Boddi V, Monami M, Melani C, Balzi D, et al. Prolactin levels independently predict major cardiovascular events in patients with erectile dysfunction. Int J Androl. 2011;34:217–24.
Corona G, Mannucci E, Fisher AD, Lotti F, Ricca V, Balercia G, et al. Effect of hyperprolactinemia in male patients consulting for sexual dysfunction. J Sex Med. 2007;4:1485–93.
Corona G, Rastrelli G, Ricca V, Jannini EA, Vignozzi L, Monami M, et al. Risk factors associated with primary and secondary reduced libido in male patients with sexual dysfunction. J Sex Med. 2013;10:1074–89.
Salonia A, Bettocchi C, Boeri L, Capogrosso P, Carvalho J, Cilesiz NC, et al. European Association of Urology guidelines on sexual and reproductive health-2021 update: male sexual dysfunction. Eur Urol. 2021;80:333–57.
Corona G, Wu FC, Rastrelli G, Lee DM, Forti G, O’Connor DB, et al. Low prolactin is associated with sexual dysfunction and psychological or metabolic disturbances in middle-aged and elderly men: the European Male Aging Study (EMAS). J Sex Med. 2014;11:240–53.
Garcia A, Herbon L, Barkan A, Papavasiliou S, Marshall JC. Hyperprolactinemia inhibits gonadotropin-releasing hormone (GnRH) stimulation of the number of pituitary GnRH receptors. Endocrinology. 1985;117:954–9.
Lookingland KJ, Moore KE. Effects of estradiol and prolactin on incertohypothalamic dopaminergic neurons in the male rat. Brain Res. 1984;323:83–91.
Burnett AL, Nehra A, Breau RH, Culkin DJ, Faraday MM, Hakim LS, et al. Erectile dysfunction: AUA guideline. J Urol. 2018;200:633–41.
Corona G, Cucinotta D, Di Lorenzo G, Ferlin A, Giagulli VA, Gnessi L, et al. The Italian Society of Andrology and Sexual Medicine (SIAMS), along with ten other Italian Scientific Societies, guidelines on the diagnosis and management of erectile dysfunction. J Endocrinol Invest. 2023;46:1241–74.
Johri AM, Heaton JP, Morales A. Severe erectile dysfunction is a marker for hyperprolactinemia. Int J Impot Res. 2001;13:176–82.
Krysiak R, Okopień B. Sexual functioning in hyperprolactinemic patients treated with cabergoline or bromocriptine. Am J Ther. 2019;26:e433–e40.
Johnson AR 3rd, Jarow JP. Is routine endocrine testing of impotent men necessary? J Urol. 1992;147:1542–3.
Corona G, Ricca V, Bandini E, Mannucci E, Petrone L, Fisher AD, et al. Association between psychiatric symptoms and erectile dysfunction. J Sex Med. 2008;5:458–68.
Corona G, Mannucci E, Petrone L, Fisher AD, Balercia G, De Scisciolo G, et al. Psychobiological correlates of delayed ejaculation in male patients with sexual dysfunctions. J Androl. 2006;27:453–8.
Petrone L, Mannucci E, Corona G, Bartolini M, Forti G, Giommi R, et al. Structured interview on erectile dysfunction (SIEDY): a new, multidimensional instrument for quantification of pathogenetic issues on erectile dysfunction. Int J Impot Res. 2003;15:210–20.
Rosen RC, Cappelleri JC, Smith MD, Lipsky J, Peña BM. Development and evaluation of an abridged, 5-item version of the International Index of Erectile Function (IIEF-5) as a diagnostic tool for erectile dysfunction. Int J Impot Res. 1999;11:319–26.
Carter JN, Tyson JE, Tolis G, Van Vliet S, Faiman C, Friesen HG. Prolactin-screening tumors and hypogonadism in 22 men. N Engl J Med. 1978;299:847–52.
Franks S, Jacobs HS, Martin N, Nabarro JD. Hyperprolactinaemia and impotence. Clin Endocrinol (Oxf). 1978;8:277–87.
Laufer N, Yaffe H, Margalioth EJ, Livshin J, Ben-David M, Schenker JG. Effect of bromocriptine treatment on male infertility associated with hyperprolactinemia. Arch Androl. 1981;6:343–6.
Prescott RW, Johnston DG, Kendall-Taylor P, Crombie A, Hall K, McGregor A, et al. Hyperprolactinaemia in men-response to bromocriptine therapy. Lancet. 1982;1:245–8.
Johnston DG, Prescott RW, Kendall-Taylor P, Hall K, Crombie AL, Hall R, et al. Hyperprolactinemia: long-term effects of bromocriptine. Am J Med. 1983;75:868–74.
