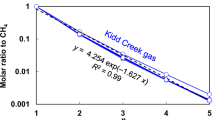
Abstract
THE idea that natural gas is formed by thermal decomposition of sedimentary organic matter1,2 enjoys almost universal acceptance3–6. But pyrolysis experiments on organic matter7–13 have failed to reproduce the composition of natural gas (typically 90% methane). It has recently been suggested14 that natural gas may instead be generated catalytically: transition metals are often found in carbonaceous sedimentary rocks, and might promote the reaction between hydrogen and n-alkenes (which are themselves formed during thermal decomposition of kerogen) to give light hydrocarbons and natural gas. We report here experimental results that support this hypothesis. In particular, we find that n-alkenes, hydrogen and a carbonaceous sedimentary rock containing moderately high concentrations of transition metals react under mild conditions (∼200 °C) to generate a light-hydrocarbon product indistinguishable from natural gas in both molecular and carbon isotope composition. Our results demonstrate that the reaction is indeed catalytic, and could alter the way in which we view the generation and distribution of oil and gas in the Earth.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Schoell, M. Geochim. cosmochim. Acta. 44, 649–661 (1980).
James, A. T. Bull. Am. Ass. Petrol. Geol. 67, 1176–1191 (1983).
Tissot, B. P. & Welte, D. H. Petroleum Formation and Occurence (Springer, New York, 1984).
Takach, N. E., Barker, C. & Kemp, M. K. Bull. Am. Ass. Petrol. Geol. 71, 322–333 (1987).
Barker, C. Bull. Am. Ass. Petrol. Geol. 74, 1254–1261 (1990).
Ungerer, P. Org. Geochem. 16, 1–25 (1990).
Evans, R. J. & Felbeck, G. T. Jr Org. Geochem. 4, 135–144 (1983).
Saxby, J. D. & Riley, K. W. Nature 308, 177–175 (1984).
Espitalie, J., Ungerer, P., Irwin, I. & Marquis, F. Org. Geochem. 13, 893–899 (1987).
Horsfield, B., Schenk, H. J., Mills, N. & Welte, D. H. Org. Geochem. 19, 191–204 (1991).
McNab, J. G., Smith, P. V. & Betts, R. L. Petrol. Engr. Chem. 44, 2556–2563 (1952).
Fabuss, B. M., Smith, J. O., Lait, R. I., Borsanyi, A. S. & Satterfield, C. N. Ind. Engr. Chem., Proc. Dev. Div. 1, 293–299 (1962).
Appleby, W. G., Avery, W. H. & Meerbott, W. K. J. Am. chem. Soc. 69, 2279–2285 (1947).
Mango, F. D. Geochim. cosmochim. Acta. 56, 553–555 (1992).
Laidler, K. J., Sagert, N. H. & Wojciechowske, B. W. Proc. R. Soc. A270, 242–253 (1962).
Lewan, M. D., Winters, J. C. & McDonald, J. H. Science 203, 897–899 (1979).
Tegelaar, E. W. et al. J. analyt. appl. Pyrolysis 15, 29–54 (1989).
Derenne, S., Largeau, C., Casadevall, E., Berkaloff, C. & Rousseau, B. Geochim. cosmochim. Acta. 55, 1041–1050 (1991).
Harwood, R. J. Bull Am. Ass. Petrol. Geol. 61, 2082–2102 (1977).
Shock, E. L. Geology 16, 889–890 (1988).
James, A. T. Bull. Am. Ass. Petrol. Geol. 74, 1441–1458 (1990).
Fuex, A. N. J. Geochem. Explor. 7, 155–188 (1977).
Price, L. C. & Wenger, L. M. Org. Geochem. 19, 141–159 (1992).
Lewan, M. D. Phil. Trans. R. Soc. A315, 123–1134 (1985).
Monthioux, M., Landais, P. & Monin, J. Org. Geochem. 8, 275–292 (1985).
Hamak, J. E. & Sigler, S. Information Circular No. 9301 (Bureau of Mines, US Dep. Interior, Pittsburgh, 1991).
Goebel, E. D., Coveney, R. M. Jr, Angino, E. E., Zeller, E. J. & Dreschhoff, G. A. M. Oil Gas. J. 5 May, 215–222 (1984).
Quigley, T. M. & Mackenzie, A. S. Nature 333, 549–552 (1988).
Sweeney, J. J., Burnham, A. K. & Braun, R. L. Bull. Am. Ass. Petrol. Geol. 71, 967–985 (1987).
Price, L. C., Clayton, J. L. & Rumen, L. L. Trans. Gulf. Coast Ass. Geol. Soc. 29, 352–370 (1979).
Price, L. Geochim. cosmochim. Acta 57, 3261–3280 (1993).
Mango, F. D. Nature 352, 146–148 (1991).
Isaacs, C. M. et al. Open-File Report 92-539-C (US Geological Survey, Menlo Park, California, 1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mango, F., Hightower, J. & James, A. Role of transition-metal catalysis in the formation of natural gas. Nature 368, 536–538 (1994). https://doi.org/10.1038/368536a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/368536a0
This article is cited by
-
Weathered and Volcanic Provenance-Sedimentary System and Its Influence on Reservoir Quality in the East of the Eastern Depression, the North Yellow Sea Basin
Journal of Earth Science (2018)
-
Hydrothermal history in the eastern margin of Tunisia: inferred magmatic rocks alterations, new paragenesis and associated gas occurrences
Arabian Journal of Geosciences (2015)
-
Trace-gas metabolic versatility of the facultative methanotroph Methylocella silvestris
Nature (2014)
-
Kinetics of N–S Atom Exchange in the Pyrrole–H2S System
Arabian Journal for Science and Engineering (2014)
-
Low-temperature gas from marine shales
Geochemical Transactions (2009)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.