Abstract
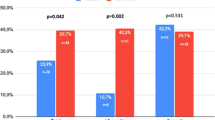
Trials of the efficacy and safety of vardenafil in the treatment of male erectile dysfunction (ED) were meta-analysed. All available databases were searched (January 1, 2001–November 30, 2003). Trials were eligible if they included men with ED, compared vardenafil with placebo, were randomized, were at least of 12 weeks duration, and assessed clinically relevant outcomes. Two reviewers independently evaluated study quality and extracted data in a standardized fashion. Nine trials (6809 men) met the inclusion criteria. In results pooled from seven fixed-dose trials, vardenafil increases the Erectile Function domain of the International Index of Erectile Function questionnaire by 6.18 units (weighted mean difference (WMD)). Vardenafil also increases the percentage of erections firm enough to allow vaginal penetration (WMD: 26) and the percentage of sexual attempts that were successful per participant (WMD: 29.8). The percentage of men agreeing with the statement that ‘the treatment they have been taking over the past 4 weeks improved their erections’, is also in favour of vardenafil (relative risk (RR): 3). These efficacy variables appeared greater at higher doses, although there are no significant differences between 10 and 20 mg dose. The same results were extracted for the two flexible ‘as needed’ dosing trials. Discontinuations are greater at the vardenafil groups compared to placebo (RR: 2.25). Specific adverse events with vardenafil included flushing, dyspepsia, headache, and rhinitis. Vardenafil was not significantly associated with serious cardiovascular events or death. Vardenafil, in all treatment regimens, shows to possess superior efficacy to placebo in the treatment of patients with erectile dysfunction. More data is needed on patients' subgroups.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
NIH Consensus Development Panel on Impotence. Impotence. JAMA 1993; 270: 83–90.
Montague DK et al. Clinical guidelines panel on erectile dysfunction: summary report on the treatment of organic erectile dysfunction. J Urol 1996; 156: 2007–2011.
Benet AE, Melman A . The epidemiology of erectile dysfunction. Urol Clin North Am 1995; 22: 699–709.
Feldman HA et al. Impotence and its medical and psychosocial correlates: results of the Massachusetts Male Aging Study. J Urol 1994; 151: 54–61.
Andersson K-E, Wagner G . Physiology of penile erection. Physiol Rev 1995; 75: 191–236.
Burnett AL . Role of nitric oxide in the physiology of erection. Biol Reprod 1995; 52: 485–489.
Burnett AL . Nitric oxide in the penis: physiology and pathology. J Urol 1997; 157: 320–324.
Ignarro LJ et al. Mechanism of vascular smooth muscle relaxation by organic nitrates, nitrites, nitroprusside and nitric oxide: evidence for the involvement of S-nitrosothiols as active intermediates. J Pharmacol Exp Ther 1981; 218: 739–749.
Lugnier C, Komas N . Modulation of vascular cyclic nucleotide phosphodiesterases by cyclic GMP: role in vasodilatation. Eur Heart J 1993; 14(Suppl 1): 141–148.
Boolell M et al. Sildenafil: an orally active type 5 cyclic GMP-specific phosphodiesterase inhibitor for the treatment of penile erectile dysfunction. Int J Impot Res 1996; 8: 47–52.
Klotz T et al. Vardenafil increases penile rigidity and tumescence in erectile dysfunction patients: a RigiScan and pharmacokinetic study. World J Urol 2001; 19: 32–39.
Eardley I, Cartledge J . Tadalafil (Cialis) for men with erectile dysfunction. Int J Clin Pract 2002; 56: 300–304.
Rosen RC et al. The International Index of Erectile Function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology 1997; 49: 822–830.
Jadad AR et al. Assessing the quality of reports on randomized clinical trials: is blinding necessary? Control Clin Trials 1996; 17: 1–12.
Montori VM, Guyatt GH . Intention-to-treat principle. Can Med Assoc J 2001; 165: 1339–1341.
Review Manager (RevMan) [Computer program]. Version 4.2 for Windows. The Cochrane Collaboration: Oxford, England, 2002.
Moher D et al. Improving the quality of reports of meta-analyses of randomised controlled trials: the QUOROM statement. Quality of Reporting of Meta-analyses. Lancet 1999; 354: 1896–1900.
Porst H et al. The efficacy and tolerability of vardenafil, a new, oral, selective phosphodiesterase type 5 inhibitor, in patients with erectile dysfunction: the first at-home clinical trial. Int J Impot Res 2001; 13: 192–199.