Spark RF, Wills CA, O’Reilly G, Ransil BJ, Bergland R. Hyperprolactinaemia in males with and without pituitary macroadenomas. Lancet. 1982;2:129–32.
Weizman A, Weizman R, Hart J, Maoz B, Wijsenbeek H, Ben, et al. The correlation of increased serum prolactin levels with decreased sexual desire and activity in elderly men. J Am Geriatr Soc. 1983;31:485–8.
Buvat J, Lemaire A, Buvat-Herbaut M, Fourlinnie JC, Racadot A, Fossati P. Hyperprolactinemia and sexual function in men. Horm Res. 1985;22:196–203.
Maatman TJ, Montague DK. Routine endocrine screening in impotence. Urology. 1986;27:499–502.
el-Beheiry A, Souka A, el-Kamshoushi A, Hussein S, el-Sabah K. Hyperprolactinemia and impotence. Arch Androl. 1988;21:211–4.
Leonard MP, Nickel CJ, Morales A. Hyperprolactinemia and impotence: why, when and how to investigate. J Urol. 1989;142:992–4.
Akpunonu BE, Mutgi AB, Federman DJ, York J, Woldenberg LS. Routine prolactin measurement is not necessary in the initial evaluation of male impotence. J Gen Intern Med. 1994;9:336–8.
Colao A, De Rosa M, Sarnacchiaro F, Di Sarno A, Landi ML, Iervolino E, et al. Chronic treatment with CV 205–502 restores the gonadal function in hyperprolactinemic males. Eur J Endocrinol. 1996;135:548–52.
Buvat J, Lemaire A. Endocrine screening in 1,022 men with erectile dysfunction: clinical significance and cost-effective strategy. J Urol. 1997;158:1764–7.
De Rosa M, Colao A, Di Sarno A, Ferone D, Landi ML, Zarrilli S, et al. Cabergoline treatment rapidly improves gonadal function in hyperprolactinemic males: a comparison with bromocriptine. Eur J Endocrinol. 1998;138:286–93.
Wolfsberger S, Czech T, Vierhapper H, Benavente R, Knosp E. Microprolactinomas in males treated by transsphenoidal surgery. Acta Neurochir (Wien). 2003;145:935–40.
Venetikou MS, Lambou T, Gizani D. Hyperprolactinaemia due to hypothalamic-pituitary disease or drug-induced in patients with erectile dysfunction. Andrologia. 2008;40:240–4.
Arduc A, Gokay F, Isik S, Ozuguz U, Akbaba G, Tutuncu Y, et al. Retrospective comparison of cabergoline and bromocriptine effects in hyperprolactinemia: a single center experience. J Endocrinol Invest. 2015;38:447–53.
Shimon I, Hirsch D, Tsvetov G, Robenshtok E, Akirov A, Fraenkel M, et al. Hyperprolactinemia diagnosis in elderly men: a cohort of 28 patients over 65 years. Endocrine. 2019;65:656–61.
Green KI, Amadi C. Status of serum prolactin levels among male cohort in infertile couples. Int J Appl Basic Med Res. 2020;10:245–51.
Su WJ, Cai HC, Yang GC, He KJ, Wu HL, Yang YB, et al. Transsphenoidal surgery for prolactinomas in male patients: a retrospective study. Asian J Androl. 2023;25:113–8.
Cacioppo S, Bianchi-Demicheli F, Frum C, Pfaus JG, Lewis JW. The common neural bases between sexual desire and love: a multilevel kernel density fMRI analysis. J Sex Med. 2012;9:1048–54.
Stoléru S, Fonteille V, Cornélis C, Joyal C, Moulier V. Functional neuroimaging studies of sexual arousal and orgasm in healthy men and women: a review and meta-analysis. Neurosci Biobehav Rev. 2012;36:1481–509.
Bridges RS, Grattan DR. 30 years after: CNS actions of prolactin: sources, mechanisms and physiological significance. J Neuroendocrinol. 2019;31:e12669.
Farrar VS, Harris RM, Austin SH, Nava Ultreras BM, Booth AM, Angelier F, et al. Prolactin and prolactin receptor expression in the HPG axis and crop during parental care in both sexes of a biparental bird (Columba livia). Gen Comp Endocrinol. 2022;315:113940.
Kokay IC, Wyatt A, Phillipps HR, Aoki M, Ectors F, Boehm U, et al. Analysis of prolactin receptor expression in the murine brain using a novel prolactin receptor reporter mouse. J Neuroendocrinol. 2018;30:e12634.
Georgescu T, Ladyman SR, Brown RSE, Grattan DR. Acute effects of prolactin on hypothalamic prolactin receptor expressing neurones in the mouse. J Neuroendocrinol. 2020;32:e12908.
Kamesh A, Black EAE, Ferguson AV. The subfornical organ: a novel site for prolactin action. J Neuroendocrinol. 2018;30:e12613.
