Abstract
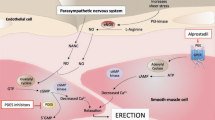
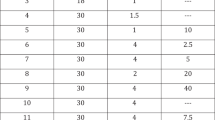
This open-label, dose-escalation study investigated the efficacy and safety of alprostadil (PGE1, prostaglandin E1) Sterile Powder (S.Po.) (Caverject®) for treatment of erectile dysfunction (ED) in 84 men with ED of various etiologies lasting ≥4 months. Doses started with 2.5 µg, then 5 µg, 10, 15, 20, 30, up to a 40 µg maximum. Eligible patients received single alprostadil injections in the physician’s office until an erection occurred. Ten minutes after injection, the patient’s erection was clinically evaluated. Optimal response was defined as erection sufficient to permit vaginal penetration and lasting 30–60 min. The patient also reported his own evaluation of response and any side effects. Patients were 24–65 y old (mean: 43.7 y), had ED of psychogenic, vascular, or neurogenic origin lasting 4 months–30 y (mean: 3.75 y). Of 84 patients enrolled, 82 completed the study. In the 82 patients who completed the study 78 (92.9%) achieved an optimal response; 18/78 patients (23.1%) had an optimal response at 2.5 µg, 9/78 (11.5%) at 5 µg, 21/78 (26.9%) at 10 µg, 12/78 (15.4%) at 15 µg, and 11/78 (14.1%) at 20 µg. Only 5/78 (6.4%) at 30 µg and 2/78 (2.6%) at 40 µg achieved an optimal response. Mean optimal alprostadil dose was 11.9 µg, and the mean minimal effective dose was 9.9 µg. Mean onset of erection was 11.2 min; mean duration of erection was 50.5 min. Penile pain in five patients (6%) was the only reported side effect.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Choi, H., Adimoelja, A., Kim, S. et al. A dose-response study of alprostadil sterile powder (S.Po.) (Caverject®) for the treatment of erectile dysfunction in Korean and Indonesian men. Int J Impot Res 9, 47–51 (1997). https://doi.org/10.1038/sj.ijir.3900264
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijir.3900264
Keywords
This article is cited by
-
Intracavernosal alprostadil is effective for the treatment of erectile dysfunction in diabetic men
International Journal of Impotence Research (2001)