Abstract
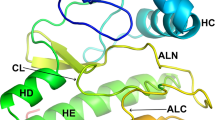
The structure of the MAP kinase ERK2, a ubiquitous protein kinase target for regulation by Ras and Raf, has been solved in its unphosphorylated low-activity conformation to a resolution of 2.3 A. The two domains of unphosphorylated ERK2 are farther apart than in the active conformation of cAMP-dependent protein kinase and the peptide-binding site is blocked by tyrosine 185, one of the two residues that are phosphorylated in the active enzyme. Activation of ERK2 is thus likely to involve both global and local conformational changes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Boulton, T. G. et al. Science 249, 64–67 (1990).
Cobb, M. H., Boulton, T. G. & Robbins, D. J. Cell Regulation 2, 965–978 (1991).
Anderson, N. G., Maller, J. L., Tonks, N. K. & Sturgill, T. W. Nature 343, 651–653 (1990).
Pulverer, B. J., Kyriakis, J. M., Avruch, J., Nikolakaki, E. & Woodgett, J. R. Nature 353, 670–674 (1991).
Gille, H., Sharrocks, A. D. & Shaw, P. E. Nature 358, 414–416 (1992).
Takishima, K., Griswold-Prenner, I., Ingebritsen, T. & Rosner, M. R. Proc. natn. Acad. Sci. U.S.A. 8, 2520–2524 (1991).
Northwood, I. C., Gonzalez, F. A., Wartmann, M., Raden, D. L. & Davis, R. J. J. biol. Chem. 266, 15266–15276 (1991).
Tsao, H., Aletta, J. M. & Greene, L. A. J. biol. Chem. 265, 15471–15480 (1990).
Ray, L. B. & Sturgill, T. W. Proc. natn. Acad. Sci. U.S.A. 84, 1502–1506 (1987).
Sturgill, T. W., Ray, L. B., Erikson, E. & Maller, J. L. Nature 334, 715–718 (1988).
Lee, R.-M., Cobb, M. H. & Blackshear, P. J. J. biol. Chem. 267, 1088–1092 (1992).
Schaap, D., van der Wal, J., Howes, L. R., Marshall, C. J. & van Blitterswijk, W. J. J. biol. Chem. 268, 20232–20236 (1993).
Lange-Carter, C. A., Pleiman, C. M., Gardner, A. M., Blumer, K. J. & Johnson, G. L. Science 260, 315–319 (1993).
Moodie, S. A., Willumsen, B. M., Weber, M. J. & Wolfman, A. Science 260, 1658–1661 (1993).
Robbins, D. J. et al. in Advances in Cancer Research Vol. 63, 93–116 (Academic, San Diego, 1994).
Sontag, E. et al. Cell 75, 887–897 (1993).
Drewes, G. et al. EMBO J. 11, 2131–2138 (1992).
Ahn, N. G. et al. J. biol. Chem. 266, 4220–4227 (1991).
Crews, C. M., Alessandrini, A. & Erikson, R. L. Science 258, 478–480 (1992).
Robbins, D. J. et al. J. biol. Chem. 268, 5097–5106 (1993).
Seger, R. et al. Proc. natn. Acad. Sci. U.S.A. 88, 6142–6146 (1991).
Rossomando, A. J. et al. Proc. natn. Acad. Sci. U.S.A. 89, 5779–5783 (1992).
Boulton, T. G. & Cobb, M. H. Cell Regulation 2, 357–371 (1991).
Zheng, C.-F. & Guan, K.-L. J. biol. Chem. 268, 16116–16119 (1993).
Alessi, D. R., Smythe, C. & Keyse, S. M. Oncogene 8, 2015–2020 (1993).
Sun, H., Charles, C. H., Lau, L. F. & Tonks, N. K. Cell 75, 487–493 (1993).
Zhang, F., Robbins, D. J., Cobb, M. H. & Goldsmith, E. J. J. molec. Biol. 233, 550–552 (1993).
Ringe, D. & Petsko, G. A. Prog. Biophys. molec. Biol. 45, 197–235 (1985).