Ozerdogan N, Mizrak Sahin B, Gursoy E, Zeren F. Sexual dysfunction in the third trimester of pregnancy and postpartum period: a prospective longitudinal study. J Obstet Gynaecol. 2022;42:2722–8.
Asratie MH, Andualem Z. Predictors of early resumption of post-partum sexual intercourse among post-partum period women in Ethiopia: a multilevel analysis based on Ethiopian demographic and health survey 2016. PLoS One. 2022;17:e0271372.
Matthies LM, Wallwiener M, Sohn C, Reck C, Müller M, Wallwiener S. The influence of partnership quality and breastfeeding on postpartum female sexual function. Arch Gynecol Obstet. 2019;299:69–77.
Isidori AM, Aversa A, Calogero A, Ferlin A, Francavilla S, Lanfranco F, et al. Adult- and late-onset male hypogonadism: the clinical practice guidelines of the Italian Society of Andrology and Sexual Medicine (SIAMS) and the Italian Society of Endocrinology (SIE). J Endocrinol Invest. 2022:45:2385–2403.
Lin CS. Phosphodiesterase type 5 regulation in the penile corpora cavernosa. J Sex Med. 2009;6:203–9.
Corona G, Gacci M, Baldi E, Mancina R, Forti G, Maggi M. Androgen deprivation therapy in prostate cancer: focusing on sexual side effects. J Sex Med. 2012;9:887–902.
Corona G, Rastrelli G, Marchiani S, Filippi S, Morelli A, Sarchielli E, et al. Consequences of anabolic-androgenic steroid abuse in males; sexual and reproductive perspective. World J Mens Health. 2021;40:165–78.
Kaplan SA, Chung DE, Lee RK, Scofield S, Te AE. A 5-year retrospective analysis of 5α-reductase inhibitors in men with benign prostatic hyperplasia: finasteride has comparable urinary symptom efficacy and prostate volume reduction, but less sexual side effects and breast complications than dutasteride. Int J Clin Pr. 2012;66:1052–5.
Martínez-Giner G, Giménez-De Llano E, Romero-Rubio D, Abad-Pérez MJ, Sánchez-Martínez V. Sexual dysfunction in people treated with long-acting injectable antipsychotics in monotherapy or polypharmacy: a naturalistic study. Int J Ment Health Nurs. 2022;31:576–90.
Bonete Llácer JM, Martínez Hortelano A, Richart Albelda B. Hyperprolactinemia in psychotic patients treated in monotherapy with long-acting injectable antipsychotics. Int J Psychiatry Clin Pr. 2019;23:189–93.
Zhao YH, Wu ZX, Zhao X, Jiang J, Jiang R. Low androgen level impairs erectile function of rat by regulating the Ng/CaN/AKT/eNOS pathway in penile corpus cavernosum. Andrology. 2022;10:1189–96.
Bivalacqua TJ, Champion HC, Usta MF, Cellek S, Chitaley K, Webb RC, et al. RhoA/Rho-kinase suppresses endothelial nitric oxide synthase in the penis: a mechanism for diabetes-associated erectile dysfunction. Proc Natl Acad Sci USA 2004;101:9121–6.
Vignozzi L, Morelli A, Filippi S, Ambrosini S, Mancina R, Luconi M, et al. Testosterone regulates RhoA/Rho-kinase signaling in two distinct animal models of chemical diabetes. J Sex Med. 2007;4:620–32.
Yildirim MK, Yildirim S, Utkan T, Sarioglu Y, Yalman Y. Effects of castration on adrenergic, cholinergic and nonadrenergic, noncholinergic responses of isolated corpus cavernosum from rabbit. Br J Urol. 1997;79:964–70.
Reilly CM, Stopper VS, Mills TM. Androgens modulate the alpha-adrenergic responsiveness of vascular smooth muscle in the corpus cavernosum. J Androl. 1997;18:26–31.
Wang XJ, Xu TY, Xia LL, Zhong S, Zhang XH, Zhu ZW, et al. Castration impairs erectile organ structure and function by inhibiting autophagy and promoting apoptosis of corpus cavernosum smooth muscle cells in rats. Int Urol Nephrol. 2015;47:1105–15.
Hu Z, Zhang Y, Chen J, Luo M, Wang N, Xiao Y, et al. Testosterone attenuates senile cavernous fibrosis by regulating TGFβR1 and galectin-1 signaling pathways through miR-22-3p. Mol Cell Biochem. 2022. https://doi.org/10.1007/s11010-022-04641-8.
Corona G, Rastrelli G, Vignozzi L, Maggi M. Androgens and male sexual function. Best Pr Res Clin Endocrinol Metab. 2022;36:101615.
