Abstract
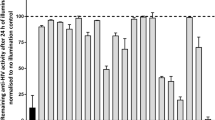
HERPES simplex viruses (HSV) types 1 and 2 encode their own ribonucleotide reductases (RNRs) (EC 1.17.4.1) to convert ribo-nucleoside diphosphates into the corresponding deoxyribo-nucleotides1. Like other iron-dependent RNRs, the viral enzyme is formed by the reversible association of two distinct homodimeric subunits2. The carboxy terminus of the RNR small subunit (R2) is critical for subunit association3,4 and synthetic peptides containing these amino-acid sequences selectively inhibit the viral enzyme by preventing subunit association4–9. Increasing evidence indicates that the HSV RNR is important for virulence and reactivation from latency10–14. Previously, we reported on the design of HSV RNR inhibitors with enhanced inhibitory potency in vitro4,15,16. We now report on BILD 1263, which to our knowledge is the first HSV RNR subunit-association inhibitor with antiviral activity in vivo. This compound suppresses the replication of HSV-1, HSV-2 and acyclovir-resistant HSV strains in cell culture, and also strongly potentiates the antiviral activity of acyclovir. Most importantly, its anti-herpetic activity is shown in a murine ocular model of HSV-1-induced keratitis, providing an example of potent non-substrate-based antiviral agents that prevent protein–protein interactions. The unique antiviral properties of BILD 1263 may lead to the design of new strategies to treat herpesvirus infections in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Reichard, P. A. Rev. Biochem. 57, 349–374 (1988).
Eriksson, S. & Sjöoberg, B. M. in Allosteric Enzymes (ed Hervé G. 189–215 (CRC Press, Boca Raton, 1989).
Filatov, D., Ingemarson, R., Graslund, A. & Thelander, L. J. biol. Chem. 267, 15816–15822 (1992).
Krogsrud, R. L., Welchner, E., Scouten, E. & Liuzzi, M. Analyt. Biochem. 213, 386–394 (1993).
Dutia, B. M., Frame, M. C., Subak-Sharpe, J. H. Clark, W. N. & Marsden, H. S. Nature 321, 439–441 (1986).
Cohen, E. A., Gaudreau, P., Brazeau, P. & Langelier, Y. Nature 321, 441–443 (1986).
Cosentino, G. et al. Biochem. cell. Biol. 69, 79–83 (1991).
Paradis, H., Gaudreau, P., Brazeau, P. & Langelier, Y. J. biol. Chem. 263, 16045–16050 (1988).
McClements, W. et al. Virology 162, 270–273 (1988).
Jacobson, J. G. et al. Virology 173, 276–283 (1989).
Cameron, J. M. et al. J. gen. Virol. 67, 2607–2612 (1988).
Brandt, C. R., Kintner, R. T., Visalli, R. J., Pumfrey, A. M. & Grau, D. R. J. gen. Virol. 72, 2043–2049 (1991).
Yamada, Y. et al. J. infect. Dis. 164, 1091–1097 (1991).
Idowu, A. D., Fraser-Smith, A. B., Poffenberg, K. L. & Herman, R. C. Antiviral Res. 17, 145–156 (1992).
Liuzzi, M. & Déziel, R., in The search for Antiviral Drugs (eds Adams, J. & Merluzzi, V. J.) 225–238 (Birkhauser, Boston, 1993).
Moss, N. et al. J. med. Chem. 36, 3005–3009 (1993).
Langlois, M., Allard, J. P., Nugier, F. & Aymard, M. J. biol. Standardization 14, 201–211 (1986).
Denizot, F. & Lang, R. J. immun. Meth. 89, 271–277 (1986).
Knipe, D. M. & Spang, A. E. J. Virol. 43, 314–324 (1982).
Chatis, P. A. & Crumpacker, C. S. Antimicrob. Agents Chemother. 36, 1589–1595 (1992).
Reardon, J. E. & Spector, T. Adv. Pharmac. 22, 1–27 (1991).
Coen, D. M. Antiviral Res. 15, 287–300 (1991).
Sacks, S. L. et al. Ann. int. Med. 111, 893–899 (1989).
Furman, P. A., Coen, D. M., St Clair, M. H. & Schaffer, P. A. J. Virol. 40, 936–941 (1981).
Coen, D. M. et al. Proc. natn. Acad. Sci. U.S.A. 86, 4736–4740 (1989).
Coen, D. M., Goldstein, D. J. & Weller, S. K. Antimicrob. Agents. Chemother. 33, 1395–1399 (1989).
Brandt, C. R., Coakley, L. M. & Grau, D. R. J. virol. Meth. 36, 209–222 (1992).
Brazeau, P. et al. Proc. natn. Acad. Sci. U.S.A. 79, 7909–7913 (1982).
Garrett, C. & Santi, D. V. Analyt. Biochem. 99, 268–273 (1979).
Sühnel, J. Antiviral Res. 13, 23–40 (1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liuzzi, M., Déziel, R., Moss, N. et al. A potent peptidomimetic inhibitor of HSV ribonucleotide reductase with antiviral activity in vivo. Nature 372, 695–698 (1994). https://doi.org/10.1038/372695a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/372695a0
This article is cited by
-
Targeting secret handshakes of biological processes for novel drug development
Frontiers in Biology (2016)
-
Amino acids special issue ‘Protein interactions in the virus–host relationship’
Amino Acids (2011)
-
Determination, by inductively coupled plasma mass spectrometry, of changes in cellular metal content resulting from herpes simplex virus-1 (HSV-1) infection
Analytical and Bioanalytical Chemistry (2007)
-
Antiherpesvirus drugs: a promising spectrum of new drugs and drug targets
Nature Reviews Drug Discovery (2003)
-
Herpes simplex virus helicase-primase inhibitors are active in animal models of human disease
Nature Medicine (2002)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.