Knighton, D. R. et al. Science 253, 407–413 (1991).
Taylor, S. S. et al. Trends biochem. Sci. 18, 84–89 (1993).
De Bondt, H. L. et al. Nature 363, 595–602 (1993).
Hanks, S. K., Quinn, A. M. & Hunter, T. Science 241, 42–52 (1988).
Hanks, S. K., Curr. Opin. Struct. Biol. 1, 369–383 (1991).
Karlsson, R., Zheng, J., Xuong, N.-H., Taylor, S. S. & Sowadski, J. M. Acta crystallogr. D49, 381–388 (1993).
Bossemeyer, D., Engh, R. A., Kinzel, V., Ponstingl, H. & Huber, R. EMBO J. 12, 849–859 (1993).
Knighton, D. R. et al. Science 253, 414–420 (1991).
Clark-Lewis, I., Sanghera, J. S. & Pelech, S. L. J. biol. Chem. 266, 15180–15184 (1991).
Haycock, J. W., Ahn, N. G., Cobb, M. H. & Krebs, E. G. Proc. natn. Acad. Sci. U.S.A. 89, 2365–2369 (1992).
Gonzalez, F. A., Raden, D. L. & Davis, R. J. J. biol. Chem. 266, 22159–22163 (1991).
Hurley, J. H. et al. Proc. natn. Acad. Sci. U.S.A. 86, 8635–8639 (1989).
Sprang, S. R., Withers, S. G., Goldsmith, E. J., Fletterick, R. J. & Madsen, N. B. Science 254, 1367–1371 (1991).
Schulz, G. E. Curr. Opin. Struct. Biol. 1, 883–888 (1991).
Perutz, M. F. Nature 228, 726–739 (1970).
Bennett, W. S. Jr & Steitz, T. A. Proc. natn. Acad. Sci. U.S.A. 75, 4848–4852 (1978).
Howard, A. J., Nielsen, C. & Xuong, N-H. Meth. Enzym. 114, 452–4782 (1985).
Steigemann, W. PROTEIN, A Program System for the Crystal Structure Analysis of Proteins (Max-Planck-Institute für Biochimie, Martinsried, Germany, 1989).
Wang, B.-C. in Meth. Enzym. (eds Hirs, C. H. & Wyckoff, H. W.) Vol. 115, 90–112 (Academic, New York, 1985).
Jones, T. A. J. appl. Crystallogr. 11, 268–272 (1978).
Jones, T. A. Zou, J.-Y. & Cowan, S. W. Acta crystallogr. A47, 110–119 (1991).
Brünger, A. T., Kuriyan, J. & Karplus, M. Science 235, 458–460 (1987).
Carson, M. J. molec. Graph. 5, 103–106 (1987).
Boulton, T. G. et al. Cell 65, 663–675 (1991).
Tsai, L.-H. & Meyerson, M. Nature 353, 174–177 (1991).
Uhler, M. D. et al. Proc. natn. Acad. Sci. U.S.A. 83, 1300–1304 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhang, F., Strand, A., Robbins, D. et al. Atomic structure of the MAP kinase ERK2 at 2.3 Å resolution. Nature 367, 704–711 (1994). https://doi.org/10.1038/367704a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/367704a0
This article is cited by
-
Uncoupling p38α nuclear and cytoplasmic functions and identification of two p38α phosphorylation sites on β-catenin: implications for the Wnt signaling pathway in CRC models
Cell & Bioscience (2023)
-
A non-catalytic herpesviral protein reconfigures ERK-RSK signaling by targeting kinase docking systems in the host
Nature Communications (2022)
-
ERK signalling: a master regulator of cell behaviour, life and fate
Nature Reviews Molecular Cell Biology (2020)
-
Tyrosine nitration of human ERK1 introduces an intra-hydrogen bond by molecular dynamics simulations
Structural Chemistry (2019)
-
Analysis of crystal structure of Arabidopsis MPK6 and generation of its mutants with higher activity
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.