Hellstrom WJ et al. Vardenafil for treatment of men with erectile dysfunction: efficacy and safety in a randomized, double-blind, placebo-controlled trial. J Androl 2002; 23: 763–771.
Goldstein I et al. Vardenafil, a new phosphodiesterase type 5 inhibitor, in the treatment of ED in men with diabetes. Diabetes Care 2003; 26: 777–783.
Brock G et al. Safety and efficacy of vardenafil for the treatment of men with erectile dysfunction after radical retropubic prostatectomy. J Urol 2003; 170: 1278–1283.
Porst H, Young JM, Schmidt AC, Buvat J . Efficacy and tolerability of vardenafil for treatment of ED in patients subgroups. Urology 200; 362: 519–524.
Montorsi F et al. Reliable efficacy over time of vardenafil, a potent, highly selective PDE-5 inhibitor in men with erectile dysfunction: a retrospective analysis of two pivotal phase III studies. Second International Consultation on Erectile and Sexual Dysfunctions. 28 June–1 July##2003 Paris.
Hatzichristou D, Lording D . Vardenafil in efficacious in men with erectile dysfunction unresponsive to prior Sildenafil therapy: results of a phase-III clinical trial—patient response with vardenafil in sildenafil nonresponders (PROVEN). Int J Impot Res 2003; 15(Suppl 6): S5.
Porst H, Padma-Nathan H, Thibonnier M, Eardley I . Efficacy and safety of vardenafil, a selective phosphodoesterase 5 inhibitor in men with ED on antihypertensive therapy. Eur Urol 2001; 1(Suppl 1): 152.
Hatzichristou D et al. A flexible dose regimen of vardenafil for erectile dysfunction: a placebo-controlled trial. Eur Urol 2003; 1(Suppl 2): 176.
US Food and Drug Administration. Web site Center for Drug Evaluation and Research. Available at http://www.fda.gov/cder/foi/nda/2003/21-400_Levitra.htm. Assessed October 30 2003.
Perimenis P et al. Switching from long-term treatment with self-injections to oral sildenafil in diabetic patients with severe erectile dysfunction. Eur Urol 2002; 41: 387–391.
Saenz de Tejada I et al. The phosphodiesterase inhibitory selectivity and the in vitro and in vivo potency of the new PDE5 inhibitor vardenafil. Int J Impot Res 2001; 13: 282–290.
Bischoff E, Schneider K . A conscious-rabbit model to study vardenafil hydrochloride and other agents that influence penile erection. Int J Impot Res 2001; 13: 230–235.
Kim NN et al. Inhibition of cyclic GMP hydrolysis in human corpus cavernosum smooth muscle cells by vardenafil, a novel, selective phosphodiesterase type 5 inhibitor. Life Sci 2001; 69: 2249–2256.
Juni P, Rutjes AW, Dieppe PA . Are selective COX 2 inhibitors superior to traditional non steroidal anti-inflammatory drugs? BMJ 2002; 324: 1287–1288.
Egger M, Davey Smith G, O'Rourke K . Rationale, potentials and promise of systematic reviews. In: Egger M, Davey Smith G, Altman DG (eds). Systematic Reviews in Health Care: Meta-Analysis in Context. BMJ Books: London, 2001 pp 3–19.
Perimenis P et al. A comparative, crossover study of the efficacy and safety of sildenafil and apomorphine in men with evidence of arteriogenic erectile dysfunction. Int J Impot Res 2004; 16: 2–7.
Acknowledgements
We thank Matthias Egger and Peter Juni for their assistance and suggestions and Artemis Athanasiou for her assistance in literature collection.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Markou, S., Perimenis, P., Gyftopoulos, K. et al. Vardenafil (Levitra) for erectile dysfunction: a systematic review and meta-analysis of clinical trial reports. Int J Impot Res 16, 470–478 (2004). https://doi.org/10.1038/sj.ijir.3901258
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijir.3901258
Keywords
This article is cited by
-
The application of quantitative methods for identifying and exploring the presence of bias in systematic reviews: PDE-5 inhibitors for erectile dysfunction
International Journal of Impotence Research (2008)
-
A meta-regression analysis of treatment effect modifiers in trials with flexible-dose oral sildenafil for erectile dysfunction in broad-spectrum populations
International Journal of Impotence Research (2006)
-
Indirect comparison of interventions using published randomised trials: systematic review of PDE-5 inhibitors for erectile dysfunction
BMC Urology (2005)