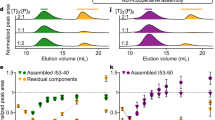
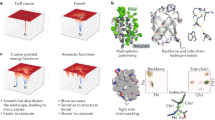
Abstract
THE construction of synthetic molecules that fold or assemble predictably into large, well defined structures represents a fertile area of chemistry. Many supramolecular systems have been reported that self-assemble as a result of non-covalent interactions1-7; and the control of higher-order protein structure by de novo design has also been demonstrated8,9. Protein secondary structural motifs have also been stabilized by incorporating artificial groups that impose constraints on the folded architecture10-12. Here we describe the synthesis of molecules that will fold in water into a pleated structure, as a result of interactions between alternating electron-rich donor groups and electron-deficient acceptor groups. We verify the pleated structure using absorption and NMR spec-troscopy. Donor-acceptor interactions have been used previously to engineer specific supramolecular geometries2,13, and are energetically favourable in organic as well as in aqueous solutions. But whereas previously such interactions have been used to effect self-assembly of distinct molecules, our results show that they can also determine the secondary structure of complex synthetic molecules in solution.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lindsey, J. S. New J. Chem. 15, 153–180 (1991).
Amabilino, D. B. et al. J. Am. chem. Soc. 117, 1271–1294 (1995).
Whitesides, G. M., Mathias, J. P. & Seto, C. T. Science 254, 1312–1319 (1991).
Lehn, J.-M. Angew. Chem. int. Edn engl. 29, 1304–1319 (1990).
Chambron, J. C., Dietrich-Buchecker, C. O., Heitz, V., Nierengarten, J.-F. & Sauvage, J.-P. in Transition Metals in Supramolecular Chemistry (eds Fabbrizzi, L. & Poggi, A.) 371–390 (Kluwer Academic, Dordrecht, 1994).
Ashton, P. R., Philp, D., Spencer, J. & Stoddart, J. F. J. chem. Soc., chem. Commun. 1677–1679 (1991).
LaBrenz, S. R. & Kelly, J. W. J. Am. chem. Soc. 117, 1655–1656 (1995).
Betz, S. F., Raleigh, D. P. & Degrado, W. F. Curr. Opin. struct. Biol. 3, 601–610 (1993).
Quinn, T. P., Tweedy, N. B., Richardson, J. S., Williams, R. W. & Richardson, D. C. Proc. natn. Acad. Sci. U.S.A. 91, 8747–8751 (1994).
Mutter, M. et al. J. Am. chem. Soc. 114, 1463–1470 (1992).
Ghadiri, M. R., Soares, C. & Choi, C. J. Am. chem. Soc. 114, 825–831 (1992).
Åkerfeldt, K. S., Kim, R. M., Comac, D., Groves, J. T., Lear, J. D. & DeGrado, W. F. J. Am. chem. Soc. 114, 9656–9657 (1992).
Hunter, C. A. & Sanders, J. R. M. J. Am. chem. Soc. 112, 5525–5534 (1990).
Hanna, M. W. & Ashbaugh, A. L. J. phys. Chem. 68, 811–816 (1964).
Deranleau, D. A. J. Am. chem. Soc. 91, 4044–4054 (1969).
Benniston, A. C., Harriman, A. & Lynch, V. M. Tetrahedron Lett. 35, 1473–1476 (1994).
Cantor, R. C. & Schimmel, P. R. Biophysical Chemistry Part II 390–408 (Freeman, New York, 1980).
Chudek, J. A., Foster, R. & Twiselton, D. R. J. chem. Soc. Perkin Trans. II 1385–1389 (1983).
Barlos, K. et al. Tetrahedron Lett. 30, 3943–3946 (1989).
Atherton, E. & Shepard, R. C. Solid Phase Peptide Synthesis (IRL, Oxford, 1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Scott Lokey, R., Iverson, B. Synthetic molecules that fold into a pleated secondary structure in solution. Nature 375, 303–305 (1995). https://doi.org/10.1038/375303a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/375303a0
This article is cited by
-
Numerical representations of AB-type copolymer complexes: analysis of 1H NMR chemical shift patterns in terms of a Smith–Cantor set
Journal of Mathematical Chemistry (2024)
-
Using supramolecular machinery to engineer directional charge propagation in photoelectrochemical devices
Nature Chemistry (2023)
-
Molecular Tetris by sequence-specific stacking of hydrogen bonding molecular clips
Communications Chemistry (2022)
-
Designing cooperatively folded abiotic uni- and multimolecular helix bundles
Nature Chemistry (2018)
-
Photomodulation of fluoride ion binding through anion-π interactions using a photoswitchable azobenzene system
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.