Abstract
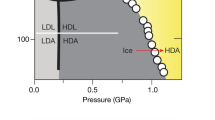
THE discovery1 in 1984 that an ice crystal can be transformed by pressure to an amorphous phase has since been followed by other examples of pressure-induced amorphization2. This transition, like melting, involves loss of long-ranged order, prompting the question of whether the two transitions are related. Here I describe experiments probing this relationship for a form of crystalline ice (denoted Ih) which is melted and amorphized by pressure. To avoid the complication of crystal–crystal transformations interrupting the melting process I use an ice emulsion, in which the very small particle size (about 5μm) suppresses nucleation of other crystal phases. As the temperature is decreased, I see a smooth crossover from (pressure-induced) equilibrium melting to sluggish amorphization at around 140–165 K. In this temperature range, ice Ih became 'supercompressed' before melting to a highly viscous liquid which seemed to be related to an imperfectly relaxed amorphous ice. Below about 140 K, ice Ih was transformed to an unrelaxed phase apparently related to the high-density amorphous form of ice. This sequence of transitions can be viewed as a crossover from a two-phase melting process (which is determined by the relative free energies of the solid and liquid phases) towards a one-phase amorphization process (where the transition is induced by a mechanical instability limit of the solid).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Mishima, O., Calvert, L. D. & Whalley, E. Nature 310, 393–395 (1984).
Wolf, G. H. et al., in High-Pressure Research: Application to Earth and Planetary Sciences (eds Syono, Y. & Manghnani, M. H.) 503–517 (Terra Science, Tokyo/Am. Geophys. Union, Washington DC, 1992).
Tse, J. S. & Klein, M. L. J. Chem. Phys. 92, 3992–3994 (1990).
Sciortino, F. et al. Phys. Rev. E 52, 6484–6491 (1995).
Kanno, H., Speedy, R. J. & Angell, C. A. Science 189, 880–881 (1975).
Handa, Y. P., Mishima, O. & Whalley, E. J. Chem. Phys. 84, 2766–2770 (1986).
Johari, G. P., Hallbrucker, A. & Mayer, E. J. Phys. Chem. 94, 1212–1214 (1990).
Mishima, O., Calvert, L. D. & Whalley, E. Nature 314, 76–78 (1985).
Mishima, O. J. Chem. Phys. 100, 5910–5912 (1994).
Angell, C. A. Science 267, 1924–1935 (1995).
Lüdemann, H.-D. Ber. Bunsenges. Phys. Chem. 94, 325–332 (1990).
Johnson, W. L. Prog. Mater. Sci. 30, 81–134 (1986).
Cahn, R. W. & Johnson, W. L. J. Mater. Res. 1, 724–732 (1986).
Wolf, D., Okamoto, P. R., Yip, S., Lutsko, J. F. & Kluge, M. J. Mater. Res. 5, 286–301 (1990).
Fecht, H. J. Mater. Trans. Jpn Inst. Metals 36, 777–793 (1995).
Bertie, J. E., Calvert, L. D. & Whalley, E. J. Chem. Phys. 38, 840–846 (1963).
Handa, Y. P., Klug, D. D. & Whalley, E. Can. J. Chem. 66, 919–924 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mishima, O. Relationship between melting and amorphization of ice. Nature 384, 546–549 (1996). https://doi.org/10.1038/384546a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/384546a0
This article is cited by
-
Observing growth and interfacial dynamics of nanocrystalline ice in thin amorphous ice films
Nature Communications (2024)
-
Atmospheric ice nucleation
Nature Reviews Physics (2023)
-
A new technical approach for preparing frozen biological samples for electron microscopy
Plant Methods (2020)
-
Experimental tests for a liquid-liquid critical point in water
Science China Physics, Mechanics & Astronomy (2020)
-
Hydrophilic and hydrophobic competition in water-methanol solutions
Science China Physics, Mechanics & Astronomy (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.