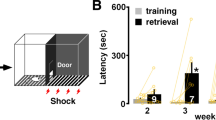
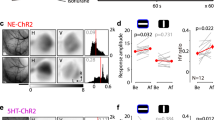
Abstract
LONG-TERM potentiation (LTP) is a lasting enhancement of excitatory synaptic transmission that follows specific patterns of electrical stimulation1. Although the mechanism of LTP has been intensively studied, particularly in the hippocampus, its significance for normal brain function remains unproven. It has been proposed that LTP-like mechanisms may contribute to naturally occurring, experience-dependent synaptic modifications in the visual cortex2-8. The formation of normal binocular connections within the visual cortex requires simultaneous input from both eyes during a postnatal critical period9-12 that can be delayed by rearing animals in complete darkness13,14. To explore the role of LTP in this experience-dependent maturation process, we induced LTP in visual cortical slices taken at different ages from light-reared and dark-reared rats. Susceptibility to LTP coincides with the critical period and, like the critical period, can be prolonged by rearing animals in darkness. These findings support the hypothesis that LTP reflects a normal mechanism of experience-dependent synaptic modification in the developing mammalian brain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bliss, T. V. P. & Collingridge, G. L. Nature 361, 31–39 (1993).
Komatsu, Y., Toyama, K., Maeda, J. & Sakaguchi, H. Neurosci. Lett. 26, 269–274 (1981).
Artola, A. & Singer, W. Nature 330, 649–652 (1987).
Berry, R. L., Teyler, T. J. & Taizhen, H. Brain Res. 481, 221–227 (1989).
Kimura, F., Nishigori, A., Shirokawa, T. & Tsumoto, T. J. Physiol., Lond. 414, 125–144 (1989).
Bear, M. F., Press, W. A. & Connors, B. W. J. Neurophysiol. 67, 1–11 (1992).
Kirkwood, A. & Bear, M. F. J. Neurosci. 14, 1634–1645 (1994).
Frégnac, Y., Burke, J. P., Smith, D. & Friedlander, M. J. J. Neurophysiol. 71, 1403–1421 (1994).
Hubel, D. H. & Wiesel, T. N. J. Physiol., Lond. 206, 419–436 (1970).
Blakemore, C. & vanSluyters, R. C. J. Physiol., Lond. 237, 195–216 (1974).
Olson, C. R. & Freeman, R. D. Expl Brain Res. 39, 17–21 (1980).
Fagiolini, M., Pizzorusso, T., Berardi, N., Domenici, L. & Maffei, L. Vision Res. 34, 709–720 (1994).
Cynader, M. & Mitchel, D. E. J. Neurophysiol. 43, 1026–1039 (1980).
Mower, G. D. Devl Brain Res. 58, 151–158 (1991).
Sherman, M. S. & Spear, P. D. Physiol. Rev. 62, 738–855 (1982).
Domenici, L., Berardi, N., Carmignoto, G., Vantini, G. & Maffei, L. Proc. natn. Acad. Sci. USA. 88, 8811–8815 (1991).
Kirkwood, A., Dudek, S. D., Gold, J. T., Aizenman, C. D. & Bear, M. F. Science 260, 1518–1521 (1993).
Komatsu, Y., Fujii, K., Maeda, J., Sakaguchi, H. & Toyama, K. J. Neurophysiol. 59, 124–141 (1988).
Perkins, A. T. & Teyler, T. J. Brain Res. 439, 222–229 (1988).
Kato, N., Artola, A. & Singer, W. Devl Brain Res. 60, 43–50 (1991).
Stevens, C. F., Tonegawa, S. & Wang, Y. Curr. Biol. 4, 687–693 (1994).
Fox, K., Daw, N., Sato, H. & Czepita, D. Nature 350, 342–344 (1991).
Carmignoto, G. & Vicini, S. Science 258, 1007–1011 (1992).
Berry, R. L., Perkins, T. A. & Teyler, T. J. Brain Res. 628, 99–104 (1993).
Blue, M. E. & Parnavelas, J. G. J. Neurocytol. 12, 697–712 (1983).
Komatsu, Y. Devl Brain Res. 8, 136–139 (1983).
Bode-Gruel, K. M. & Singer, W. Devl Brain Res. 46, 197–204 (1989).
Luhmann, H. J. & Prince, D. A. Devl Brain Res. 54, 287–290 (1990).
Friauf, E. & Shatz, C. J. J. Neurophysiol. 66, 2059–2071 (1991).
Komatsu, Y. J. Neurosci. 14, 6488–6499 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kirkwood, A., Lee, HK. & Bear, M. Co-regulation of long-term potentiation and experience-dependent synaptic plasticity in visual cortex by age and experience. Nature 375, 328–331 (1995). https://doi.org/10.1038/375328a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/375328a0
This article is cited by
-
Nanoscale rules governing the organization of glutamate receptors in spine synapses are subunit specific
Nature Communications (2022)
-
A calcium-based plasticity model for predicting long-term potentiation and depression in the neocortex
Nature Communications (2022)
-
Decreased reproducibility and abnormal experience-dependent plasticity of network dynamics in Fragile X circuits
Scientific Reports (2020)
-
A spike-timing-dependent plasticity rule for dendritic spines
Nature Communications (2020)
-
The Ontogenesis of Mammalian Sleep: Form and Function
Current Sleep Medicine Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.