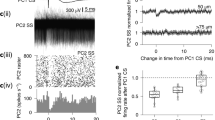
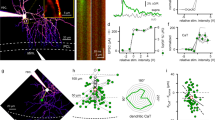
Abstract
IN the classical view, a central neuron integrates incoming synaptic information by simple algebraic summation of the resultant bioelectrical signals that coincide in time. The voltage dependence of the NMDA (N-methyl-D-aspartate) type of iono-tropic glutamate receptor endows neurons with an additional tool that allows one synaptic input to influence another, providing, again, that the two are active simultaneously1. Here we identify a new mechanism by which non-coincident signals generated by different synaptic inputs are integrated. The device serves to regulate neuronal excitation through G-protein-coupled, metabotropic glutamate receptors (mGluRs)2 in a powerful and specific manner. We show that, in cerebellar Purkinje cells, a single activation of the climbing–fibre input markedly potentiatesmGluR-mediated excitation at parallel–fibre synapses3. The potentiation results from a transient rise in cytosolic Ca2+ which is 'memorized' in such a way that it promotes excitation through mGluRs for about two minutes. A Ca2+ -transient is also effective if imposed up to two seconds after parallel-fibre stimulation. By allowing temporally and spatially dispersed synaptic signals to be assimilated, this mechanism adds a new element to the computational power of central neurons.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bliss, T. V. P. & Collingridge, G. L. Nature 361, 31–39 (1993).
Tanabe, Y., Masu, M., Ishii, T., Shigemoto, R. & Nakanishi, S. Neuron 8,169–179 (1992).
Batchelor, A. M., Madge, D. J. & Garthwaite, J. Neuroscience 63, 911–915 (1994).
Llano, I., Marty, A., Armstrong, C. M. & Konnerth, A. J. Physiol. (Lond.) 434,183–213 (1991).
Conquet, F. et al. Nature 372, 237–243 (1994).
Masu, M., Tanabe, Y., Tsuchida, K., Shigemoto, R. & Nakanishi, S. Nature 349, 760–765 (1991).
Berridge, M. J. Nature 361, 315–325 (1993).
Staub, C., Vranesic, I. & Knöpfel, T. Eur. J. Neurosci. 4, 832–839 (1992).
Merrill, E. G., Wall, P. D. & Yaksh, T. L. J. Physiol. (Lond.) 284,127–145 (1978).
Eccles, J. C., Ito, M. & Szentágothai, J. The Cerebellum as a Neuronal Machine (Springer, Berlin, 1967).
Llinás, R. & Sugimori, M. J. Physiol. (Lond.) 305,171–195 (1980).
Ross, W. N. & Werman, R. J. Physiol. (Lond.) 389, 319–336 (1987).
Ellis-Davies, G. C. R. & Kaplan, J. H. Proc. Natl Acad. Sci. USA 91,187–191 (1994).
Abe, T. et al. J. Biol. Chem. 267,13361–13368 (1992).
Miyakawa, H., Lev-Ram, V., Lasser-Ross, N. & Ross, W. N. J. Neurophysiol. 68, 1178–1189 (1992).
Sugimori, M. & Llinás, R. R. Proc. Natl Acad. Sci. USA 87, 5084–5088 (1990).
Huang, K. -P. Trends Neurosci. 12, 425–432 (1989).
Marsh, S. J., Trouslard, J., Leaney, J. L. & Brown, D. A. Neuron 15, 729–737 (1995).
Eilers, J., Augustine, G. J. & Konnerth, A. Nature 373,155–158 (1995).
Albus, J. S. Math. Biosci. 10, 25–61 (1971).
Marr, D. A. J. Physiol. (Lond.) 202, 437–470 (1969).
Ito, M. Annu. Rev. Neurosci. 12, 85–102 (1989).
Sakurai, M. Proc. Natl Acad. Sci. USA 87, 3383–3385 (1990).
Garthwaite, J. & Batchelor, A. M. J. Neurosci. Meth. 64, 189–197 (1996).
Batchelor, A. M. & Garthwaite, J. Neuropharmacol. J. 32, 11–20 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Batchelor, A., Garthwaite, J. Frequency detection and temporally dispersed synaptic signal association through a metabotropic glutamate receptor pathway. Nature 385, 74–77 (1997). https://doi.org/10.1038/385074a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/385074a0
This article is cited by
-
Electrophysiological and Imaging Analysis of GFP-Tagged Protein Kinase C γ Translocation in Cerebellar Purkinje Cells
The Cerebellum (2022)
-
Group I metabotropic glutamate receptors in the primate motor thalamus: subsynaptic association with cortical and sub-cortical glutamatergic afferents
Brain Structure and Function (2019)
-
Synapse elimination in the developing cerebellum
Cellular and Molecular Life Sciences (2013)
-
Spatiotemporal firing patterns in the cerebellum
Nature Reviews Neuroscience (2011)
-
Role of the metabotropic glutamate receptor subtype 1 in the Harmaline-induced tremor in rats
Journal of Neural Transmission (2009)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.