Boloña ER, Uraga MV, Haddad RM, Tracz MJ, Sideras K, Kennedy CC, et al. Testosterone use in men with sexual dysfunction: a systematic review and meta-analysis of randomized placebo-controlled trials. Mayo Clin Proc. 2007;82:20–8.
Isidori AM, Giannetta E, Gianfrilli D, Greco EA, Bonifacio V, Aversa A, et al. Effects of testosterone on sexual function in men: results of a meta-analysis. Clin Endocrinol (Oxf). 2005;63:381–94.
Elliott J, Kelly SE, Millar AC, Peterson J, Chen L, Johnston A, et al. Testosterone therapy in hypogonadal men: a systematic review and network meta-analysis. BMJ Open. 2017;7:e015284.
Ponce OJ, Spencer-Bonilla G, Alvarez-Villalobos N, Serrano V, Singh-Ospina N, Rodriguez-Gutierrez R, et al. The efficacy and adverse events of testosterone replacement therapy in hypogonadal men: a systematic review and meta-analysis of randomized, placebo-controlled trials. J Clin Endocrinol Metab. 2018. https://doi.org/10.1210/jc.2018-00404.
Zhu J, Zhang W, Ou N, Song Y, Kang J, Liang Z, et al. Do testosterone supplements enhance response to phosphodiesterase 5 inhibitors in men with erectile dysfunction and hypogonadism: a systematic review and meta-analysis. Transl Androl Urol. 2020;9:591–600.
Algeffari M, Jayasena CN, MacKeith P, Thapar A, Dhillo WS, Oliver N. Testosterone therapy for sexual dysfunction in men with type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Diabet Med. 2018;35:195–202.
Cozzi R, Ambrosio MR, Attanasio R, Battista C, Bozzao A, Caputo M, et al. Italian Association of Clinical Endocrinologists (AME) and International Chapter of Clinical Endocrinology (ICCE). Position statement for clinical practice: prolactin-secreting tumors. Eur J Endocrinol. 2022;186:P1–33.
Duckles SP, Miller VM. Hormonal modulation of endothelial NO production. Pflug Arch. 2010;459:841–51.
Molinari C, Grossini E, Mary DA, Uberti F, Ghigo E, Ribichini F, et al. Prolactin induces regional vasoconstriction through the beta2-adrenergic and nitric oxide mechanisms. Endocrinology. 2007;148:4080–90.
Montes de Oca P, Macotela Y, Nava G, López-Barrera F, de la Escalera GM, Clapp C. Prolactin stimulates integrin-mediated adhesion of circulating mononuclear cells to endothelial cells. Lab Invest. 2005;85:633–42.
Rosen RC, Riley A, Wagner G, Osterloh IH, Kirkpatrick J, Mishra A. The international index of erectile function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology. 1997;49:822–30.
Romano L, Pellegrino R, Sciorio C, Barone B, Gravina AG, Santonastaso A, et al. Erectile and sexual dysfunction in male and female patients with celiac disease: a cross-sectional observational study. Andrology. 2022;10:910–8.
Mistretta FA, de Cobelli O, Verze P, Botticelli F, Jannello L, Luzzago S, et al. A comprehensive evaluation of sexual and reproductive outcomes following robot-assisted retroperitoneal lymph node dissection for nonseminomatous germ cell tumor. Asian J Androl. 2022;24:579–83.
D’Andrea S, Minaldi E, Castellini C, Cavallo F, Felzani G, Francavilla S, et al. Independent association of erectile dysfunction and low testosterone levels with life dissatisfaction in men with chronic spinal cord injury. J Sex Med. 2020;17:911–8.
Xi Y, Colonnello E, Ma G, Limoncin E, Ciocca G, Zhang H, et al. Validity of erectile function assessment questionnaires in premature ejaculation patients: a comparative study between the abridged forms of the international index of erectile function and proposal for optimal cutoff redefinition. J Sex Med. 2021;18:440–7.
Funding
The paper was partially supported by an unrestricted grant “PE8 -Conseguenze e sfidedell’invecchiamento -AGE-IT –CUP B83C22004800006.
Author information
Authors and Affiliations
Contributions
Each author has contributed sufficiently to the intellectual content of the submission. In particular, MM and GC conceived and/or designed the work; AF, NB, and CS acquired data, GR, GC, LV, and MM played an important role in interpreting the results; GC and MM drafted or revised the manuscript. All authors approved the final version. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Corona, G., Rastrelli, G., Bianchi, N. et al. Hyperprolactinemia and male sexual function: focus on erectile dysfunction and sexual desire. Int J Impot Res (2023). https://doi.org/10.1038/s41443-023-00717-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41443-023-00717-1
This article is cited by
-
Effects of cabergoline and dimethylcabergoline on the sexual behavior of male rats
Psychopharmacology (2023)