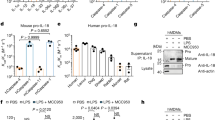
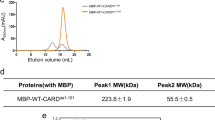
Abstract
Interleukin-lβ converting enzyme (ICE) processes an inactive precursor to the proinflammatory cytokine, interleukin-lβ, and may regulate programmed cell death in neuronal cells. The high-resolution structure of human ICE in complex with an inhibitor has been determined by X-ray diffraction. The structure confirms the relationship between human ICE and cell-death proteins in other organisms. The active site spans both the 10 and 20K subunits, which associate to form a tetramer, suggesting a mechanism for ICE autoactivation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Black, R. A., Kronheim, S. R. & Sleath, P. R. FEBS Lett. 247, 386–390 (1989).
Kostura, M. J. et al. Proc. natn. Acad. Sci. U.S.A. 86, 5227–5231 (1989).
Sleath, P. R., Hendrickson, R. C., Kronheim, S. R., March, C. J. & Black, R. A. J. biol. Chem. 265, 14526–14528 (1990).
Howard, A. D. et al. J. Immun. 147, 2964–2969 (1991).
March, C. J. Nature 315, 641–647 (1985).
Singer, I. I. et al. J. exp. Med. 167, 389–407 (1988).
Thornberry, N. A. et al. Nature 356, 768–774 (1992).
Cerretti, D. P. et al. Science 256, 97–100 (1992).
Yuan, J., Shaham, S., Ledoux, S., Ellis, H. M. & Horvitz, H. R. Cell 75, 641–652 (1993).
Miura, M., Zhu, H., Rotello, R., Hartweig, E. A. & Yuan, J. Cell 75, 653–660 (1993).
Nett-Fiordalisi, M. A., Berson, D. R. & Chaplin, D. D. J. Cell Biochem. 17B, 117 (1993).
Ray, C. A. et al. Cell 69, 597–604 (1992).
Gagliardini, V. et al. Science 263, 826–828 (1994).
Marx, J. & Barinaga, M. Science 259, 760–762 (1993).
Chapman, K. T. Biorg. med. Chem. Lett. 2, 613–618 (1992).
Miller, D. K. et al. Ann. NY Acad. Sci. 696, 133–148 (1993).
Kamphuis, I. G., Drenth, J. & Baker, E. N. J. molec. Biol. 182, 317–329 (1985).
Nett, M. A. et al. J. Immun. 149, 3254–3259 (1992).
Shivers, B. D., Giegel, D. A. & Keane, K. M. J. Cell Biochem. 17B, 119 (1993).
Polgar, L. Eur. J. Biochem. 33, 104–109 (1973).
Baker, E. N. & Drenth, J. in Biological Macromolecules and Assemblies Vol. 3 (eds Jurnak, F. & McPherson, A.) 312–368 (Wiley, New York, 1987).
Menard, R. et al. Biochemistry 30, 8924–8928 (1991).
Menard, R. et al. Biochemistry 30, 5531–5538 (1991).
Miller, S., Lesk, A. M., Janin, J. & Chothia, C. Nature 328, 834–836 (1987).
Bender, P. E. & Lee, J. C. A. Rep. med. Chem. 25, 185–193 (1989).
Dinarello, C. A. Immun. Rev. 127, 119–146 (1992).
Harris, E. D. Jr New Engl. J. Med. 322, 1277–1289 (1990).
Mandrup-Poulsen, T. et al. Cytokine 5, 185–191 (1993).
Hammerburg, C. et al. J. clin. Invest. 90, 571–583 (1992).
Noronha, I. L., Kruger, C., Andrassy, K., Ritz, E. & Waldherr, R. Kidney Int. 43, 682–692 (1993).
Dinarello, C. A., Gelfand, J. A. & Wolff, S. M. J. Am. med. Ass. 269, 1829–1835 (1993).
Geiger, T. et al. Clin. exp. Rheumatol. 11, 515–522 (1993).
Ohlsson, K., Bjork, P., Bergenfeldt, M., Hageman, R. & Thompson, R. C. Nature 348, 550–552 (1990).
Wooley, P. H. et al. Arthritis Rheumatol. 36, 1305–1314 (1993).
Sandberg, J-O., Eizirik, D. L., Sandler, S., Tracey, D. E. & Andersson, A. Diabetes 42, 1845–1851 (1993).
Griffin, W. S. T. et al. Proc. natn. Acad. Sci. U.S.A. 86, 7611–7615 (1989).
Breitner, J. C. S. et al. Neurology 44, 227–232 (1994).
CCP4-Collaborative Computing Project No. 4 A Suite of Programs for Protein Crystallography (Daresbury Laboratory, Warrington, 1979).
Quanta version 4.0b (Molecular Simulations Inc., Burlington MA, 1992).
Brünger, A. T. Acta crystallogr. A46, 46–57 (1990).
Brünger, A. T., Krukowski, A. & Erickson, J. Acta crystallogr. A46, 585–593 (1990).
Blundell, T. L. & Johnson, L. N. Protein Crystallography (Academic, New York, 1976).
Carson, M. J. appl. Crystallogr. 24, 958–961 (1991).
Eklund, H. J. molec. Biol. 102, 27 (1976).
Holmgren, A., Soderberg, B.-O., Eklund, H. & Branden, C.-I. Proc. natn. Acad. Sci. U.S.A. 72, 2305–2309 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wilson, K., Black, JA., Thomson, J. et al. Structure and mechanism of interleukin-lβ converting enzyme. Nature 370, 270–275 (1994). https://doi.org/10.1038/370270a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/370270a0
This article is cited by
-
Extracellular vesicles derived from mesenchymal stem cells — a novel therapeutic tool in infectious diseases
Inflammation and Regeneration (2023)
-
A Review on Caspases: Key Regulators of Biological Activities and Apoptosis
Molecular Neurobiology (2023)
-
Caspases in Alzheimer’s Disease: Mechanism of Activation, Role, and Potential Treatment
Molecular Neurobiology (2023)
-
NaCl exposure results in increased expression and processing of IL-1β in Meniere’s disease patients
Scientific Reports (2022)
-
Generic nature of the condensed states of proteins
Nature Cell Biology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.